Current Concepts in Surgical Management of Neck Metastases from Head and Neck Cancer
This article will address modified, selective, and radical neck dissection as well as other surgical considerations, and will review the surgical techniques currently available for neck treatment.
The surgical management of neck metastases from head and neck cancer consists primarily of neck dissection. An awareness of both the cervical anatomy and natural history of squamous cell carcinoma of the upper aerodigestive tract is necessary to understand the role of neck dissection and to appreciate when it may be appropriate to modify the standard radical neck dissection. The use of imaging to augment palpation of the neck has resulted in greater accuracy in identifying neck metastases and has further clarified the role of modified neck dissections. Depending on the site and size of the primary tumor, radiation therapy, modified neck dissection, or selective neck dissection are all appropriate options for the elective treatment of the neck when there is a high risk of occult metastasis. Therapeutic neck dissection alone is adequate for early-stage neck disease, but must be combined with irradiation for more advanced stages. Regional control of tumor metastases is highly dependent on the stage of neck disease and the presence of extranodal cancer spread.
Introduction
Radical neck dissection, first described by Crile in 1906 [1], is the standard against which all surgical approaches to neck metastases is compared. Consideration was subsequently given to more conservative surgical techniques, but they did not gain widespread acceptance and were condemned by Martin et al in the 1950s [2].
Surgical management of the neck has continued to evolve toward more conservative approaches, however. Even selective neck dissection techniques are now widely accepted. Current controversies center around the appropriate use of modified and selective neck dissections, indications for elective neck treatment, and selection of the best therapeutic modality for elective neck treatment. This article will address these as well as other surgical considerations, and will review the surgical techniques currently available for neck treatment.
Anatomic Considerations
A neck dissection is, in fact, an anatomic dissection of the neck. Therefore, a clear grasp of the anatomy of the cervical lymph node groups and cervical aponeurotic system is essential for proper understanding of neck dissection.
FIGURE 1
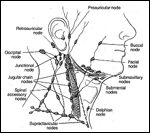
Lymph node groups are most commonly referred to by the names in this illustration.FIGURE 2
Lymph node groups are subdivided for ease of communication into levels as demonstrated by this figure.
Cervical Lymph Node Groups
There are approximately 150 to 350 lymph nodes above the clavicles. Since the lymphatic system is embryologically related to the venous system, the lymph nodes are closely related to veins. The cervical lymph nodes have commonly been divided into surgical levels to facilitate discussions of patterns of metastases and types of neck dissection (Figures 1 and 2).
The submandibular and submental nodes constitute the level I group. The submental lymph nodes are located in the midline within the triangle defined by the anterior bellies of the digastric muscles and the hyoid bone. The submandibular nodes are situated within a triangle bounded by the anterior and posterior bellies of the digastric muscle and the mandibular body. These nodes are in close relationship to the submandibular gland and the posterior facial vein. Notably, the gland itself does not include any lymph nodes.
The internal jugular vein lymph nodes are limited anteriorly by the lateral margin of the sternohyoid muscle and posteriorly by the posterior border of the sternocleidomastoid muscle, and are subdivided into three groups. The level II group, or upper internal jugular nodes, extend from the skull base superiorly to the hyoid bone or carotid bifurcation inferiorly. These nodes , also commonly referred to as jugulodigastric nodes, are closely related to the superior portion of the spinal accessory nerve as well. The middle jugular group (level III group) continues inferiorly to the omohyoid muscle. The lower jugular group (level IV) nodes extend from the omohyoid muscle to the level of the clavicle.
The level V group, or posterior triangle lymph nodes, are bounded by the posterior edge of the sternocleidomastoid muscle, anterior border of the trapezius muscle, and clavicle. Superiorly, these nodes closely approximate the spinal accessory nerve, and are often referred to as the spinal accessory chain. The nodes that are located near the confluence of the posterior triangle and upper jugular groups are sometimes termed junctional nodes. The supraclavicular nodes are part of the posterior triangle group.
The anterior compartment group (level VI) is bounded superiorly by the hyoid bone, inferiorly by the sternum, and laterally by the common carotid arteries. Included in this group are the paratracheal, pretracheal, precricoid (Delphian), and tracheoesophageal groove nodes.
Additional groups that are sometimes dissected outside the normally described boundaries of the neck include the facial, parotid, preauricular, retroauricular, suboccipital, and retropharyngeal nodes.
FIGURE 3

The contents of the neck are divided into several spaces by a variety of fascial interconnections. These spaces and fascial layers are demonstrated in this figure.
Cervical Aponeurotic System
The cervical aponeurotic system divides the neck into compartments, including the lateral and paravisceral spaces, that are removed with a neck dissection (Figure 3). The superficial cervical fascia is a thin layer that invests the platysma muscle. The deep cervical fascia is subdivided into three layers: investing (superficial), pretracheal (middle), and prevertebral (deep).
The investing layer completely encircles the neck, splitting to enclose the sternocleidomastoid, omohyoid, and trapezius muscles. Superiorly, the investing fascia splits to enclose the subman- dibular and parotid glands, and attaches to the lower border of the mandible. The pretracheal layer encompasses the trachea, thyroid gland, and esophagus.
The prevertebral layer covers the prevertebral, scalene, levator scapulae, splenius, and semispinalis capitis muscles, and constitutes the floor of the lateral neck. The phrenic nerve, brachial plexus, sympathetic trunk, and dorsal scapular nerve all lie deep to the prevertebral fascia.
The carotid sheath consists of all three layers of deep cervical fascia and contains the carotid artery and vagus nerve. The jugular vein is located within an extension of the carotid sheath as well.
Critical to an understanding of modified and selective neck dissections is the concept that the lymph nodes lie within the lateral and paravisceral spaces, not within the fascial wrappings of the muscles or the carotid artery. The lymph nodes may, however, be in very close association with these fascial layers. The spinal accessory nerve must be uniquely considered in neck dissection, as it actually travels through the lateral space of the neck just deep to the investing fascia and superficial to the prevertebral fascia.
Tumor Biology
Squamous cell carcinoma of the upper aerodigestive tract is the most common tumor to metastasize to the cervical nodes. Less commonly, neck dissection is performed for malignant neoplasms of the thyroid gland, parotid gland, or skin. Distant metastases from below the clavicle occur, but neck dissection is generally of no benefit in the management of these neoplasms.
Once a metastasis reaches a lymph node, tumor growth results in an indurated and more rounded node. Increasing size of the lymph node is directly associated with increased risk of tumor invasion through the capsule, and eventually, fixation to adjacent structures.
The risk of cervical metastases from different primary sites and stages has been estimated by examining the percentage of patients with similar stage tumors who present with clinically positive node [3], the distribution of pathologically positive nodes in an electively dissected, clinically negative neck, and failure rates in untreated necks. In this fashion, the relative risk of cervical metastases may be approximated for specific sites. For example, the glottis has a low risk of metastasizing to the neck(< 20%), whereas the nasopharynx has a high risk (> 30%). Finally, lesions such as early supraglottic laryngeal tumors are considered to be of intermediate risk (20% to 30%) [4].
In many cases, these fairly consistent patterns of metastases allow for the application of selective removal of nodal groups for specified lesions. There are, however, situations in which the normal pattern of nodal metastases do not hold. For example, the right and left sides of the cervical lymphatic system normally do not shunt. However, previous neck surgery, prior irradiation, or multiple positive nodes obstructing the lymphatic system may alter the patterns of cervical metastases such that shunting across the submental region occurs, or unusual nodal groups are involved for a given primary. Furthermore, recurrent lesions are associated with a higher risk of cervical metastasis.
Diagnosis
Palpation of the neck for metastases, by itself, may be associated with significant error rates. Therefore, CT is used extensively to image both the primary site and neck, and may increase the accuracy of neck staging from 70% to 93% [5]. Computed tomography is especially useful in detecting occult nodal disease, determining the presence or absence of extranodal disease, demonstrating the resectability of extensive nodal disease, and evaluating inaccessible areas, such as the parapharyngeal space, retropharyngeal space, and tracheoesophageal groove (Figure 4).
Although MRI can detect nodal disease as well [6], we currently limit its use to selected primary sites, such as the nasopharynx, or to those cases in which a specific issue, such as perineural infiltration, needs to be addressed. Ultrasound is used more extensively in Europe than in the United States to evaluate nodal disease. Magnetic resonance imaging and ultrasound may be helpful in determining nodal fixation to the carotid artery, but no imaging method is definitive in this regard.
Selection of Treatment
The choice between irradiation or dissection for treatment of the neck is based on the treatment modality planned for the primary site and the clinical status of the neck. More advanced neck disease is usually managed with combined therapy.
Management of the Clinically Negative Neck
Traditionally, elective neck treatment has been utilized to manage tumors with a high risk (> 20% to 30%) of occult metastatic disease because of the improved neck control associated with this strategy [7]. The alternative is to adopt a "wait and see" approach, which may be associated with an increased rate of distant metastases and a worse prognosis, even if the neck failure is successfully salvaged [8,9]. However, it has been recently suggested that elective neck treatment may no longer be justified, since the use of CT and MRI to assess the neck should decrease the risk of occult disease to approximately 12% [10].
Because the morbidity of a modified radical neck dissection or elective neck irradiation is minimal, follow-up is simplified, and the cost of delivering the treatment in one instance is lower than that of an additional treatment period for a neck failure, we continue to advocate elective neck treatment in patients with a high risk of occult cervical metastases. An interesting use of decision analysis tools by Weiss et al further supported the elective management of the neck with either dissection or irradiation for those tumors at high risk for occult metastases [11].
TABLE 1

AJCC Staging System for Regional Lymph Node Metastases in Head and Neck Cancer, 1992
Radiation and surgery can achieve similar regional control rates for N0 neck (Table 1) disease [9], and it is desirable to use only one therapeutic modality when possible to save the other for either recurrence or a second primary tumor. Therefore, when the primary tumor is to be treated with surgery, an elective neck dissection is indicated for primary sites with a greater than 20% risk of cervical metastasis. If, on the other hand, the primary site is to be treated with radiation, elective neck irradiation is indicated when the risk of cervical metastasis exceeds 20%.
Management of the Clinically Positive Neck
If the primary tumor is to be managed surgically, dissection is indicated for clinically positive necks. Preoperative irradiation is rarely used, except when a node is fixed to the carotid artery or deep cervical fascia. The node will usually become resectable, and in many instances, no viable tumor is found in the neck dissection pathologic specimen. When the primary site is to be irradiated, radiation alone is typically adequate for small N1 (1 to 2 cm) disease, especially for nasopharyngeal tumors. More advanced neck disease, particularly when regression is incomplete, requires a neck dissection 4 to 6 weeks after the completion of radiation therapy in order to improve regional control [12].
FIGURE 5

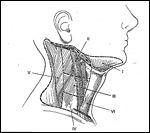
The outlined area demonstrates the extent of the section and the structures removed in a radical neck dissection. The structures removed include the stemocleidomastoid muscle, the spinal accessory nerve, and the internal jugular vein.
Selection of Dissection Technique
The terminology for describing neck dissections has become quite confusing since the initial description of the "classic" radical neck dissection in 1906 [1]. An understanding of the classification of neck dissections is essential for selecting the appropriate type of neck dissection and for comparing treatment results. The description of neck dissections that follows is useful for such purposes [13].
FIGURE 6
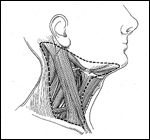
The outlined area demonstrates the extent of dissection in a modified radical neck dissection. The extent of dissection is simiar to that of a radical neck dissection, but the stemocleidomastoid muscle, the spinal accessory nerve, and the internal jugular vein are preserved.FIGURE 7

The outlined area demonstrates the extent of a supraomohyoid neck dissection. This is a form of a selective neck dissection, which would most commonly be used for tunors of the tongue and floor of mouth. The outlined area is included in a lateral neck dissection, which would most commonly be used for tumors of the larynx.FIGURE 8
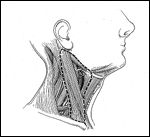
The outlined area is that included in a lateral neck dissection, which would most commonly be used for tunors of the larynx.
Radical neck dissection entails the removal of lymph node levels I to V in continuity with the spinal accessory nerve, sternocleidomastoid muscle, and internal jugular vein (Figure 5). A modified radical neck dissection involves the removal of the same lymph node groups with preservation of one or more of the nonlymphatic structures (Figure 6). A selective neck dissection describes a cervical lymphadenectomy, in which one or more of the lymph node groups is not removed. Supraomohyoid and lateral neck dissections are selective neck dissections that remove nodal levels I to III and II to IV, respectively (Figures 7 and 8). Finally, an extended neck dissection includes the removal of lymph node groups or nonlymphatic structures not encompassed by a radical neck dissection.
The choice of a neck dissection is based on the primary site as well as the number, size, and location of positive lymph nodes. In addition, the results and morbidity associated with each type of neck dissection must be considered.
Radical vs Modified Radical Dissection
We prefer a modified radical neck dissection over a radical neck dissection in any situation in which equal regional control rates may be obtained. The removal of the spinal accessory nerve, sternocleidomastoid muscle, and internal jugular vein in a radical neck dissection is associated with trapezius muscle dysfunction, alteration of the neck contour, and venous engorgement. Furthermore, bilateral modified neck dissection may be performed simultaneously rather than in a staged fashion, as with a radical neck dissection.
Multiple authors advocate a modified radical neck dissection for N1 disease [14-16]. Since no lymph nodes are contained within the fascial wrappings of the sternocleidomastoid muscle or carotid sheath, all lymph nodes may be removed with a modified neck dissection, unless extensive extranodal spread and/or fixation are present. These are, in fact, our only indications for a radical neck dissection, as suggested by Bocca and Pignaturo [17].
Selective Neck Dissection
The advantage of a selective neck dissection is that it provides the same therapeutic benefit as a comprehensive neck dissection, with potentially less morbidity and decreased operative time. Cases must be carefully evaluated to ensure that they are appropriate for a selective dissection, however.
Use of selective neck dissection is based on the concept that metastases from a specific primary site occur in a predictable fashion. It is indicated for N0 or N1 neck disease associated with the primary sites described below. In the setting of more advanced neck disease or after previous treatment, lymphatic drainage is no longer predictable, and a comprehensive neck dissection is indicated.
The most commonly employed selective neck dissection is the supraomohyoid dissection for clinically negative necks associated with oral cavity primaries. Lateral neck dissection is best suited for laryngeal, oropharyngeal, and hypopharyngeal primaries [18]. If significant unexpected lymph node metastases are encountered during a selective neck dissection, the dissection should be converted to a comprehensive neck dissection due to the unpredictable pattern of metastases in that event.
Extended Neck Dissection
Extended neck dissection is indicated for removal of the following specific nodal groups associated with selected primary sites: (1) anterior compartment nodes (level VI) with thyroid gland, esophageal, and subglottic laryngeal primaries; (2) facial nodes for lip and mid-face skin primaries; (3) periparotid and preauricular nodes for skin lesions of the temple, cheek, and anterior auricle; and (4) retroauricular and suboccipital nodes for posterior scalp and posterior auricular lesions.
A variation of an extended neck dissection is the inclusion of a retropharyngeal node dissection for pharyngeal tumors that have a particularly high incidence of nodal metastases to that area. The survival benefit of this dissection, however, has not been proven [19,20].
Indications for Postoperative Irradiation
The decision to use postoperative irradiation in head and neck cancer is based on the initial tumor stage and pathologic findings. Both the primary site and neck must be considered in this regard.
Specific to the neck, postoperative irradiation is indicated for extranodal tumor spread or multiple positive nodes, especially at multiple levels. In addition, irradiation is useful for controlling the opposite, undissected neck when it is at risk and the primary site will need postoperative irradiation.
Management of Neck Metastases From an Unknown Primary
Cervical metastases arise overwhelmingly from upper aerodigestive tract squamous cell carcinoma primary sites, although metastases that appear first in the lower portion of the neck may be of infraclavicular origin. When upper- or middle-third cervical metastases are present without an obvious primary site, we recommend a thorough search for that site, including imaging and endoscopy of the upper aerodigestive tract. If no primary is found, histopathology of the lymph node is almost always obtainable by means of fine-needle aspiration cytology.
It is rarely necessary to perform a cervical node excision for diagnostic purposes. If a node excision is required, or is inadvisably performed, radiation must be the next modality utilized in order to achieve a reasonable rate of regional control [21]. This approach may, however, unduly complicate the management of the patient.
Results of Treatment
Survival rates in head and neck cancer vary widely, due to the large number of primary sites and different primary tumor sizes. However, various factors have been examined relative to their influence on the control of regional metastases by neck dissection. Extranodal spread of tumor, which increases in proportion to lymph node size, has been identified as a very important factor affecting regional control and survival [22-24]. An increased risk of distant metastases and a decrease in survival have been demonstrated with an increasing number of positive lymph nodes, particularly when located in the lower neck [22,25,26].
Regional control rates may also be examined with regard to treatment selection. Regional control rates for negative necks or necks with a single, positive node less than 3 cm managed with any type of neck dissection should be 90% or greater [12]. Several retrospective studies have demonstrated no difference in nodal recurrence rates between modified and radical neck dissections for appropriately selected patients [15,27,28].
Similar control rates (90% or greater) may be obtained with irradiation for N0 or early N1 neck disease [29]. For more advanced nodal disease, control rates are improved with a combination of neck dissection and radiation, as compared with either modality alone [12,30].
The salvage rate for recurrence in a previously dissected or previously irradiated neck is low, ranging from 5% to 40%, and salvage appears to be more likely if irradiation has not been previously employed [30,31].
Surgical Considerations
Prevention of Carotid Artery Rupture
Major vascular complications of neck dissection, especially carotid artery rupture, are often catastrophic. In an isolated neck dissection, carotid rupture is an extremely rare event. Carotid artery exposure occurs almost exclusively after the development of a pharyngocutaneous or orocutaneous fistula. Carotid exposure appears to be much less common after modified radical neck dissection, due to the protection provided by the sternocleidomastoid muscle.
Proper skin flap thickness and the placement of skin incisions away from the carotid bifurcation are helpful in preventing carotid exposure. In addition, the adventitia should be left on the carotid, unless there is a positive node partially fixed to the artery.
Once the outer layer of the carotid is exposed, it desiccates and weakens, most commonly at the level of the carotid bifurcation. Full-thickness destruction occurs within approximately 1 to 2 weeks. The first sign of an impending carotid rupture may be a sentinel bleed, which is then usually followed by an actual rupture 24 to 48 hours later. If rupture occurs, the common, internal, and external carotid arteries are ligated, and the bifurcation is resected.
Ligation and resection of the carotid artery for rupture have been associated with death rates of 33% to 50% and neurologic sequelae rates of 15% to 25% among survivors [32]. Elective resection for impending rupture is associated with decreased morbidity and mortality when compared with emergent resection. In highly selected cases, balloon occlusion of the carotid may be useful as a temporizing measure and for the prediction of neurologic outcome. However, despite the results of such a test, the artery most often will still require ligation.
Various methods of tissue coverage have been suggested for the prevention of carotid exposure after radical neck dissection; however, the effectiveness of such attempts is not clear [32]. Although dermal grafts are employed most commonly, the use of levator scapulae muscle flaps, omentum, and prevertebral fascia has been described. Dermal grafting is most likely to be of benefit in cases of radical neck dissection that have a possibility of a pharyngocutaneous or orocutaneous fistula and a history of prior irradiation, especially if the carotid adventitia was removed.
Sacrifice of the Internal Carotid Artery
If irradiation has not been successful in freeing a fixed node from the carotid artery, a decision must be made relative to carotid artery resection. Even though carotid artery resection with bypass may result in acceptable perioperative morbidity and mortality [33], we generally do not perform this maneuver, as survival is not improved with carotid artery resection [34,35].
Reference Guide
Therapeutic Agents
Mentioned in This Article
Bleomycin
Carboplatin
Carmustine
Cisplatin
Cetuximab (Erbitux)
Docetaxel (Taxotere)
Erlotinib (Tarceva)
Fluorouracil
Gefitinib (Iressa)
Hydroxyurea
Lomustine
Methotrexate
Mitomycin
Paclitaxel
Procarbazine
Brand names are listed in parentheses only if a drug is not available generically and is marketed as no more than two trademarked or registered products. More familiar alternative generic designations may also be included parenthetically.
Benefit of Saving the Spinal Accessory Nerve
A variety of neural injuries may be evident after neck dissection, but injuries to the spinal accessory nerve predominate. The spinal accessory nerve passes through the lymph node-bearing tissue of the neck, requiring that it either be sacrificed or dissected for preservation.
The majority of permanent spinal accessory nerve injuries are associated with the purposeful sacrifice of the nerve in conjunction with a radical neck dissection. This results in denervation of the trapezius muscle. The associated "shoulder syndrome" consists of shoulder pain, weakness, impaired mobility in abduction of the arm over 90°, shoulder droop, and scapular winging. The shoulder pain is due to the increased demands for support of the scapula on the levator scapulae and rhomboid muscles, and is worse if the dorsal scapular nerve is also injured. An adhesive capsulitis of the shoulder joint may develop as well, and predominate symptomatically [36].
Because loss of the spinal accessory nerve is generally the most debilitating part of a radical neck dissection, various methods of preserving trapezius function have been utilized. Given that the spinal accessory nerve receives contributions from the second, third, and fourth cervical nerves, preservation of these branches was proposed as a method of preserving some trapezius function. However, it now seems clear that these branches are mostly proprioceptive in nature, and any residual shoulder function was more likely due to preservation of the nerve to the levator scapulae muscle.
Resection of the spinal accessory nerve and cable grafting achieves some diminution of the shoulder syndrome. A "save XI" neck dissection still results in significant atrophy of the trapezius in up to 50% of patients, due to the extensive trauma to the nerve and devascularization associated with this procedure.
Modified radical neck dissection with preservation of the sternocleidomastoid muscle and spinal accessory nerve is associated with the least trauma to and devascularization of the spinal accessory nerve. This ultimately results in the best trapezius function, although there is still a temporary phase of dysfunction, which recovers significantly by 6 months after neck dissection, as documented by electrodiagnostic testing [37]. Since modified radical neck dissection is associated with acceptable nodal recurrence rates, this has become the nerve-sparing procedure of choice.
Benefit of Saving the Internal Jugular Vein
The internal jugular vein is typically conserved in modified radical and selective neck dissections. This allows the performance of simultaneous bilateral neck dissections with a diminished risk of increased intracranial pressure, the syndrome of inappropriate antidiuretic hormone secretion, facial edema, blindness, stroke, and airway obstruction.
The benefit of saving the internal jugular vein during a unilateral neck dissection is not as clear, but logically would seem to be of benefit, to a lesser degree. The internal jugular vein has been demonstrated to be reliably patent following conservation procedures. Factors that are associated with the rare instance of internal jugular vein thrombosis include prior irradiation, nodal recurrence, and compression by a musculocutaneous flap [38-40].
Conclusion
The surgical management of cervical metastases from head and neck cancer has undergone a significant change to more conservative techniques for appropriately selected individuals. Improved imaging modalities facilitate the staging of cervical metastases. The selection of the appropriate treatment for the neck is best done by a multidisciplinary group of surgeons, radiation oncologists, medical oncologists, path- ologists, and radiologists due to the complex relationship between the management of the primary site and the neck.
References:
1. Crile G: Excision of cancer of the head and neck with special reference to the plan of dissection based upon one hundred thirty-two operations. JAMA 47:1780-1786, 1906.
2. Martin HE, De Valle B, Ehrlich H, et al: Neck dissection. Cancer 4:441-449, 1951.
3. Lindberg RD: Distribution of cervical lymph node metastases from squamous cell carcinoma of upper respiratory and digestive tracts. Cancer 29:1446-1449, 1972.
4. Mendenhall WM, Million RR: Elective neck irradiation for squamous cell carcinoma of the head and neck: Analysis of time-dose factors and causes of failure. Int J Radiat Oncol Biol Phys 12:741-746, 1986.
5. Stevens MH, Harnsberger HR, Mancuso AA, et al: Computed tomography of cervical lymph nodes. Arch Otolaryngol Head Neck Surg 111:735-739, 1985.
6. van den Brekel MW, Castelijns JA, Croll GA, et al: Magnetic resonance imaging vs palpation of cervical lymph node metastasis. Arch Otolaryngol Head Neck Surg 117:663-673, 1991.
7. Hughes CJ, Gallo O, Spiro RH, et al: Management of occult neck metastases in oral cavity squamous ca. Am J Surg 166:380-383, 1993.
8. Jesse RH, Barkley HT, Lindberg RD, et al: Cancer of the oral cavity: Is elective neck dissection beneficial? Am J Surg 120:505-508, 1970.
9. Mendenhall WM, Million RR, Cassisi NJ: Elective neck irradiation in squamous cell ca of the head and neck. Head Neck 3:15-20, 1980.
10. Friedman M, Mafee MF, Pacella BL: Rationale for elective neck dissection in 1990. Laryngoscope 100:54-59, 1990.
11. Weiss MH, Harrison LB, Isaacs RS: Use of decision analysis in planning a management strategy for the stage N0 neck. Arch Otolaryngol Head Neck Surg 120:699-702, 1994.
12. Byers RM: Modified neck dissection: A study of 967 cases from 1970 to 1980. Am J Surg 150:414-421, 1985.
13. Medina JE: A rational classification of neck dissections. Otolaryngol Head Neck Surg 100:169-176, 1989.
14. Molinari R, Chiesa F, Cantu G, et al: Retrospective comparison of conservative and radical neck dissection in laryngeal cancer. Ann Otol 89:578-581, 1980.
15. Lingeman RE, Stephens R, Helmus C, et al: Neck dissection: Radical or conservative. Ann Otol 86:737-744, 1977.
16. Gavilan C, Gavilan J: Five-year results of functional neck dissection for cancer of the larynx. Arch Otolaryngol Head Neck Surg 115:1193-1196, 1989.
17. Bocca E, Pignataro O: A conservation technique in radical neck dissection. Ann Otol Rhinol Laryngol 76:975-987, 1967.
18. Wenig BL, Applebaum EL: The submandibular triangle in squamous cell ca of the larynx and hypopharynx. Laryngoscope 101:516, 1991.
19. Ballantyne AJ: Significance of retropharyngeal nodes in cancer of the head and neck. Am J Surg 108:500-504, 1964.
20. Hasegawa Y, Matsuura H: Retropharyngeal node dissection in cancer of the oropharynx and hypopharynx. Head Neck 16:173-180, 1994.
21. Parsons JT, Million RR, Cassisi NJ: The influence of excisional or incisional biopsy of metastatic neck nodes on the management of head and neck cancer. Int J Radiat Oncol Biol Phys 11:1447-1454, 1985.
22. Snow GB, Annyas AA, van Slooten EA, et al: Prognostic factors of neck node metastasis. Clin Otolaryngol 7:185-192, 1982.
23. Johnson JT, Barnes EL, Myers EN, et al: The extracapsular spread of tumor in cervical node metastasis. Arch Otolaryngol Head Neck Surg 107:725-729, 1981.
24. Hirabayashi H, Koshii K, Uno K, et al: Extracapsular spread of squamous cell carcinoma in neck lymph nodes: Prognostic factor of laryngeal cancer. Laryngoscope 101:502-506, 1991.
25. Ellis ER, Mendenhall WM, Rao PV, et al: Does node location affect the incidence of distant metastases in head and neck squamous cell ca? Int J Radiat Oncol Biol Phys 17:293-297, 1989.
26. Strong EW: Preoperative radiation and neck dissection. Surg Clin North Am 49:271-276, 1969.
27. Skolnik EM, Deutsch EC: Conservative neck dissection. Laryngoscope 95:561-565, 1985.
28. Jesse RM, Ballantyne AJ, Larson D: Radical or modified neck dissection: A therapeutic dilemma. Am J Surg 136:516-519, 1978.
29. Barkley HT, Fletcher GH, Jesse RH, et al: Management of cervical lymph node metastases in squamous cell carcinoma of the tonsillar fossa, base of tongue, supraglottic larynx, and hypopharynx. Am J Surg 124:462-467, 1972.
30. Mendenhall WM, Million RR, Cassisi NJ: Squamous cell carcinoma of the head and neck treated with radiation therapy: The role of neck dissection for clinically positive neck nodes. Int J Radiat Oncol Biol Phys 12:733-740, 1986.
31. Grandi C, Mingardo M, Guzzo M, et al: Salvage surgery of cervical recurrences after neck dissection or radiotherapy. Head Neck 15:292-295, 1993.
32. Hillerman BL, Kennedy TL: Carotid rupture and tissue coverage. Laryngoscope 92:985-988, 1982.
33. Biller HF, Urken M, Lawson W, et al: Carotid artery resection and bypass for neck carcinoma. Laryngoscope 98:181-183, 1988.
34. Brennan JA, Jafek BW: Elective carotid artery resection for advanced squamous cell ca of the neck. Laryngoscope 104:259-263, 1994.
35. Meleca RJ, Marks SC: Carotid artery resection for cancer of the head and neck. Arch Otolaryngol Head Neck Surg 120:974-978, 1994.
36. Patten C, Hillel AD: The 11th nerve syndrome: Accessory nerve palsy or adhesive capsulitis? Arch Otolaryngol Head Neck Surg 119:215-220, 1993.
37. Remmler D, Byers R, Scheetz J, et al: A prospective study of shoulder disability resulting from radical and modified neck dissections. Head Neck 8:280-286, 1986.
38. Cotter CS, Stringer SP, Landau S, et al: Patency of the internal jugular vein following modified radical neck dissection. Laryngoscope 104:841-845, 1994.
39. Lake GM, Dinardo LJ, Demeo JH: Performance of the internal jugular vein after functional neck dissection. Otolaryngol Head Neck Surg 111:201-204, 1994.
40. Docherty JG, Carter R, Sheldon CD, et al: Relative effect of surgery and radiotherapy on the internal jugular vein following functional neck dissection. Head Neck 15:553-556, 1993.