Woman With Rare Cause of Rectal Bleeding
A 56-year-old Caucasian woman presented to her primary care physician with a 3-month history of intermittent bright red rectal blood with defecation. At her initial visit, a digital rectal examination, anoscopy, and a pelvic examination with DNA testing for high-risk HPV were performed; all results were negative. She was referred for a colonoscopy, which revealed an abnormal area with a 3 × 4–cm mass in the rectum at a distance of 10 cm from the anal verge.
Oncology (Williston Park). 32(1):28-31.

Figure 1. Histologic Appearance of Rectal Squamous Cell Carcinoma
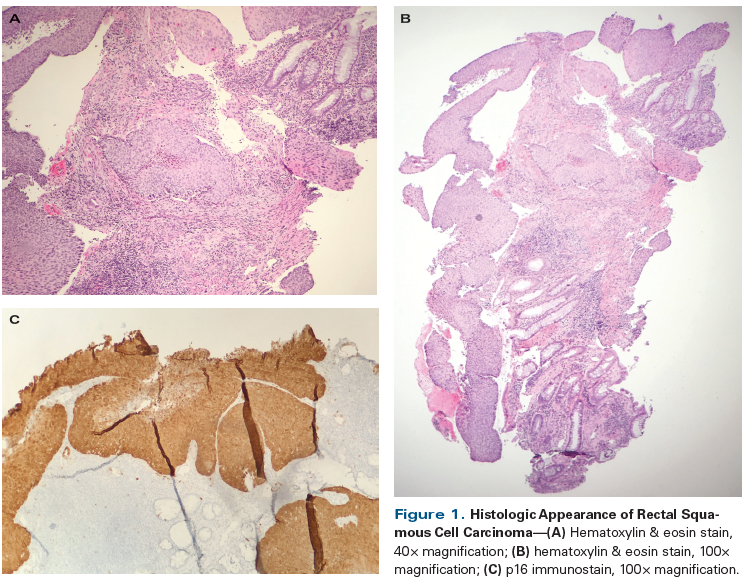
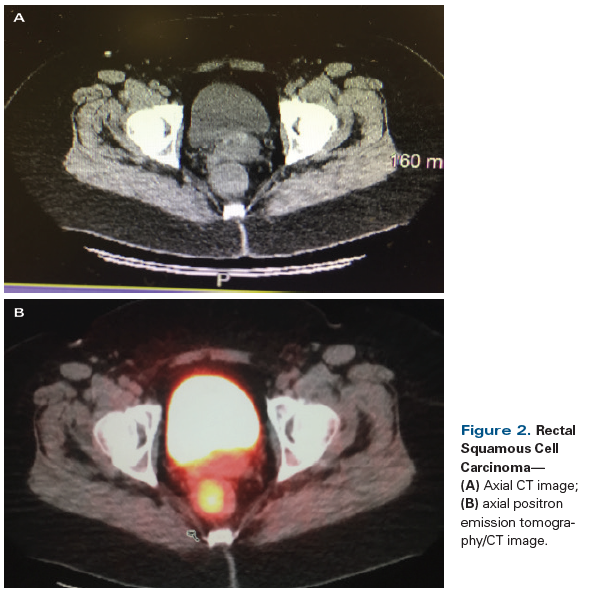
Figure 2. Rectal Squamous Cell Carcinoma
The Case
A 56-year-old Caucasian woman presented to her primary care physician with a 3-month history of intermittent bright red rectal blood with defecation. At her initial visit, a digital rectal examination, anoscopy, and a pelvic examination with DNA testing for high-risk human papillomavirus (HPV) were performed; all results were negative. She was referred for a colonoscopy, which revealed an abnormal area with a 3 × 4–cm mass in the rectum at a distance of 10 cm from the anal verge. Colonoscopic biopsy of this mass was consistent with invasive squamous cell carcinoma (Figures 1A and 1B); p16 immunohistochemistry staining of the biopsy specimen was positive (Figure 1C). Staging workup with a total body CT scan (Figure 2A) and positron emission tomography (PET)/CT scan (Figure 2B) confirmed the presence of a rectal mass, which demonstrated a standardized uptake value of 10. There was no evidence of nodal or distant metastasis. The patient was referred to medical oncology for further treatment recommendations.
For which of the following treatment plans does the available evidence provide the strongest support?
A. Neoadjuvant therapy with continuously infused fluorouracil (5-FU) and concurrent radiation therapy, followed by total mesorectal excision
B. Low anterior resection or abdominoperineal resection followed by adjuvant chemoradiation therapy
C. Radiation therapy only
D. Definitive chemoradiation therapy with continuously infused 5-FU on days 1–4 and days 29–32 and mitomycin on days 1 and 29, along with concurrent radiation therapy
Discussion
Here we present a rare case of squamous cell carcinoma of the rectum, which represents 0.1% to 0.2% of all colorectal cancers and which occurs more frequently in women.[1] While the underlying pathogenesis is unclear, several mechanisms for the development of rectal cancer of squamous histology have been proposed; these include: proliferation of stem cells capable of multidirectional differentiation,[2] chronic irritation by conditions such as ulcerative colitis,[3] radiation exposure,[4] HPV infection,[5] and squamous differentiation from an underlying adenoma or adenocarcinoma.[6] Historically, surgery with curative intent has been the most frequently used treatment approach for squamous cell rectal cancer. Depending on the anatomic location and TNM classification of the tumor, either a low anterior resection or an abdominoperineal resection with permanent ostomy has been performed. Each type of surgery has come with associated significant morbidity (13% to 46%) and mortality (1% to 7%).[7] Mesorectal excision following a course of preoperative neoadjuvant 5-FU–based chemoradiation[8,9] has been the standard of care for the much more common histologic type of rectal cancer-ie, adenocarcinoma. Answer A would have been appropriate if the histology of the tumor had been adenocarcinoma. However, this is not the correct answer for a rectal cancer of squamous cell histology.
Chemoradiation as a postoperative treatment for squamous cell rectal cancer has been investigated[10] and found to be tolerable, with a low toxicity profile. However, this multimodality treatment approach has not been shown to offer any advantage in terms of overall survival compared with surgery alone. Thus, Answer B also is not correct.
In recent years, selective nonoperative management of rectal adenocarcinoma has received increasing attention. Several anecdotal retrospective case series, as well as a few small prospective studies, have demonstrated that a nonoperative approach, such as close surveillance, may be feasible in a select group of patients with rectal adenocarcinoma who have had a complete clinical response to neoadjuvant chemoradiation.[11,12] However, Answer C-no surgery, and with treatment consisting of radiation only-would not be correct, since there are no data to support the use of radiation treatment without chemotherapy in the setting of squamous cell rectal cancer. This approach would not be appropriate even for rectal adenocarcinoma.
KEY POINTS
- Squamous cell carcinoma of the rectum is a rare malignancy; it has some anatomic and histologic similarities to anal squamous cell carcinoma.
- Optimal management of this entity is evolving in parallel to the emerging trend in anal squamous cell carcinoma-ie, toward the use of chemoradiation either as definitive treatment or in conjunction with surgery as a salvage option.
- Recent published data on anal squamous cell carcinoma show that a complete response to chemoradiation can sometimes take up to 6 months from the completion of treatment, suggesting that longer follow-up may be appropriate and that surgery should be reserved for cases of treatment failure after 6 months.
Validation of the Nigro protocol (preoperative use of concurrent radiation with 5-FU and mitomycin chemotherapy, followed by surgical excision if necessary) in multiple randomized controlled trials has made it the accepted standard treatment for patients with anal squamous cell carcinoma. Surgery, which had previously been the preferred management approach for squamous cell anal cancer, now has been relegated to a salvage role.[13,14] The anatomic and histologic similarities between squamous cell rectal and anal cancers have led to an emerging trend of utilizing chemoradiation either as definitive treatment or in conjunction with salvage surgery, if needed, in the management of squamous cell rectal carcinoma. Because of improvements in chemotherapy and radiotherapy, as well as in the imaging technology used to determine stage and response in this disease, a number of recent studies utilizing an anal squamous cell carcinoma–based treatment approach have shown promising results in patients with rectal squamous cell carcinoma.[15-21] A review of cases reported in the literature makes clear that patients undergoing definitive chemoradiation therapy have had outcomes far superior to those that have historically been reported.[22] For patients whose tumors show frank progression during chemoradiation, salvage surgery can be undertaken as the next line of treatment to ensure an optimal outcome. However, in the setting of a partial response to definitive chemoradiation or stable disease, a more prolonged assessment of up to 6 months from completion of treatment can be appropriate before considering salvage surgery. Many patients who eventually have a pathologic complete response have been known to have clinical and radiologic findings suggestive of persistent disease in the early post-chemoradiation phase. This also is in line with the accepted management of anal squamous cell cancers, in which a delayed tumor response may occur as long as 6 months after the completion of chemoradiation.[23] Thus, as with squamous cell anal cancer, nonoperative management of squamous cell cancer of the rectum with definitive chemoradiation represents the most appropriate approach, making Answer D correct here. Surgery should be reserved for cases of treatment failure that persists after 6 months.
Outcome of This Case
The patient was treated with definitive chemoradiation therapy, using the anal squamous cell carcinoma treatment regimen. Chemotherapy consisted of continuous infusion of 5-FU 1,000 mg/m2 on days 1–4 and days 29–32, plus mitomycin 10 mg/m2 on days 1 and 29. Radiotherapy was delivered via a linear accelerator with energy of 6–15 MV, using a three-dimensional conformal technique. The planning target volume 2 (PTV2) included the site of disease and pelvic nodal stations (internal iliac, external iliac, obturator, mesorectal, presacral, and inguinal nodes). The planning target volume 1 (PTV1) encompassed the site of disease with an isotropic margin of 2 cm. PTV2 received 45 Gy in 25 daily fractions of 1.8 Gy each. PTV1 received a 59.4-Gy boost via the addition of 8 fractions, using a multiple-field technique. The patient was reevaluated at 8 weeks after the end of chemoradiation with a restaging PET/CT scan and at 12 weeks with a proctoscopic examination. There was no evidence of tumor on PET/CT scan. Proctoscopic examination at 12 weeks also revealed no residual disease. Currently, she remains free of disease 19 months out from the date of her diagnosis.
Financial Disclosure:The authors have no significant financial interest in or other relationship with the manufacturer of any product or provider of any service mentioned in this article.
E. David Crawford, MD, serves as Series Editor for Clinical Quandaries. Dr. Crawford is Professor of Surgery, Urology, and Radiation Oncology, and Head of the Section of Urologic Oncology at the University of Colorado School of Medicine; Chairman of the Prostate Conditions Education Council; and a member of ONCOLOGY's Editorial Board.
If you have a case that you feel has particular educational value, illustrating important points in diagnosis or treatment, you may send the concept to Dr. Crawford at david.crawford@ucdenver.edu for consideration for a future installment of Clinical Quandaries.
References:
1. Kang H, O’Connell JB, Leonardi MJ, et al. Rare tumors of the colon and rectum: a national review. Int J Colorectal Dis. 2007;22:183-9.
2. Ouban A, Nawab RA, Coppola D. Diagnostic and pathogenetic implications of colorectal carcinomas with multidirectional differentiation: a report of 4 cases. Clin Colorectal Cancer. 2002;1:243-8.
3. Michelassi F, Montag AG, Block GE. Adenosquamous-cell carcinoma in ulcerative colitis: report of a case. Dis Colon Rectum. 1988;31:323-6.
4. Leung KK, Heitzman J, Madan A. Squamous cell carcinoma of the rectum 21 years after radiotherapy for cervical carcinoma. Saudi J Gastroenterol. 2009;15:196-8.
5. Audeau A, Han HW, Johnston MJ, et al. Does human papilloma virus have a role in squamous cell carcinoma of the colon and upper rectum? Eur J Surg Oncol. 2002; 28:657-60.
6. Lundquest DE, Marcus JN, Thorson AG, Massop D. Primary squamous cell carcinoma of the colon arising in a villous adenoma. Hum Pathol. 1988;19:362-4.
7. Martling AL, Holm T, Rutqvist LE, et al. Effect of a surgical training programme on outcome of rectal cancer in the County of Stockholm. Stockholm Colorectal Cancer Study Group, Basingstoke Bowel Cancer Research Project. Lancet. 2000;356:93-6.
8. Heald RJ, Ryall RD. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet. 1986;1:1479-82.
9. Glynne-Jones R, Mawdsley S, Pearce T, Buyse M. Alternative clinical end points in rectal cancer-are we getting closer? Ann Oncol. 2006;17:1239-48.
10. Schneider TA 2nd, Birkett DH, Vernava AM 3rd. Primary adenosquamous and squamous cell carcinoma of the colon and rectum. Int J Colorectal Dis. 1992;7:144-7.
11. Maas M, Beets-Tan RG, Lambregts DM, et al. Wait-and-see policy for clinical complete responders after chemoradiation for rectal cancer. J Clin Oncol. 2011;29:4633-40.
12. Smith JD, Ruby JA, Goodman KA, et al. Nonoperative management of rectal cancer with complete clinical response following neoadjuvant therapy. Ann Surg. 2012;256:965-7.
13. Glynne-Jones R, Nilsson PJ, Aschele C, et al. Anal cancer: ESMO-ESSO-ESTRO clinical practice guidelines for diagnosis, treatment and follow-up. Radiother Oncol. 2014;111:330-9.
14. Nigro ND. An evaluation of combined therapy for squamous cell cancer of the anal canal. Dis Colon Rectum. 1984;27:763-6.
15. Yeh J, Hastings J, Rao A, Abbas MA. Squamous cell carcinoma of the rectum: a single institution experience. Tech Coloproctol. 2012;16:349-54.
16. Rasheed S, Yap T, Zia A, et al. Chemo-radiotherapy: an alternative to surgery for squamous cell carcinoma of the rectum-report of six patients and literature review. Colorectal Dis. 2009;11:191-7.
17. Péron J, Bylicki O, Laude C, et al. Nonoperative management of squamous-cell carcinoma of the rectum. Dis Colon Rectum. 2015;58:60-4.
18. Wang ML, Heriot A, Leong T, Ngan SY. Chemoradiotherapy in the management of primary squamous-cell carcinoma of the rectum. Colorectal Dis. 2011;13:296-301.
19. Clark J, Cleator S, Goldin R, et al. Treatment of primary rectal squamous cell carcinoma by primary chemoradiotherapy: should surgery still be considered a standard of care? Eur J Cancer. 2008;44:2340-3.
20. Jeong BG, Kim DY, Kim SY. Concurrent chemoradiotherapy for squamous cell carcinoma of the rectum. Hepatogastroenterology. 2013;60:512-6.
21. Musio D, De Felice F, Manfrida S, et al. Squamous cell carcinoma of the rectum: the treatment paradigm. Eur J Surg Oncol. 2015;41:1054-8.
22. Guerra GR, Kong CH, Warrier SK, et al. Primary squamous cell carcinoma of the rectum: an update and implications for treatment. World J Gastrointest Surg. 2016;8:252-65.
23. Glynne-Jones R, Sebag-Montefiore D, Meadows HM, et al. Best time to assess complete clinical response after chemoradiotherapy in squamous cell carcinoma of the anus (ACT II): a post-hoc analysis of randomized controlled phase 3 trial. Lancet Oncol. 2017;18:347-56.