Pharmacologic manipulation of the immune system is emerging as a viable and robust treatment for some cancer patients. Exercise-induced modulation of the immune system may be another adjunctive strategy for inhibiting tumor initiation and progression. In healthy individuals, exercise has been shown to modulate a number of cell subsets involved in innate and adaptive immunity. Here, we provide an overview of the current state of knowledge pertaining to exercise modulation of the inflammation-immune axis in cancer. The current evidence suggests that exercise may be a promising adjunctive strategy that can favorably alter numerous components of the immune system, which, in turn, may modulate tumorigenesis. However, many important knowledge gaps are evident. To this end, we propose a framework to guide future research efforts investigating the immune effects of exercise in cancer.
Introduction
Over the past 2 decades, a steadily growing and increasingly diverse evidence base has emerged investigating the tolerability and initial efficacy of general physical activity, as well as of structured aerobic exercise training interventions (hereafter referred to as “exercise”), in preventing and/or mitigating the adverse physiologic and psychosocial impact of cancer and associated cytotoxic and supportive care therapies.[1] On the basis of available evidence, several international agencies have published cancer-specific exercise guidelines intended specifically for use by cancer patients both during and following the completion of primary therapy.[2-4] Symptom control is, and will likely continue to be, the major focus of research and clinical efforts in “exercise oncology” over the next 2 decades. In conjunction with such efforts, however, a new line of investigation has started to emerge exploring whether the benefit of exercise extends beyond symptom control-that is, exploring the efficacy of exercise as a primary and/or combination treatment for cancer (ie, the use of exercise with therapeutic intent).[5]
Promising data from a growing but limited number of observational studies indicate that, in general, higher levels of self-reported postdiagnosis exercise exposure are associated with reductions in the risk of recurrence and cancer-specific mortality (as well as overall mortality) in patients with nonmetastatic breast, prostate, and colorectal cancer.[6,7] A limited number of preclinical studies also indicate that exercise exhibits antitumor activity; these studies have been conducted mostly in immune-competent animals bearing syngeneic allografts, as well as in genetically engineered mouse models of breast and prostate cancer.[8-13] Collectively, these data have led to the provocative hypothesis that postdiagnosis exposure to exercise may inhibit disease progression-prompting calls from many stakeholders regarding the need for large-scale definitive clinical trials to test this question. As with the development of all anticancer agents, successful development of exercise as a candidate anticancer strategy will be contingent on elucidation of the underlying mechanisms of action.[5]
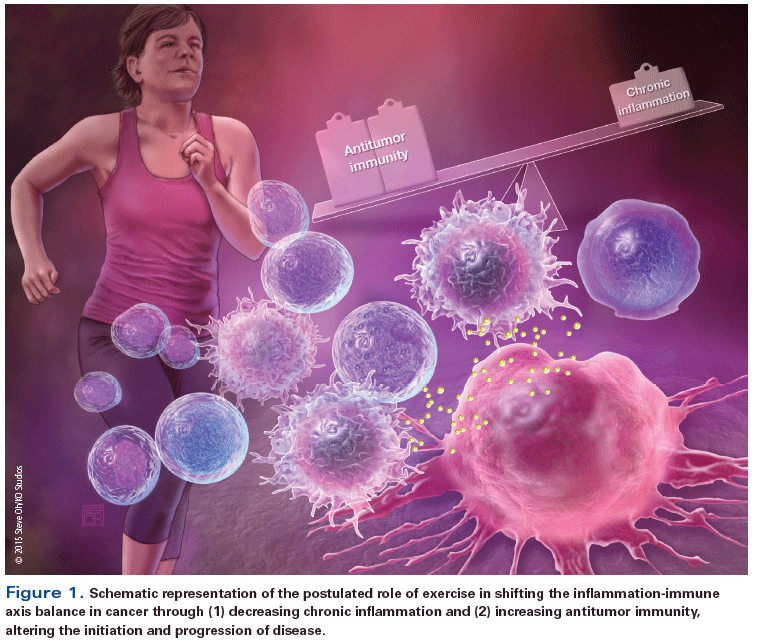
It is established that tumor progression is regulated by complex, multifaceted interactions between the systemic milieu (host), the tumor microenvironment, and cancer cells.[14] The tumor microenvironment, whether in primary or distant ectopic sites, is directly influenced by and responsive to circulating growth factors, cytokines and angiogenic factors, hormones, and numerous other cell types-including immune cells-that comprise the systemic host milieu.[15,16] Higher circulating (systemic) levels of certain inflammatory cytokine and angiogenic factors (eg, hepatocyte growth factor, tumor necrosis factor [TNF], interleukin [IL]-6),[17-19] as well as of metabolic growth hormones (eg, insulin, glucose, leptin),[20] are associated with higher risks of recurrence and cancer-specific mortality in a number of solid malignancies.[21] To date, few studies have investigated the ability of exercise treatment to modulate host factors.[7] A host-related pathway that also may play a critical role in underpinning the antitumor effects of exercise is the inflammation-immune axis (Figure 1).[22]
The role of the immune system in eradicating malignant cells has been recognized for almost a century; over the past decade, research efforts in this area have been significantly bolstered by seminal discoveries of how T cells recognize, regulate, and eradicate cancer cells.[23] These efforts have spearheaded a paradigm shift in the treatment of certain solid tumors, with numerous immune activators and immune checkpoint inhibitors either approved or currently under active investigation.[24,25] Intriguingly, emerging evidence indicates that exercise significantly alters the number and function of circulating cells of the innate immune system (eg, neutrophils, monocytes, and natural killer [NK] cells) and, to a lesser degree, of the adaptive immune system (eg, T and B cells).[26] However, whether modulation of the immune system contributes to the potential antitumor properties of exercise has not been thoroughly examined.
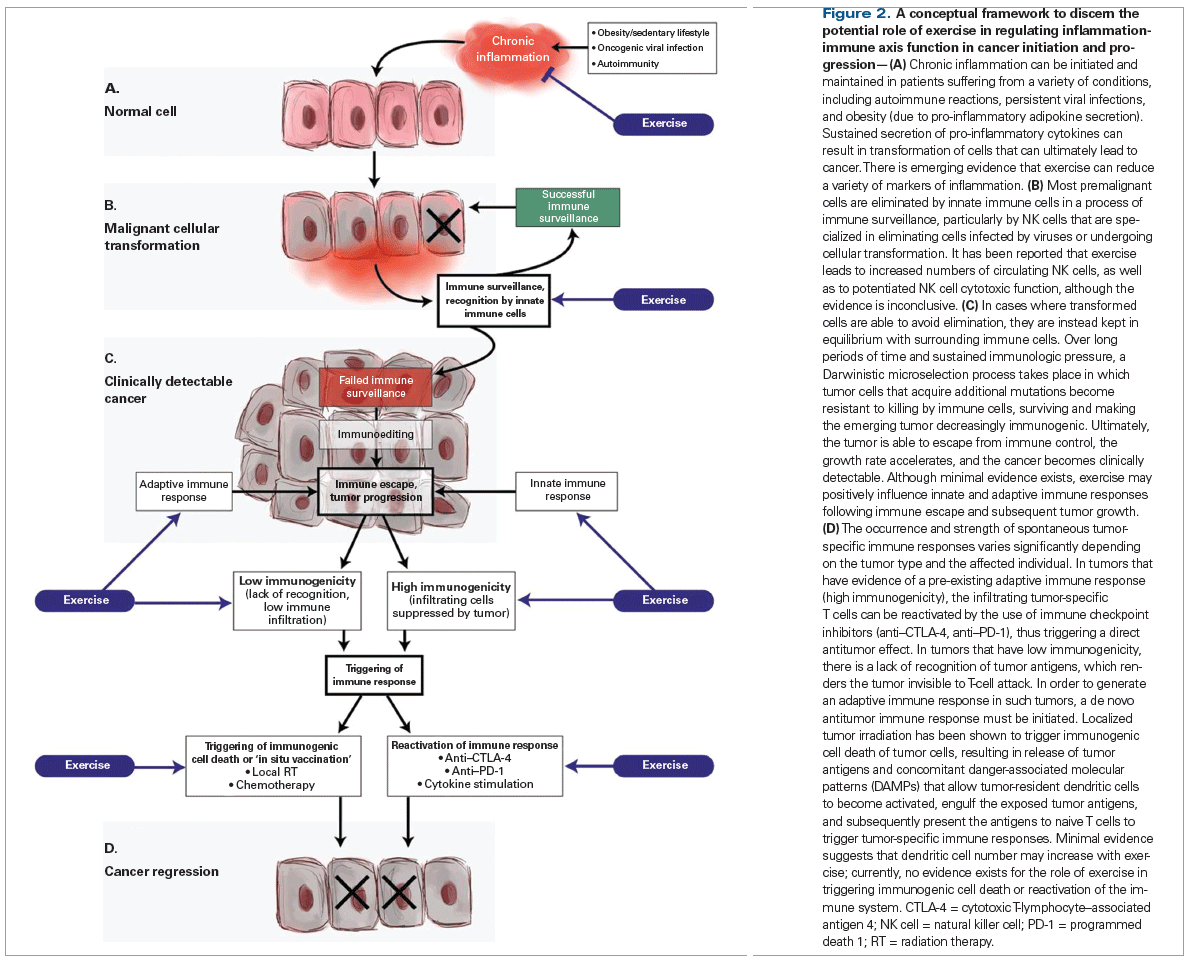
Accordingly, here we provide an overview of the current state of knowledge regarding exercise modulation of the inflammation-immune axis in cancer. Specifically, we present a framework that focuses on the effects of long-term exercise (as opposed to acute, single bouts of exercise) on specific steps at which normal cells interact with the immune system, and at which-when the immune response fails to eliminate malignantly transformed cells-immune escape and cancer growth may ensue (Figure 2). The subsequent sections of this paper will thus provide an overview of: (A) chronic inflammation and malignant cellular transformation (cancer initiation); (B) immune surveillance of malignantly transformed cells; (C) immune escape and subsequent tumor progression, with emphasis on innate immune cell tumor infiltration and adaptive immune responses; and (D) immune activation in response to a clinically detectable cancer. For each step, we describe the known interaction between the host and the inflammation-immune axis, as well as look at the known or potential effects of exercise both in health and in cancer. Finally, we highlight current research gaps, with the hope that this will inform areas of future research.
A. Chronic Inflammation and Malignant Cellular Transformation
Cells of the innate immune system are equipped with germline-encoded pattern-recognition receptors (PRRs), including the Toll-like receptors (TLRs), which directly bind to conserved microbial structures that are characteristic of microbes such as viruses and bacteria (pathogen-associated molecular patterns [PAMPs]).[27] Signaling downstream of TLRs leads to activation of the central inflammatory transcription factor nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), which triggers a local acute inflammatory response whose purpose is to contain and/or eliminate the pathogen. Vascular endothelial cells upregulate adhesion molecules and chemokines to allow recruitment and extravasation of immune cells. Tissue-resident neutrophils and macrophages produce pro-inflammatory cytokines, such as interferon-γ (IFN-γ), IL-6, and TNF-α, which, via both autocrine and paracrine signaling, trigger the recruitment and activation of additional innate immune cells, including NK cells, in order to eliminate infected cells. Simultaneously, tissue-resident dendritic cells (DCs) phagocytize microbial structures, resulting in the maturation of DCs and the subsequent presentation of the microbial antigens to naive CD8+ and CD4+ T cells and B cells in secondary lymphoid organs, thus initiating the specific adaptive immune responses required for infection eradication.[28]
An inflammatory response that does not eventually resolve can transition into a state of chronic “smoldering” inflammation that is permissive for tumorigenesis; this process has been described in detail in numerous prior excellent reviews.[29,30] In brief, under certain pathologic conditions involving dysregulated immune system function, including chronic infections, some autoimmune diseases, and obesity, smoldering inflammation causes prolonged cellular stress, which can result in malignant cellular transformation and cancer (Figure 2A).[31-35] It is important to note that in this review, we are focusing on the prophylactic effect of reducing chronic inflammation (ie, the ability of such a reduction to prevent cancer initiation); however, this focus is not intended to exclude or negate a treatment effect of reducing chronic inflammation (ie, the inhibition of already diagnosed cancer), particularly in inflammation-driven cancer. Multiple strategies have been deployed in an attempt to reverse low-grade inflammation and thus to prevent and/or slow cancer initiation and progression; such strategies have met with mixed success.[36]
Exercise and inflammation in health
Exercise has been shown to modulate local and systemic inflammatory processes. As described in excellent reviews by Gleeson et al[37] and Walsh et al,[26] exercise is postulated to modulate inflammation in humans via a number of processes. Exercise leads to a reduction in visceral fat mass and a subsequent reduction in pro-inflammatory adipokine secretion, as well as a reduction in macrophage infiltration into adipose tissue.[38,39] Exercise can also induce macrophage polarization in adipose tissue towards an anti-inflammatory M2 phenotype, thereby reducing systemic inflammation independent of changes in fat mass-although to our knowledge, this has only been shown in preclinical models.[39] Each individual bout of exercise also results in the production and release of cytokines from contracting skeletal muscle (such cytokines are thus termed “myokines”).[40,41] For example, IL-6 released from skeletal muscle appears to be responsible for subsequent elevations in circulating anti-inflammatory cytokines IL-1Ra and IL-10, as well as for cortisol release from the adrenal gland,[42] which is known to have potent anti-inflammatory properties.[43] Combined with the exercise-induced activation of the hypothalamic-pituitary-adrenal axis and the release of cortisol and catecholamines, which can downregulate the lipopolysaccharide (LPS)-induced production of cytokines (eg, TNF-α and IL-1β) by antigen-presenting cells such as macrophages and DCs,[44] these alterations cumulatively result in an anti-inflammatory systemic host environment that can be maintained with long-term, sustained exercise.[37] Exercise also reduces circulating pro-inflammatory (CD14+CD16+) monocytes and decreases LPS-stimulated monocyte-specific TNF-α production.[45]
Exercise can also alter autonomic nervous system function by increasing parasympathetic (vagal) nervous system activity,[46] the effects of which may regulate the inflammatory reflex.[47] Efferent vagus nerve activity can induce catecholamine-mediated activation of T-cell–derived acetylcholine release in the spleen and release of acetylcholine from vagal nerve endings in other organs. The result is inhibition of NF-κB nuclear translocation and reduced pro-inflammatory cytokine secretion by macrophages and other immune cells.[47] Proof of this concept with exercise, however, has not been discerned.
Exercise also favorably alters plasma lipoprotein levels,[48] in particular by increasing high-density lipoprotein (HDL) concentrations.[49,50] These favorable alterations in the lipid profile are associated with reduced inflammation.[51,52]
Exercise and inflammation in cancer
Few studies have evaluated the effects of exercise on pro-inflammatory effectors in cancer, although preliminary data appear promising. For example, Jones et al[53] randomized 20 patients with locally advanced breast cancer to either a 12-week nonlinear exercise prescription in combination with neoadjuvant doxorubicin-cyclophosphamide (AC) or AC alone. Results demonstrated significant reductions in several circulating cytokines (eg, IL-1β, IL-2). However, there were no changes in the pro-inflammatory cytokines IL-6 and TNF-α, a finding that was supported by Ergun et al[54]; the latter researchers also reported no changes in a panel of inflammatory cytokines (eg, IL-6, IL-8, and TNF-α) after 12 weeks of exercise following treatment in breast cancer patients. Glass et al[55] found that 12 weeks of supervised exercise was associated with a significant reduction in circulating levels of IL-4, macrophage inflammatory protein beta (MIP-1β), and TNF-α in a heterogeneous study cohort of 44 patients with solid tumors who were receiving cytotoxic therapy. Despite these promising initial data, there is currently insufficient evidence to draw definitive conclusions regarding the ability of exercise to modulate circulating levels of inflammatory effectors in any cancer setting. The current data are also confounded by the considerable heterogeneity in study methodology, which stems from small sample sizes, analysis of different pro/anti-inflammatory biomarker panels, heterogeneous patient populations, and use of nonstandardized doses of exercise. In addition, whether exercise-induced alterations in circulating concentrations of inflammatory effectors are associated with and/or directly underpin (1) tissue-specific changes in inflammation (in the tumor/microenvironment), or (2) the potential antitumor effects of exercise is not known.
B. Immune Surveillance
The role of the immune system in protecting against cancer development and in shaping the types of cancers that arise in immune-competent hosts has been unequivocally demonstrated.[56] Within the immune system, NK cells play a critical role in the immune surveillance or “elimination” phase.[57-59] NK cells are part of the innate immune system, specialized in the recognition of cells with increased expression of stress-induced ligands (MICA/B, UL16 binding proteins [ULBPs], TNF-related apoptosis-inducing ligand [TRAIL] receptors) and low expression of major histocompatibility complex (MHC) class I, a common characteristic of transformed cells.[60-64] IFN-γ produced by NK cells promotes cross-presentation of tumor cell–derived antigens to T cells by antigen-presenting myeloid cells, leading to the development of adaptive immunity. This adaptive immunity not only eliminates many incipient cancers, but also provides selective pressure that results in the progression only of cancers that have decreased immunogenicity and that have acquired the ability to suppress the immune response (Figure 2B).[56]
Exercise and immune surveillance in health
There is some evidence to suggest that exercise increases NK cell number and function, although not all studies support this conclusion.[26] For example, in adults with suspected impaired immune surveillance (eg, older individuals), 10 weeks of exercise increased circulating NK cell–mediated cytotoxicity (NKCC) against K562 tumor cells in vitro.[65] However, in a similar cohort, 24 weeks of exercise showed no significant changes in NKCC[66]; in addition, a 12-month trial found no change in NKCC in postmenopausal women.[67]
Exercise and immune surveillance in cancer
Several studies have evaluated the effect of exercise on NK cell function in cancer.[22,68] For example, after 15 weeks of stationary cycle ergometry in 52 postmenopausal breast cancer patients following the completion of primary adjuvant therapy, Fairey et al[69] found increased cytotoxic activity of NK cells in peripheral blood mononuclear cells that target K562 cells, compared with a nonexercise control group. However, Saxton et al[70] found no effect on NKCC in 85 overweight breast cancer patients who were 3 to 18 months posttreatment and who were randomized to either a 6-month exercise-plus-hypocaloric-diet intervention or usual care. These contrasting findings likely reflect the differences in exercise dose, as well as the heterogeneity of the patient populations. Another limitation is that the assays used in these studies only measure NKCC in vitro. Performing studies in mouse models of cancer that use NK cell–depleted mice (with NK cell depletion mediated by anti–asialo-GM1– and anti-NK1.1–depleting antibodies) would provide direct evidence for the role of exercise on NK cell–mediated antitumor effects. Furthermore, the genotyping of killer-cell immunoglobulin-like receptors (KIRs) from peripheral blood NK cells in clinical cohorts could give an indication of whether exercise has an effect on NK cell development and licensing in healthy individuals and cancer patients, further clarifying the role of exercise in modulating NK cell “immunosurveillance” activity. It is also important to note that, although it is beyond the scope of this review, a considerable literature base also exists on the effect of a single bout of exercise on NK cells. In brief, NK cell number significantly increases following a single bout of exercise,[71] which may have implications in the cancer setting, particularly in allogeneic adoptive NK-cell transfer.[26]
C. Immune Escape and Tumor Progression
If complete elimination of the malignant cells fails, the continued immunologic pressure on the emerging tumor will instead lead to the selection of increasingly less immunogenic tumor cells (Figure 2C). This phase of immunoediting is driven largely by T cells and occurs over several years.[72] Malignant cells with high chromosomal instability escape antigen-specific T-cell recognition by antigenic loss or upregulation of immunosuppressive mediators. Accumulation of de novo mutations eventually results in an invasive phenotype that favors escape from immune surveillance, leading ultimately to an overt (detectable) tumor.[73] Traits acquired by tumor cells that enable them to avoid recognition by adaptive immune cells include downregulated surface expression of MHC class I, as well as the shedding of antigens and natural killer group 2D (NKG2D) ligands.[74-76] Moreover, tumors gradually become more resistant to elimination by immune cells due to upregulation of immune-inhibitory molecules, such as ligands for the T-cell–inhibitory receptor programmed death 1 (PD-1). The shedding of death receptors and the overexpression of anti-apoptotic proteins increase resistance to apoptosis.[77-80] Importantly, tumor cells also manipulate the cellular composition of the tumor microenvironment by secreting chemokines and cytokines that can recruit and polarize myeloid cells and some lymphoid cells to an immunosuppressive, protumorigenic phenotype. Unfortunately, the role of exercise in modulating these processes remains unexplored.
Innate immune response
Macrophages typically infiltrate human tumors, where they play an important role in shaping the tumor microenvironment (see Figure 2C). In the early stages of tumor development, tumor-associated macrophages (TAMs) are polarized by IFN-γ to an inflammatory M1 phenotype characterized by tumoricidal activity and IL-12 secretion, which supports NK-cell activation and the development of T helper 1 (Th1) immunity.[81,82] In more advanced stages of tumor development, TAMs are recruited to the tumor microenvironment primarily via monocyte chemoattractant protein (CCL2)-induced chemotaxis; they are converted to an M2 phenotype via tumor-derived immunosuppressive cytokines, including transforming growth factor (TGF)-β and IL-10, and promote tissue remodeling and angiogenesis.[83] M2-polarized TAMs are enriched in hypoxic and ischemic areas of tumors, where expression of hypoxia-inducible factor 1α (HIF-1α) and vascular endothelial growth factor (VEGF) are central regulators of macrophage recruitment. Macrophage tumor infiltration is therefore associated with adverse outcomes and shorter survival across multiple cancer subtypes.[84-86] In preclinical models, pharmacologic modulation of CCL2-mediated migration and VEGF receptor 2 (VEGFR-2)–mediated tumor infiltration is associated with reduced tumor burden, demonstrating the therapeutic potential of inhibiting TAM recruitment.[87,88] In addition, downregulation of placental growth factor (PlGF) using histidine-rich glycoprotein skewed the polarization of macrophages to an M1 phenotype, resulting in improved antitumor immune responses.[89] Tumor-associated neutrophils (TANs) possess a wide range of traits that can allow them to effectively kill tumor cells (via secretion of oxidants and proteases), as well as recruit and stimulate effector immune cells via secretion of cytokines (IL-8, C-X-C motif ligand 10 [CXCL10], IL-12, TNF-α, IL-1β, and IFN-γ).[90] However, tumor-derived TGF-β has also been shown to cause polarization of TANs from an antitumorigenic N1 phenotype to an N2 phenotype that promotes angiogenesis and metastasis and induces oxidative stress in effector T cells through production of reactive oxygen species, thus inhibiting the antitumor immune response.[91]
Exercise and innate immune response in health. In noncancer populations, research has focused on the capacity of exercise modulation of the innate immune system to (1) mitigate low-grade chronic inflammation (discussed earlier, in section A) and (2) enhance response to infections and other environmental injuries[92]; these capacities may provide clues to the role of exercise in response to tumor escape and progression. Interestingly, Kizaki et al[93] showed that 3 weeks of treadmill exercise in BALB/c mice increased LPS-induced release of nitric oxide (NO) and production of IFN-γ and TNF-α, and lowered levels of IL-10 production in peritoneal macrophages, suggesting a potential shift towards a more pronounced M1 phenotype and enhanced innate immune response. These results corroborated findings from Sigiura et al[94] that showed that 8 weeks of treadmill exercise also significantly increased LPS-induced production of NO and IL-1β by peritoneal macrophages. The results of the work by Kizaki et al and Sigiura et al, however, stand in contrast to other researchers’ findings of a decreased LPS response of circulating monocytes to exercise (decreased LPS-stimulated TNF-α production) in humans.[45] Thus, we speculate that exercise may reduce the inflammatory phenotype (and number) of circulating monocytes, but upon differentiation of monocytes into macrophages within specific tissues, exercise may favor an inflammatory (M1) phenotype, which would be advantageous for antitumor immunity. With regard to neutrophil function following exercise, little is currently known, although exercise does not appear to significantly alter neutrophil counts.[26,65] Acutely, exercise does rapidly induce a profound neutrophilia, followed by a delayed increase in the blood neutrophil count, 1 to 2 hours following exercise cessation; this is modulated by both the length and intensity of exercise.[95] Repeated bouts of high-intensity exercise may reduce neutrophil respiratory burst,[96,97] although this has only been shown in athlete populations.
Exercise and innate immune response in cancer. To our knowledge, no clinical studies have examined the effect of exercise on TAMs and TANs. Preclinical work found that exercise might modulate the polarization and resultant function of macrophages external to the tumor. Specifically, in a chemically induced model of mammary carcinogenesis, exercise promoted M1 polarization of isolated peritoneal macrophages stimulated with LPS (via increased levels of the cytokines IFN-γ, TNF-α, and IL-12) compared with sedentary mice, which exhibited a more pronounced M2 phenotype (increased levels of IL-10 and TGF-β).[98] Furthermore, as M2 TAMs are enriched in hypoxic areas of the tumor, “normalization” of these hypoxic regions could in turn alter the recruitment, retention, and polarization of TAMs. Interestingly, our group found that voluntary wheel running in immunocompetent BALB/c and C57BL/6 mice injected orthotopically with estrogen-negative 4T1 and estrogen-positive E0771 tumor cells reduced tumor growth compared with sedentary controls; the reduced tumor growth occurred in conjunction with increased microvessel density, vessel maturity, and perfusion, leading to reduced intratumoral hypoxia.[11] Thus, exercise-induced tumor normalization via alteration in the recruitment, polarization, and function of TAMs appears biologically plausible. Functionally, 4 months of treadmill running in BALB/c mice elicited greater cytolysis of p815 tumor cells by resident peritoneal macrophages in vitro than in sedentary control mice,[99] suggesting again a predominant M1 phenotype. Exercise may also limit TAM/TAN infiltration into the tumor. Exercise (forced swimming) in male Swiss mice injected with Ehrlich tumor cells (6 weeks of exercise, with tumor cell injection at 4 weeks) resulted in lower tumor volume and concomitant decrease in macrophage and neutrophil infiltration.[100] This is supported by a study from Zielinski et al[101] showing that 2 weeks of treadmill running in BALB/c mice injected with EL4 lymphoma cells decreased macrophage and neutrophil infiltration compared with sedentary control mice. Further, McClellan et al[8] showed that in the ApcMin/+ mouse model of intestinal tumorigenesis, 12 weeks of treadmill running reduced the number of large polyps alongside decreased overall macrophage presence and reduction of M1 (eg, IL-12) and also M2 (eg, CD206, CCL22, and Arg-1) gene expression in intestinal tissue. Overall, exercise may enhance the antitumor immune response following immune escape and resultant tumor progression, through alterations in macrophage and neutrophil infiltration and macrophage polarization-although validation of these findings in vivo is required. Specifically, studies to discern the potential mechanisms underlying the role of exercise in reducing monocyte recruitment and macrophage/neutrophil infiltration into the tumor, as well as in polarization of TAMs and TANs once within the tumor microenvironment, are required.
Adaptive immune response
Adaptive immune cells play a critical role in the control of tumor development (see Figure 2C). In recent years, the mapping of spontaneous adaptive antitumor immune responses in cancer patients has provided highly accurate prognostic markers, as well as predictive markers of response to chemotherapy and immunotherapy.[102-105] For example, using analysis of the spatio-temporal distribution of different immune cell types in colorectal cancer specimens, Galon et al found that high intratumoral density of Th1-polarized T cells (particularly CD8+ central memory T cells and CD4+ follicular T helper cells) and B cells, as well as expression of T-cell homing chemokines (C-X3-C motif ligand 1 [CX3CL1] and CXCL10) and adhesion molecules (intercellular adhesion molecules and vascular cell adhesion molecules), was predictive of prognosis.[106-110] Regulatory T cells (Tregs) are also important for maintaining peripheral tolerance and are capable of suppressing the adaptive immune response to tumors via secretion of immunosuppressive cytokines, including IL-10 and TGF-β.[109] Recruitment and conversion of conventional Foxp3− CD4+ T cells into Foxp3+ Tregs via secretion of TGF-β is a common immune escape strategy employed by tumors, and high Treg infiltration in solid tumors has been correlated to poor prognosis.[110,111]
Exercise and adaptive immune response in health. Exercise in younger sedentary populations has failed to demonstrate any marked changes in T-cell function[26]; however, exercise in older individuals may have an effect.[112] For example, 6 months of exercise increased the T-cell proliferative response to mitogen stimulation,[65] and 10 months of a similar exercise protocol in sedentary older individuals significantly improved influenza vaccine response (via increased seroprotection and reduced overall illness severity), compared with controls.[113] Preclinical work in C57BL/6 mice found that 6 weeks of high-intensity treadmill running, but not moderate-intensity running, increased the proportion of CD4+CD25+ Tregs in the spleen and the expression of Foxp3,[114] suggesting that higher-intensity training may elicit a Treg-mediated dampened adaptive immune response. Cumulatively, exercise may maintain immunocompetent T-cell populations, and the intensity of training is likely important in mediating the adaptive immune cell response.
Exercise and adaptive immune response in cancer. Exercise-induced changes in intratumoral T-cell composition and function in the clinical cancer setting have received scant attention.[22] In preclinical studies, an 8-week moderate swimming intervention in mice injected with DMBA increased quantities of splenic lymphocytes producing IFN-γ, IL-2, IL-12, and TNF-α; decreased quantities of lymphocytes (and macrophages) expressing IL-4, IL-10, and TGF-β; and lowered proportions of splenic Tregs compared with sedentary mice.[115] In patients, Glass et al[55] showed nonsignificant increases in the percent change in circulating T-lymphocyte population subsets (CD8+, CD4+, CD8+/CD45RA, and naive cytotoxic T cells) following a 12-week supervised exercise program in a cohort of heterogeneous solid tumor patients. However, Saxton et al[70] found that patients undergoing a 6-month exercise intervention exhibited a reduced total lymphocyte count compared with patients who did not undergo the intervention, although the lymphocyte proliferative response to phytohemagglutinin (PHA) after exercise was unchanged. In terms of intratumoral T-cell alterations, preclinical work from Goh et al[116] showed that voluntary wheel running in a transgenic polyoma middle T oncoprotein (PyMT) mouse model of invasive breast cancer resulted in decreased intratumoral gene expression of CCL22, a cytokine associated with Treg recruitment. McClellan et al[8] showed greater CD8 expression and reduced Foxp3 expression in lymphocytes isolated from polyp-laden intestinal tissue in ApcMin/+ mice exposed to 12 weeks of treadmill running. Further, Zielinski et al[101] showed greater lymphocyte density in tumors following 2 weeks of treadmill running in BALB/c mice injected with EL4 lymphoma cells, compared with sedentary mice. These data suggest that exercise may positively alter both the circulating and the intratumoral adaptive immune response in cancer.
D. Triggering an Immunogenic Response to the Tumor
As previously described, the degree of infiltration of effector T cells in tumors is a strong prognostic marker in a variety of tumor subtypes.[106,117-119] In addition, tumors with high CD8+ T-cell infiltration are more likely to respond to immune checkpoint inhibitors, which unleash T-cell function.[120] However, in most cancer patients, there is a lack of spontaneous antitumor immunity, as well as absent or minimal intratumoral T-cell infiltration, making the tumor somewhat “invisible” to the immune system. In such patients, any therapeutic approach will need to stimulate a de novo immune response. DCs are essential for the initiation of immune responses, and although they are found in tumors, they are often in a tolerogenic state in which they are unable to provide costimulatory signals to T cells during antigen presentation, rendering the T cells anergic.[121] However, DCs can be activated locally in the tumor following stimulation by danger-associated molecular patterns (DAMPs), including calreticulin, high-mobility group protein B1 (HMGB1), and adenosine triphosphate (ATP). Tumor cells undergoing immunogenic cell death, induced by certain anticancer therapies, translocate calreticulin to the cell surface, which provides an “eat me” signal to surrounding DCs and stimulates engulfment of the dying cells.[122] Moreover, extracellular HMGB1 and ATP released by dying cancer cells activate DCs in the tumor microenvironment via ligation with TLR4 and purinergic P2X receptors, respectively.[123] Activated DCs upregulate MHC class II and costimulation molecules, as well as induce chemokine receptors C-C chemokine receptor type 7 (CCR7) and C-X-C motif receptor 4 (CXCR4) to migrate to tumor-draining lymph nodes, where the processed tumor antigens are presented to T cells.[122,124-126] In addition, the sensing of tumor cell–derived DNA by tumor-resident DCs, which is mediated by the cGAS/STING pathway, activates the production of type I IFNs, which act in both an autocrine and paracrine fashion to stimulate the antigen-presenting function of DCs.[127,128]
TO PUT THAT INTO CONTEXT
[[{"type":"media","view_mode":"media_crop","fid":"44116","attributes":{"alt":"","class":"media-image","id":"media_crop_8121462691357","media_crop_h":"0","media_crop_image_style":"-1","media_crop_instance":"4910","media_crop_rotate":"0","media_crop_scale_h":"0","media_crop_scale_w":"0","media_crop_w":"0","media_crop_x":"0","media_crop_y":"0","title":" ","typeof":"foaf:Image"}}]]
Connie J. Rogers, PhD, MPH
Department of Nutritional Sciences
Pennsylvania State University
University Park, PennsylvaniaWhat Two Recent Developments Undergird Research Into Exercise and Immune Function in Cancer?This review highlights recent developments in two areas of cancer research that until not long ago were seemingly unrelated. First, there have been promising advances in cancer immunotherapy, including recent clinical successes that may signal the beginning of a transition for cancer immunotherapy from experimental to established therapy. Thus, there is heightened interest in novel therapies and modalities that target the immune system. Second, emerging population data indicate an important role for exercise in the prevention of cancer recurrence and mortality, suggesting that exercise may be a vital intervention in cancer survivors. The biological mechanisms that might explain a relationship between these two developments are not well studied, but it has been hypothesized that an exercise-induced enhancement of antitumor immunity and/or reduction in immunosuppression may be the mechanism that underlies the ability of exercise to reduce cancer recurrence and increase survival.What Are the Important Questions That Now Need to Be Answered?The field of exercise oncology is in its infancy and many unanswered questions remain. Critical information about the dose, duration, frequency, and type of exercise required to achieve a cancer prevention effect is essential to advance the field. Furthermore, the efficacy and safety of exercise across the cancer continuum needs to be studied to determine whether exercise can have a beneficial effect on the inflammation-immune axis during the treatment phase, as well as during the survivorship window. Interdisciplinary collaborations between investigators in the fields of exercise physiology, immunology, cancer biology, and oncology are crucial to adequately design and implement preclinical experiments and clinical trials addressing these key unsolved problems in exercise oncology.
Exercise-induced triggering of an immune response in health
To our knowledge, no studies to date have analyzed the effects of exercise on DC function in humans. Preclinical work in Sprague-Dawley rats found that a 5-week course of exercise significantly increased the mixed lymphocyte reaction of bone marrow–derived DCs (used to evaluate the T-cell stimulatory capacity of DCs) compared with controls, as well as increasing cytokine secretion capacity (IL-12) and expression of MHC class II molecules.[129] Together, these findings suggest greater DC antigen presentation ability, which may translate to greater antitumor immunity.
Exercise-induced triggering of an immune response in cancer
Further preclinical work in Sprague-Dawley rats[130] found a significant increase in DC number in a group subjected to 5 weeks of exercise compared with a nonexercise control group, yet there were no differences in expression of maturation markers CD80 and CD86. Moreover, peripheral blood mononuclear cells and splenocytes isolated from the rats in the exercise group showed greater ability to inhibit human leukemia U937 and murine lymphoma YAC-1 cell lines in vitro. These results need to be confirmed in human studies.
Reactivation of the immune system
The activation of T cells requires their interaction with antigen-presenting DCs in lymph nodes (priming phase) and is strictly regulated by concomitant engagement of costimulatory and coinhibitory receptors, which are known as immune checkpoints, and which play a critical role in the maintenance of self-tolerance under normal conditions. However, in cancer, the costimulatory function of DCs is often reduced, and monoclonal antibodies blocking the coinhibitory molecule cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4) can recover activation of tumor-specific T cells.[24] Another negative regulator of T-cell activation, PD-1, which interacts with ligands expressed in the tumor microenvironment, is also a major obstacle to the function of antitumor T cells.[131] Antibodies targeting CTLA-4 and PD-1 have shown remarkable clinical activity and have been approved by the US Food and Drug Administration for the treatment of melanoma and lung cancer.[132-136] However, in tumors that lack a pre-existing immune response and that have low lymphocytic infiltration, treatment with immune checkpoint inhibitors remains largely ineffective and is likely to require combination with other treatments that enhance tumor immunogenicity.[137,138] In this context, it is intriguing to consider the possibility that exercise, if capable of enhancing antitumor immune responses, could be an effective combination treatment with immune checkpoint inhibitors.
Triggering of immunogenic cell death or 'in situ vaccination'
Immunogenic cell death of tumor cells can be induced by treatment with chemotherapeutic agents or radiation therapy.[139,140] Preclinical work by our group has shown that radiation therapy, in combination with immune checkpoint blockade, can stimulate tumor-specific adaptive immune responses.[141,142] Clinical evidence for a similar synergy in patients is beginning to emerge. For instance, a patient with chemotherapy-refractory metastatic non–small-cell lung cancer underwent complete tumor regression after localized radiation therapy treatment of a single tumor in combination with anti–CTLA-4 antibody therapy.[143] The role of exercise in eliciting immunogenic cell death has not been discerned; however, we speculate that exercise-induced alterations in tumor physiology (improved tumor vascularization, decreased hypoxia, and increased apoptosis)[11] may increase T-cell infiltration, DC activation, and antigen presentation, leading to tumor inhibition.
Future Directions
On the basis of current knowledge, a full understanding of the complex and multifaceted interaction between the host, exercise regulation of the inflammation-immune axis, and tumor biology is not yet possible. A summary of current evidence and future directions are presented in the Table.
Conclusions
Exercise is a pleiotropic strategy that may be a promising candidate therapy for the treatment of cancer.[5] The evidence reviewed in this paper suggests that exercise has immunomodulatory effects that could alter multiple critical phases of immune system–tumor cross-talk in both tumor initiation and progression, although this area of investigation clearly remains in its infancy. The recent success of immunotherapy in multiple solid tumors highlights the power of this approach, which, when properly activated, can effectively control treatment of refractory tumors. Thus, understanding the efficacy and mechanisms of exercise as a potential strategy for enhancing the immunogenic response to cancer is germane. There now exists an unprecedented opportunity for exercise-oncology researchers to investigate the key questions that can unlock the potential therapeutic promise of exercise in cancer initiation and progression. To do this will require the design of research studies that involve multidisciplinary teams with expertise in exercise science, immunology, cancer biology, and clinical oncology. A better understanding of the relationship between exercise and the inflammation-immune axis may be able to optimize the efficacy of exercise as an effective adjuvant and combination anticancer treatment.
Financial Disclosure:The authors have no significant financial interest in or other relationship with the manufacturer of any product or provider of any service mentioned in this article. Dr. Demaria receives research support from the Breast Cancer Research Foundation, the Department of Defense Breast Cancer Research Program, and the Chemotherapy Foundation. Dr. Jones receives research support from the National Cancer Institute and AKTIV Against Cancer.
Acknowledgement:The authors would like to thank Sophia Ceder for her help with Figure 2.
References:
1. Rock CL, Doyle C, Demark-Wahnefried W, et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J Clin. 2012;62:243-74.
2. Schmitz KH, Courneya KS, Matthews C, et al. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sport Exerc. 2010;42:1409-26.
3. Hayes SC, Spence RR, Galvao DA, Newton RU. Australian Association for Exercise and Sport Science position stand: optimising cancer outcomes through exercise. J Sci Med Sport/Sport Med Aust. 2009;12:428-34.
4. van den Berg JP, Velthuis MJ, Gijsen BC, et al. Guideline cancer rehabilitation. Ned Tijdschr Geneeskd. 2011;155:A4104.
5. Jones LW. Precision oncology framework for investigation of exercise as treatment for cancer. J Clin Oncol. 2015 Oct 12. [Epub ahead of print]
6. Ballard-Barbash R, Friedenreich CM, Courneya KS, et al. Physical activity, biomarkers, and disease outcomes in cancer survivors: a systematic review. J Natl Cancer Inst. 2012;104:815-40.
7. Betof AS, Dewhirst MW, Jones LW. Effects and potential mechanisms of exercise training on cancer progression: a translational perspective. Brain Behav Immun. 2013;30(suppl):S75-S87.
8. McClellan JL, Steiner JL, Day SD, et al. Exercise effects on polyp burden and immune markers in the ApcMin/+ mouse model of intestinal tumorigenesis. Int J Oncol. 2014;45:861-8.
9. Zheng X, Cui XX, Huang MT, et al. Inhibition of progression of androgen-dependent prostate LNCaP tumors to androgen independence in SCID mice by oral caffeine and voluntary exercise. Nutr Cancer. 2012;64:1029-37.
10. Jones LW, Antonelli J, Masko EM, et al. Exercise modulation of the host-tumor interaction in an orthotopic model of murine prostate cancer. J Appl Physiol (1985). 2012;113:263-72.
11. Betof AS, Lascola CD, Weitzel D, et al. Modulation of murine breast tumor vascularity, hypoxia and chemotherapeutic response by exercise. J Natl Cancer Inst. 2015;107:djv040.
12. Khori V, Amani Shalamzari S, Isanejad A, et al. Effects of exercise training together with tamoxifen in reducing mammary tumor burden in mice: possible underlying pathway of miR-21. Eur J Pharmacol. 2015;765:179-87.
13. Steiner JL, Davis JM, McClellan JL, et al. Effects of voluntary exercise on tumorigenesis in the C3(1)/SV40Tag transgenic mouse model of breast cancer. Int J Oncol. 2013;42:1466-72.
14. Straussman R, Morikawa T, Shee K, et al. Tumour micro-environment elicits innate resistance to RAF inhibitors through HGF secretion. Nature. 2012;487:500-4.
15. McAllister SS, Weinberg RA. Tumor-host interactions: a far-reaching relationship. J Clin Oncol. 2010;28:4022-8.
16. McAllister SS, Gifford AM, Greiner AL, et al. Systemic endocrine instigation of indolent tumor growth requires osteopontin. Cell. 2008;133:994-1005.
17. Sheen-Chen SM, Liu YW, Eng HL, Chou FF. Serum levels of hepatocyte growth factor in patients with breast cancer. Cancer Epidemiol Biomarkers Prev. 2005;14:715-7.
18. Sheen-Chen SM, Chen WJ, Eng HL, Chou FF. Serum concentration of tumor necrosis factor in patients with breast cancer. Breast Cancer Res Treat. 1997;43:211-5.
19. Zhang GJ, Adachi I. Serum interleukin-6 levels correlate to tumor progression and prognosis in metastatic breast carcinoma. Anticancer Res. 1999;19:1427-32.
20. Goodwin PJ, Ennis M, Pritchard KI, et al. Insulin- and obesity-related variables in early-stage breast cancer: correlations and time course of prognostic associations. J Clin Oncol. 2012;30:164-71.
21. Hursting SD, Berger NA. Energy balance, host-related factors, and cancer progression. J Clin Oncol. 2010;28:4058-65.
22. Kruijsen-Jaarsma M, Revesz D, Bierings MB, et al. Effects of exercise on immune function in patients with cancer: a systematic review. Exerc Immunol Rev. 2013;19:120-43.
23. Vesely MD, Kershaw MH, Schreiber RD, Smyth MJ. Natural innate and adaptive immunity to cancer. Annu Rev Immunol. 2011;29:235-71.
24. Sharma P, Allison JP. The future of immune checkpoint therapy. Science. 2015;348:56-61.
25. McDermott D, Lebbe C, Hodi FS, et al. Durable benefit and the potential for long-term survival with immunotherapy in advanced melanoma. Cancer Treat Rev. 2014;40:1056-64.
26. Walsh NP, Gleeson M, Shephard RJ, et al. Position statement. Part one: immune function and exercise. Exerc Immunol Rev. 2011;17:6-63.
27. Takeda K, Kaisho T, Akira S. Toll-like receptors. Annu Rev Immunol. 2003;21:335-76.
28. Lotze MT, Zeh HJ, Rubartelli A, et al. The grateful dead: damage-associated molecular pattern molecules and reduction/oxidation regulate immunity. Immunol Rev. 2007;220:60-81.
29. Trinchieri G. Cancer and inflammation: an old intuition with rapidly evolving new concepts. Annu Rev Immunol. 2012;30:677-706.
30. Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell. 2010;140:883-99.
31. Lochhead P, El-Omar EM. Helicobacter pylori infection and gastric cancer. Best Pract Res Clin Gastroenterol. 2007;21:281-97.
32. Matsuzaki K, Murata M, Yoshida K, et al. Chronic inflammation associated with hepatitis C virus infection perturbs hepatic transforming growth factor beta signaling, promoting cirrhosis and hepatocellular carcinoma. Hepatology. 2007;46:48-57.
33. Itzkowitz SH, Yio X. Inflammation and cancer IV. Colorectal cancer in inflammatory bowel disease: the role of inflammation. Am J Physiol Gastrointest Liver Physiol. 2004;287:G7-G17.
34. Khandekar MJ, Cohen P, Spiegelman BM. Molecular mechanisms of cancer development in obesity. Nat Rev Cancer. 2011;11:886-95.
35. Howe LR, Subbaramaiah K, Hudis CA, Dannenberg AJ. Molecular pathways: adipose inflammation as a mediator of obesity-associated cancer. Clin Cancer Res. 2013;19:6074-83.
36. Martinez ME, Jacobs ET, Baron JA, et al. Dietary supplements and cancer prevention: balancing potential benefits against proven harms. J Natl Cancer Inst. 2012;104:732-9.
37. Gleeson M, Bishop NC, Stensel DJ, et al. The anti-inflammatory effects of exercise: mechanisms and implications for the prevention and treatment of disease. Nat Rev Immunol. 2011;11:607-15.
38. Ross R, Bradshaw AJ. The future of obesity reduction: beyond weight loss. Nat Rev Endocrinol. 2009;5:319-25.
39. Kawanishi N, Yano H, Yokogawa Y, Suzuki K. Exercise training inhibits inflammation in adipose tissue via both suppression of macrophage infiltration and acceleration of phenotypic switching from M1 to M2 macrophages in high-fat-diet-induced obese mice. Exerc Immunol Rev. 2010;16:105-18.
40. Pedersen BK, Febbraio MA. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol. 2012;8:457-65.
41. Pedersen BK. Muscle as a secretory organ. Compr Physiol. 2013;3:1337-62.
42. Steensberg A, Fischer CP, Keller C, et al. IL-6 enhances plasma IL-1Ra, IL-10, and cortisol in humans. Am J Physiol Endocrinol Metab. 2003;285:E433-E437.
43. Cupps TR, Fauci AS. Corticosteroid-mediated immunoregulation in man. Immunol Rev. 1982;65:133-55.
44. Bergmann M, Gornikiewicz A, Sautner T, et al. Attenuation of catecholamine-induced immunosuppression in whole blood from patients with sepsis. Shock. 1999;12:421-7.
45. Timmerman KL, Flynn MG, Coen PM, et al. Exercise training-induced lowering of inflammatory (CD14+CD16+) monocytes: a role in the anti-inflammatory influence of exercise? J Leukoc Biol. 2008;84:1271-8.
46. Rosenwinkel ET, Bloomfield DM, Arwady MA, Goldsmith RL. Exercise and autonomic function in health and cardiovascular disease. Cardiol Clin. 2001;19:369-87.
47. Pavlov VA, Tracey KJ. The vagus nerve and the inflammatory reflex-linking immunity and metabolism. Nat Rev Endocrinol. 2012;8:743-54.
48. Kraus WE, Houmard JA, Duscha BD, et al. Effects of the amount and intensity of exercise on plasma lipoproteins. N Engl J Med. 2002;347:1483-92.
49. Joyner MJ, Green DJ. Exercise protects the cardiovascular system: effects beyond traditional risk factors. J Physiol. 2009;587:5551-8.
50. Lavie CJ, Arena R, Swift DL, et al. Exercise and the cardiovascular system: clinical science and cardiovascular outcomes. Circ Res. 2015;117:207-19.
51. Sang H, Yao S, Zhang L, et al. Walk-run training improves the anti-inflammation properties of high-density lipoprotein in patients with metabolic syndrome. J Clin Endocrinol Metab. 2015;100:870-9.
52. Norata GD, Pirillo A, Ammirati E, Catapano AL. Emerging role of high density lipoproteins as a player in the immune system. Atherosclerosis. 2012;220:11-21.
53. Jones LW, Fels DR, West M, et al. Modulation of circulating angiogenic factors and tumor biology by aerobic training in breast cancer patients receiving neoadjuvant chemotherapy. Cancer Prev Res (Phila). 2013;6:925-37.
54. Ergun M, Eyigor S, Karaca B, et al. Effects of exercise on angiogenesis and apoptosis-related molecules, quality of life, fatigue and depression in breast cancer patients. Eur J Cancer Care (Engl). 2013;22:626-37.
55. Glass OK, Inman BA, Broadwater G, et al. Effect of aerobic training on the host systemic milieu in patients with solid tumours: an exploratory correlative study. Br J Cancer. 2015;112:825-31.
56. Mittal D, Gubin MM, Schreiber RD, Smyth MJ. New insights into cancer immunoediting and its three component phases-elimination, equilibrium and escape. Curr Opin Immunol. 2014;27:16-25.
57. Shankaran V, Ikeda H, Bruce AT, et al. IFNgamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature. 2001;410:1107-11.
58. Smyth MJ, Crowe NY, Godfrey DI. NK cells and NKT cells collaborate in host protection from methylcholanthrene-induced fibrosarcoma. Int Immunol. 2001;13:459-63.
59. Birkeland SA, Storm HH, Lamm LU, et al. Cancer risk after renal transplantation in the Nordic countries, 1964-1986. Int J Cancer. 1995;60:183-9.
60. Raulet DH, Guerra N. Oncogenic stress sensed by the immune system: role of natural killer cell receptors. Nat Rev Immunol. 2009;9:568-80.
61. Carbone E, Neri P, Mesuraca M, et al. HLA class I, NKG2D, and natural cytotoxicity receptors regulate multiple myeloma cell recognition by natural killer cells. Blood. 2005;105:251-8.
62. Cerwenka A, Baron JL, Lanier LL. Ectopic expression of retinoic acid early inducible-1 gene (RAE-1) permits natural killer cell-mediated rejection of a MHC class I-bearing tumor in vivo. Proc Natl Acad Sci USA. 2001;98:11521-6.
63. Ashkenazi A, Dixit VM. Death receptors: signaling and modulation. Science. 1998;281:1305-8.
64. Seliger B. Different regulation of MHC class I antigen processing components in human tumors. J Immunotoxicol. 2008;5:361-7.
65. Woods JA, Ceddia MA, Wolters BW, et al. Effects of 6 months of moderate aerobic exercise training on immune function in the elderly. Mech Ageing Dev. 1999;109:1-19.â©
66. Fahlman M, Boardley D, Flynn MG, et al. Effects of endurance training on selected parameters of immune function in elderly women. Gerontology. 2000;46:97-104.
67. Campbell PT, Wener MH, Sorensen B, et al. Effect of exercise on in vitro immune function: a 12-month randomized, controlled trial among postmenopausal women. J Appl Physiol (1985). 2008;104:1648-55.
68. Bigley AB, Simpson RJ. NK cells and exercise: implications for cancer immunotherapy and survivorship. Discov Med. 2015;19:433-45.
69. Fairey AS, Courneya KS, Field CJ, et al. Randomized controlled trial of exercise and blood immune function in postmenopausal breast cancer survivors. J Appl Physiol (1985). 2005;98:1534-40.
70. Saxton JM, Scott EJ, Daley AJ, et al. Effects of an exercise and hypocaloric healthy eating intervention on indices of psychological health status, hypothalamic-pituitary-adrenal axis regulation and immune function after early-stage breast cancer: a randomised controlled trial. Breast Cancer Res. 2014;16:R39.
71. Miles MP, Mackinnon LT, Grove DS, et al. The relationship of natural killer cell counts, perforin mRNA and CD2 expression to post-exercise natural killer cell activity in humans. Acta Physiol Scand. 2002;174:317-25.
72. Schreiber RD, Old LJ, Smyth MJ. Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science. 2011;331:1565-70.
73. Dunn GP, Old LJ, Schreiber RD. The three Es of cancer immunoediting. Annu Rev Immunol. 2004;22:329-60.
74. Seliger B, Ritz U, Abele R, et al. Immune escape of melanoma: first evidence of structural alterations in two distinct components of the MHC class I antigen processing pathway. Cancer Res. 2001;61:8647-50.
75. Marincola FM, Jaffee EM, Hicklin DJ, Ferrone S. Escape of human solid tumors from T-cell recognition: molecular mechanisms and functional significance. Adv Immunol. 2000;74:181-273.
76. Groh V, Wu J, Yee C, Spies T. Tumour-derived soluble MIC ligands impair expression of NKG2D and T-cell activation. Nature. 2002;419:734-8.
77. Iwai Y, Ishida M, Tanaka Y, et al. Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PD-L1 blockade. Proc Natl Acad Sci USA. 2002;99:12293-7.
78. Hahne M, Rimoldi D, Schroter M, et al. Melanoma cell expression of Fas(Apo-1/CD95) ligand: implications for tumor immune escape. Science. 1996;274:1363-6.
79. Marques CA, Hahnel PS, Wolfel C, et al. An immune escape screen reveals CDC42 as regulator of cancer susceptibility to lymphocyte-mediated tumor suppression. Blood. 2008;111:1413-9.
80. Catlett-Falcone R, Landowski TH, Oshiro MM, et al. Constitutive activation of Stat3 signaling confers resistance to apoptosis in human U266 myeloma cells. Immunity. 1999;10:105-15.
81. Mantovani A, Sozzani S, Locati M, et al. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002;23:549-55.
82. Hagemann T, Lawrence T, McNeish I, et al. “Re-educating” tumor-associated macrophages by targeting NF-kappaB. J Exp Med. 2008;205:1261-8.
83. Mantovani A, Sica A. Macrophages, innate immunity and cancer: balance, tolerance, and diversity. Curr Opin Immunol. 2010;22:231-7.
84. Qian BZ, Pollard JW. Macrophage diversity enhances tumor progression and metastasis. Cell. 2010;141:39-51.
85. Steidl C, Lee T, Shah SP, et al. Tumor-associated macrophages and survival in classic Hodgkin’s lymphoma. N Engl J Med. 2010;362:875-85.
86. Zhang BC, Gao J, Wang J, et al. Tumor-associated macrophages infiltration is associated with peritumoral lymphangiogenesis and poor prognosis in lung adenocarcinoma. Med Oncol. 2011;28:1447-52.
87. Gazzaniga S, Bravo AI, Guglielmotti A, et al. Targeting tumor-associated macrophages and inhibition of MCP-1 reduce angiogenesis and tumor growth in a human melanoma xenograft. J Invest Dermatol. 2007;127:2031-41.
88. Dineen SP, Lynn KD, Holloway SE, et al. Vascular endothelial growth factor receptor 2 mediates macrophage infiltration into orthotopic pancreatic tumors in mice. Cancer Res. 2008;68:4340-6.
89. Rolny C, Mazzone M, Tugues S, et al. HRG inhibits tumor growth and metastasis by inducing macrophage polarization and vessel normalization through downregulation of PlGF. Cancer Cell. 2011;19:31-44.
90. Di Carlo E, Forni G, Lollini P, et al. The intriguing role of polymorphonuclear neutrophils in antitumor reactions. Blood. 2001;97:339-45.
91. Schmielau J, Finn OJ. Activated granulocytes and granulocyte-derived hydrogen peroxide are the underlying mechanism of suppression of T-cell function in advanced cancer patients. Cancer Res. 2001;61:4756-60.
92. Walsh NP, Gleeson M, Pyne DB, et al. Position statement. Part two: maintaining immune health. Exerc Immunol Rev. 2011;17:64-103.
93. Kizaki T, Takemasa T, Sakurai T, et al. Adaptation of macrophages to exercise training improves innate immunity. Biochem Biophys Res Commun. 2008;372:152-6.
94. Sugiura H, Nishida H, Sugiura H, Mirbod SM. Immunomodulatory action of chronic exercise on macrophage and lymphocyte cytokine production in mice. Acta Physiol Scand. 2002;174:247-56.
95. Peake JM. Exercise-induced alterations in neutrophil degranulation and respiratory burst activity: possible mechanisms of action. Exerc Immunol Rev. 2002;8:49-100.
96. Hack V, Strobel G, Weiss M, Weicker H. PMN cell counts and phagocytic activity of highly trained athletes depend on training period. J Appl Physiol (1985). 1994;77:1731-5.
97. Pyne DB, Baker MS, Fricker PA, et al. Effects of an intensive 12-wk training program by elite swimmers on neutrophil oxidative activity. Med Sci Sports Exerc. 1995;27:536-42.
98. Abdalla DR, Aleixo AA, Murta EF, Michelin MA. Innate immune response adaptation in mice subjected to administration of DMBA and physical activity. Oncol Lett. 2014;7:886-90.
99. Lu Q, Ceddia MA, Price EA, et al. Chronic exercise increases macrophage-mediated tumor cytolysis in young and old mice. Am J Physiol. 1999;276:R482-R489.
100. Almeida PW, Gomes-Filho A, Ferreira AJ, et al. Swim training suppresses tumor growth in mice. J Appl Physiol (1985). 2009;107:261-5.
101. Zielinski MR, Muenchow M, Wallig MA, et al. Exercise delays allogeneic tumor growth and reduces intratumoral inflammation and vascularization. J Appl Physiol (1985). 2004;96:2249-56.
102. Iwamoto T, Bianchini G, Booser D, et al. Gene pathways associated with prognosis and chemotherapy sensitivity in molecular subtypes of breast cancer. J Natl Cancer Inst. 2011;103:264-72.
103. Pages F, Kirilovsky A, Mlecnik B, et al. In situ cytotoxic and memory T cells predict outcome in patients with early-stage colorectal cancer. J Clin Oncol. 2009;27:5944-51.
104. Kalos M, June CH. Adoptive T cell transfer for cancer immunotherapy in the era of synthetic biology. Immunity. 2013;39:49-60.
105. Adams S, Gray RJ, Demaria S, et al. Prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancers from two phase III randomized adjuvant breast cancer trials: ECOG 2197 and ECOG 1199. J Clin Oncol. 2014;32:2959-66.
106. Galon J, Costes A, Sanchez-Cabo F, et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science. 2006;313:1960-4.
107. Mlecnik B, Tosolini M, Charoentong P, et al. Biomolecular network reconstruction identifies T-cell homing factors associated with survival in colorectal cancer. Gastroenterology. 2010;138:1429-40.
108. Bindea G, Mlecnik B, Tosolini M, et al. Spatiotemporal dynamics of intratumoral immune cells reveal the immune landscape in human cancer. Immunity. 2013;39:782-95.
109. Facciabene A, Motz GT, Coukos G. T-regulatory cells: key players in tumor immune escape and angiogenesis. Cancer Res. 2012;72:2162-71.
110. Chen W, Jin W, Hardegen N, et al. Conversion of peripheral CD4+CD25- naive T cells to CD4+CD25+ regulatory T cells by TGF-beta induction of transcription factor Foxp3. J Exp Med. 2003;198:1875-86.
111. deLeeuw RJ, Kost SE, Kakal JA, Nelson BH. The prognostic value of FoxP3+ tumor-infiltrating lymphocytes in cancer: a critical review of the literature. Clin Cancer Res. 2012;18:3022-9.
112. Simpson RJ, Lowder TW, Spielmann G, et al. Exercise and the aging immune system. Ageing Res Rev. 2012;11:404-20.
113. Woods JA, Keylock KT, Lowder T, et al. Cardiovascular exercise training extends influenza vaccine seroprotection in sedentary older adults: the immune function intervention trial. J Am Geriatr Soc. 2009;57:2183-91.
114. Wang J, Song H, Tang X, et al. Effect of exercise training intensity on murine T-regulatory cells and vaccination response. Scand J Med Sci Sports. 2012;22:643-52.
115. Abdalla DR, Murta EF, Michelin MA. The influence of physical activity on the profile of immune response cells and cytokine synthesis in mice with experimental breast tumors induced by 7,12-dimethylbenzanthracene. Eur J Cancer Prev. 2013;22:251-8.
116. Goh J, Tsai J, Bammler TK, et al. Exercise training in transgenic mice is associated with attenuation of early breast cancer growth in a dose-dependent manner. PLoS One. 2013;8:e80123.
117. de Jong RA, Leffers N, Boezen HM, et al. Presence of tumor-infiltrating lymphocytes is an independent prognostic factor in type I and II endometrial cancer. Gynecol Oncol. 2009;114:105-10.
118. Anraku M, Cunningham KS, Yun Z, et al. Impact of tumor-infiltrating T cells on survival in patients with malignant pleural mesothelioma. J Thorac Cardiovasc Surg. 2008;135:823-9.
119. Schumacher K, Haensch W, Roefzaad C, Schlag PM. Prognostic significance of activated CD8(+) T cell infiltrations within esophageal carcinomas. Cancer Res. 2001;61:3932-6.
120. Gajewski TF, Schreiber H, Fu YX. Innate and adaptive immune cells in the tumor microenvironment. Nat Immunol. 2013;14:1014-22.
121. Vicari AP, Caux C, Trinchieri G. Tumour escape from immune surveillance through dendritic cell inactivation. Semin Cancer Biol. 2002;12:33-42.
122. Obeid M, Tesniere A, Ghiringhelli F, et al. Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med. 2007;13:54-61.
123. Kroemer G, Galluzzi L, Kepp O, Zitvogel L. Immunogenic cell death in cancer therapy. Annu Rev Immunol. 2013;31:51-72.
124. Martins I, Wang Y, Michaud M, et al. Molecular mechanisms of ATP secretion during immunogenic cell death. Cell Death Differ. 2014;21:79-91.
125. Apetoh L, Ghiringhelli F, Tesniere A, et al. Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat Med. 2007;13:1050-9.
126. Dumitriu IE, Bianchi ME, Bacci M, et al. The secretion of HMGB1 is required for the migration of maturing dendritic cells. J Leukoc Biol. 2007;81:84-91.
127. Diamond MS, Kinder M, Matsushita H, et al. Type I interferon is selectively required by dendritic cells for immune rejection of tumors. J Exp Med. 2011;208:1989-2003.
128. Fuertes MB, Kacha AK, Kline J, et al. Host type I IFN signals are required for antitumor CD8+ T cell responses through CD8{alpha}+ dendritic cells. J Exp Med. 2011;208:2005-16.
129. Chiang LM, Chen YJ, Chiang J, et al. Modulation of dendritic cells by endurance training. Int J Sports Med. 2007;28:798-803.
130. Liao HF, Chiang LM, Yen CC, et al. Effect of a periodized exercise training and active recovery program on antitumor activity and development of dendritic cells. J Sports Med Phys Fitness. 2006;46:307-14.
131. Chen L, Han X. Anti-PD-1/PD-L1 therapy of human cancer: past, present, and future. J Clin Invest. 2015;125:3384-91.
132. Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-23.
133. Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443-54.
134. Hamid O, Robert C, Daud A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med. 2013;369:134-44.
135. Ledford H. Melanoma drug wins US approval. Nature. 2011;471:561.
136. Garon EB. Current perspectives in immunotherapy for non-small cell lung cancer. Semin Oncol. 2015;42(suppl 2):S11-S18.
137. Ji RR, Chasalow SD, Wang L, et al. An immune-active tumor microenvironment favors clinical response to ipilimumab. Cancer Immunol Immunother. 2012;61:
1019-31.
138. Taube JM, Anders RA, Young GD, et al. Colocalization of inflammatory response with B7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci Transl Med. 2012;4:127ra37.
139. Golden EB, Frances D, Pellicciotta I, et al. Radiation fosters dose-dependent and chemotherapy-induced immunogenic cell death. Oncoimmunology. 2014;3:e28518.
140. Tesniere A, Schlemmer F, Boige V, et al. Immunogenic death of colon cancer cells treated with oxaliplatin. Oncogene. 2010;29:482-91.
141. Dewan MZ, Galloway AE, Kawashima N, et al. Fractionated but not single-dose radiotherapy induces an immune-mediated abscopal effect when combined with anti-CTLA-4 antibody. Clin Cancer Res. 2009;15:5379-88.
142. Demaria S, Kawashima N, Yang AM, et al. Immune-mediated inhibition of metastases after treatment with local radiation and CTLA-4 blockade in a mouse model of breast cancer. Clin Cancer Res. 2005;11:728-34.
143. Golden EB, Demaria S, Schiff PB, et al. An abscopal response to radiation and ipilimumab in a patient with metastatic non-small cell lung cancer. Cancer Immunol Res. 2013;1:365-72.
144. Neufert C, Becker C, Neurath MF. An inducible mouse model of colon carcinogenesis for the analysis of sporadic and inflammation-driven tumor progression. Nat Protoc. 2007;2:1998-2004.
145. de Visser KE, Korets LV, Coussens LM. De novo carcinogenesis promoted by chronic inflammation is B lymphocyte dependent. Cancer Cell. 2005;7:411-23.
146. Ma Y, Kepp O, Ghiringhelli F, et al. Chemotherapy and radiotherapy: cryptic anticancer vaccines. Semin Immunol. 2010;22:113-24.
147. Dranoff G. Experimental mouse tumour models: what can be learnt about human cancer immunology? Nat Rev Immunol. 2012;12:61-6.