Diagnosis and Management of Mycosis Fungoides
Mycosis fungoides (MF), the most common cutaneous T-cell lymphoma, is a low-grade cutaneous lymphoma characterized by skin-homing CD4+ T cells. It is notable for highly symptomatic progressive skin lesions, including patches, plaques, tumors, and erytheroderma, and has a poorer prognosis at later stages. Diagnosis remains difficult owing to MF’s nonspecific skin presentation and identification of the optimal treatment strategy is challenging given the paucity of controlled trials and numerous and emerging treatment options. Management includes topical therapy with the addition of systemic therapy for patients with later-stage disease including tumors; erythroderma; and nodal, visceral, or blood involvement. Topical therapies include mechlorethamine (nitrogen mustard), carmustine (BCNU), steroids, bexarotene gel (Targretin Gel), psoralen plus ultraviolet A (PUVA), ultraviolet B (UVB), and either localized or total skin electron radiotherapy. Systemic therapies include interferon, retinoids, oral bexarotene (Targretin), denileukin diftitox (Ontak), vorinostat (Zolinza), extracorporeal photochemotherapy (photopheresis), and cytotoxic chemotherapy. Herein, we outline clinically relevant aspects of MF, including clinical presentation, pathology, diagnosis, and staging. We describe in detail existing and emerging therapeutics and offer specific recommendations for management of each stage of MF.
Mycosis fungoides (MF), the most common cutaneous T-cell lymphoma, is a low-grade cutaneous lymphoma characterized by skin-homing CD4+ T cells. It is notable for highly symptomatic progressive skin lesions, including patches, plaques, tumors, and erytheroderma, and has a poorer prognosis at later stages. Diagnosis remains difficult owing to MF’s nonspecific skin presentation and identification of the optimal treatment strategy is challenging given the paucity of controlled trials and numerous and emerging treatment options. Management includes topical therapy with the addition of systemic therapy for patients with later-stage disease including tumors; erythroderma; and nodal, visceral, or blood involvement. Topical therapies include mechlorethamine (nitrogen mustard), carmustine (BCNU), steroids, bexarotene gel (Targretin Gel), psoralen plus ultraviolet A (PUVA), ultraviolet B (UVB), and either localized or total skin electron radiotherapy. Systemic therapies include interferon, retinoids, oral bexarotene (Targretin), denileukin diftitox (Ontak), vorinostat (Zolinza), extracorporeal photochemotherapy (photopheresis), and cytotoxic chemotherapy. Herein, we outline clinically relevant aspects of MF, including clinical presentation, pathology, diagnosis, and staging. We describe in detail existing and emerging therapeutics and offer specific recommendations for management of each stage of MF.
Mycosis fungoides (MF), a low-grade cutaneous lymphoma characterized by skin-homing CD4+ T cells of unknown etiology, and its leukemic variant, Szary syndrome, constitute 4% of non-Hodgkin lymphoma but represent the most common extranodal lymphoma. MF is highly symptomatic and progressive, with poor prognoses at later stages. Patients with patch/plaque disease (T1) experience survival similar to the general population,[1] while median survival for those with extensive patches and plaques (T2), tumors (T3), and erythroderma (T4), is approximately 12,[2] 3, and 5 years.[3]
Diagnosis remains difficult owing to MF’s nonspecific skin lesions, and the optimal treatment strategy is difficult given the paucity of controlled trials and numerous and emerging treatment options.
In this article, we outline clinically relevant aspects of MF, including clinical presentation, pathology, diagnosis, and staging, and we describe existing and emerging therapeutics for its management.
Presentation and Pathology
FIGURE 1
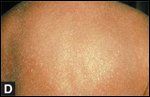
Clinical manifestations of mycosis fungoides
-Image
(A)
shows typical early patch with erythema and mild scale;
(B)
shows a typical plaque, with raised, palpable borders, central clearing, and overlying scale;
(C)
shows a large tumor with necrosis and ulceration; and
(D)
shows generalized erythroderma.
While MF disproportionately presents in black men between the ages of 55 and 60,[4] it can afflict people of any gender, race, and age. MF is characterized by heterogeneous and progressive skin lesions that begin as patches and progress to plaques and tumors (Figure 1). During the premycotic period, patients present with erythematous scaling lesions in sunshielded regions of the body that can wax and wane for years, yielding suboptimal clinical and pathologic diagnosis. In the patch phase, patches begin to involve the trunk, pelvis, and proximal extremities. While MF often is confused with eczema, pathologic diagnosis is possible with evidence of epidermotropism, atypical cells with cerebriform nuclei clustered along the basement membrane. Pautrier’s microabscesses, pathognomonic of MF, may also be seen. If untreated, patches progress to plaques with raised borders and central clearing. Microscopy often reveals a lymphoid infiltrate invading the reticular dermis.
Tumor formation results from further vertical growth of neoplastic cells into the subcutaneous fat. Tumors often present on the face, digits, and perineum and are prone to ulceration and infection. Erythroderma, defined as > 80% body surface involvement with patches and/or plaques, is associated with pruritus, leonine facies, skin atrophy, lichenification, and hyperkeratosis of the palms and soles. Szary syndrome is defined as erythroderma and hematolgic involvement with malignant Szary cells, mononuclear cells with an enfolded or grooved nucleus. It typically arises de novo and often presents with advanced T stage and extracutaneous involvement. Nodal, visceral, and bone marrow involvement are rare but are associated with advanced disease and increased skin and blood involvement.
MF variants beyond the scope of this article include follicular mucinosis, an aggressive variant characterized by follicular papules often involving the neck; pagetoid reticulosis, an indolent disease with an excellent prognosis typically presenting with a psoriaform lesion on a distal extremity; and granulomatous slack skin, characterized by lax skin in the axilla, neck, breasts, and inguinal regions.
Diagnosis and Staging
TABLE 1

Diagnostic Workup for Suspected Mycosis FungoidesTABLE 2

Proposed Algorithm for Diagnosis of Early Mycosis Fungoides
The diagnostic workup (Table 1) includes a history and physical examination to determine the extent of skin involvement, and laboratory evaluation including a complete blood count with attention to the number of Szary cells. The skin and enlarged lymph nodes are biopsied to yield a pathologic diagnosis via immunophenotyping and molecular assays. Patients with advanced disease benefit from systemic evaluation with chest X-ray or CT scan and there is increasing use of FDG-PET (fluorodeoxyglucose-positron emission tomography) in staging. A diagnostic algorithm based on clinical, pathologic, and molecular criteria has been proposed to aid in accurate diagnosis of early MF (Table 2).[5]
TABLE 3

TNM(B) Classification for Mycosis FungoidesTABLE 4
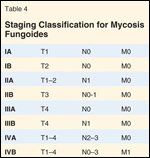
Staging Classification for Mycosis FungoidesTABLE 5
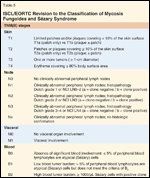
ISCL/EORTC Revision to the Classification of Mycosis Fungoides and Szary SyndromeTABLE 6
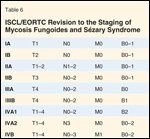
ISCL/EORTC Revision to the Staging of Mycosis Fungoides and Szary Syndrome
MF staging has traditionally been through the American Joint Committee on Cancer (AJCC), which identifies the extent and character of skin lesions, extracutaneous disease, and leukemic transformation as prognostic factors (Tables 3 and 4).[6] Recently the International Society for Cutaneous Lymphoma and the European Organisation for the Research and Treatment of Cancer (ISCL/EORTC) proposed revisions to this system (Tables 5 and 6), including: 1) an “a/b” descriptor added to T1 and T2 disease, with “a” indicating patches alone, and “b” representing patches and/or plaques; 2) N2 indicating limited nodal involvement and N3 indicating partial or complete nodal effacement; 3) B0 representing < 5% of lymphocytes involved by Szary cells, B1 limited blood involvement, and B2 significant blood involvement; 4) an “a/b” descriptor denoting the absence (“a”) or presence (“b”) of a clonal T-cell population in a lymph node or blood detected by PCR (polymerase chain reaction) or Southern blot.[7]
Treatment Overview
Skin-directed therapy is the cornerstone of MF management. For patients with skin-limited disease, topical therapy alone can produces remission and even cure. For patients with localized unilesional MF, topical therapies alone produce long-term disease-free survival rates in excess of 85%.[8] For those with more than one patch or plaque but with less than 10% body surface area involvement (T1, N0 and T1, N1), topical therapies alone produce long-term disease-free survival rates ranging from 30% to 50%. For patients with extensive patches or plaques, topical therapies infrequently produce long-term remissions. Such patients should receive intensive topical therapy to induce a complete remission followed by less intensive adjuvant topical therapy to sustain remission. Few curative options exist for patients with tumors, erythroderma, nodal, visceral, or blood disease. Therefore, treatment goals include cutaneous symptom palliation and systemic disease control. Among topical therapies, total skin electron beam therapy (TSEBT) has the highest complete response rates.[9,10] Adjuvant systemic and/or topical therapy should be administered to sustain remission. Allogeneic stem cell transplant may offer cure for a select population.[11]
TABLE 7
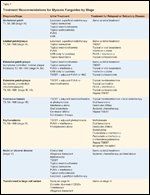
Treatment Recommendations for Mycosis Fungoides by Stage
A complete discussion of topical, systemic and emerging therapies follows and Table 7 offers a full list of treatment options.
Skin-Directed Therapy
Mechlorethamine
Topical mechlorethamine hydrochloride (HN2), also known as nitrogen mustard, is an alkylating agent used as an initial therapy for treatment of MF patches and plaques. Data from Stanford University demonstrate a 65% complete response rate, 45% 10-year relapse-free survival, and 95% 10-year disease-specific survival in patients with T1 disease, and a 34% complete response rate and 20% 10-year relapse-free survival in those with T2 disease.[12]
An irritant dermatitis occurs in approximately 50% of patients treated with aqueous HN2, but can be reduced if the nitrogen mustard is dissolved in ointment.[12] Other toxicities may include xerosis, hyperpigmentation, and, rarely, bullous reactions and Stevens-Johnson syndrome. Bone marrow suppression is not a concern owing to minimal systemic absorption. HN2 carcinogenicity remains debated, with one series reporting no increased risk,[12] and another reporting an eight-fold increase in the risk of nonmelanoma skin cancers.[13] HN2 may potentiate the carcinogenicity of other therapies including TSEBT or psoralen and ultraviolet A radiation (PUVA).[14]
Carmustine
Topical carmustine (BCNU), another alkylating agent, is associated with complete response rates of 86% and 47% and 5-year relapse-free survival of 35% and 10% for T1 and T2 patients, respectively.[15] Because carmustine is systemically absorbed and can produce bone marrow suppression, it should be applied to
< 10% of the body, treatment duration should be limited to 4 months, and patients’ complete blood counts should be monitored while they are on therapy. Cutaneous hypersensitivity is uncommon, but telangiectasias and hyperpigmentation may occur.
Topical Steroids
High-potency topical steroids, often used as first-line therapy for T1 and T2 disease, offer effective palliation and can induce lesion regression with complete response rates of 63% and 25% for T1 and T2 patients.[16] Application is limited to active lesions, as widespread application can result in serum cortisol depression.[16] Long-term use may result in skin atrophy and telangiectasias.
Topical Rexinoids
Bexarotene is a rexinoid, a vitamin A–derived compound that binds to retinoid X receptors, resulting in gene transcription that controls cellular differentiation and proliferation. Bexarotene gel was approved after phase I/II trials demonstrated a 44%–54% response rate in patients with refractory cutaneous MF.[17,18] An irritant dermatitis can occur; therefore, bexarotene should be reserved for patients with less than 15% body surface area involvement. Like systemic retinoids, bexarotene is avoided during pregnancy owing to its possible teratogenicity.
Light Therapy
Ultraviolet light, including UVB (wavelength 320–290 nm), narrow band UVB (wavelength 311 nm), and psoralen plus UVA (PUVA, wavelength 400–320 nm) is used in MF therapy. The photochemotherapeutic agent 8-methoxypsoralen (8-MOP) is ingested prior to UVA exposure. UVA activates 8-MOP, resulting in DNA cross-linking and apoptotic cell death. Because of its limited skin penetration, UVB is reserved for treating thin patches and has resulted in a complete response rate of roughly 80% and a 2-year median response duration.[19] PUVA, penetrating more deeply, effectively treats some plaque lesions producing complete responses rates of 80%–100% for T1 N0 disease and 60%–100% for T2, N0 disease.[16,20] Although relapse is common, retreatment is usually successful. Combination therapy with interferon-α is safe and associated with increased complete response rates and response duration when treating T2 disease.[21]
PUVA and UVB are administered 2 to 3 days a week. After achieving a complete response, treatment frequency is decreased to once every 2 to 4 weeks and then every 4 to 8 weeks for several years. Acute toxicities include skin erythema, hyperpigmentation, xerosis, pruritus, blistering, photoaging, and increased skin cancer risk. Eye goggles worn during treatment sessions can reduce the risk of cataract formation. Side effects of therapy with 8-MOP include nausea, vomiting, and transaminase elevations. For patients with 8-MOP intolerance, a topical “psoralen bath” is a reasonable alternative.
Radiotherapy
Radiotherapy can be administered locally to a single lesion or group of lesions or to the entire skin surface via TSEBT. Complete response rates and long-term disease-free survival for localized radiotherapy approach 91% and 85%, respectively.[22-24] A dose-response relationship exists, with best success with doses between 20 and 30 Gy.[22,23] A 1- to 2-cm field margin for local radiotherapy and a low dose per fraction, ranging from 1.2 to 2.0 Gy per day is recommended to minimize late effects and to spare skin tolerance in the event that future further irradiation is required. Preferably, a single radiation field is used, though abutting fields are often required to treat convex surfaces such as the scalp, breast, hand, or foot.
TSEBT is a complex radiation treatment which effectively treats stages T1–4 MF. For patients with limited patches and plaques, TSEBT produces approximately 90% complete response rates.[1,25] TSEBT produces complete response rates of approximately 70%–90% in patients with T2 disease and from 50%–100% in those with T3 disease.[9,25,26] For patients presenting with extensive patches and plaques (T2, N0) and tumors (T3, N0), TSEBT seems to offer superior eradication of disease and palliation, but boosts must be considered for patients with T3 disease. One study showed TSEBT resulting in higher complete response rates than HN2 (76% vs 39% for T2, and 44% vs 8% for T3, respectively) but no impact on overall survival.[9] For patients with erythroderma (T4, N0), TSEBT can induce complete responses and may result in prolonged disease-free survival in those patients without blood involvement.[10] Recurrences can be successfully salvaged with a second course of TSEBT.[27]
Long-term remission is uncommon with TSEBT monotherapy. However, adjuvant maintenance therapy with HN2 or PUVA improves outcomes in T1 and T2 disease. At Stanford, there was a 60% 10-year recurrence risk among patients treated with TSEBT followed by HN2, compared with a 90% 10-year recurrence risk among patients treated with TSEBT alone.[9] Similarly, at Yale, 7% of patients with T1 or T2 MF treated with maintenance PUVA experienced a 3-year recurrence, compared with 43% for TSEBT alone. While limited data exist regarding adjuvant therapy for T3 or T4 disease, maintenance therapy with HN2,[9] photophoresis,[28] interferon-α, bexarotene, or denileukin diftitox is reasonable.[29] Duration of adjuvant therapy is not well established.
FIGURE 2

Treatment positions for total skin electron beam therapy
• TSEBT Technique-In the Yale TSEBT program, a linear accelerator produces 6 MeV electrons with the patient standing 3.8 meters from the gantry. Patients are placed in six treatment positions that maximize skin unfolding and improve dose homogeneity (Figure 2). The anteroposterior, right posterior oblique, and left posterior oblique positions are treated on treatment day one and the posteroanterior, right anterior oblique, and left anterior oblique positions are treated with the same dose on treatment day two. Over a 2-day cycle, 2 Gy is delivered to the entire skin surface. Patients are treated 4 days per week for 9 weeks to a total dose of 36 Gy.
“Boost” treatment to underdosed regions includes the soles of feet and perineum.An electron reflector above the patient’s head provides for appropriate scalp dose. Other underdosed regions that may require boosts include the ventral penis, upper medial thighs, inframammary folds, pannicular folds, and the lateral and flatter regions of the face and trunk and tumors thicker than 4 mm. “Upfront boosts” can be administered for palliative relief of painful or bleeding lesions and supplemental boosts can be administered to persistent lesions. Conversely, the hands, wrists, ears, ankles, and dorsal penis may be overdosed and thus require shielding to limit their dose to ≤ 36 Gy. Eye and nail shielding are also incorporated.
• TSEBT Toxicity-TSEBT commonly results in pruritus, dry desquamation, hyperpigmentation, alopecia, xerosis, bullae or edema of the hands and feet, decreased perspiration, and loss of fingernails and toenails. Long-term complications are typically mild with a 36-fraction course, but they may include permanent nail dystrophy, xerosis, telangiectasias, partial scalp alopecia, and fingertip dysesthesias. Male infertility is possible owing to the superficial location of the testicles. Secondary skin cancers have been reported, particularly in patients exposed to additional potentially carcinogenic therapies, such as PUVA or HN2.[14]
Systemic Therapy
Interferon
Interferon-α-2a (IFN-α; Alferon, Roferon-A) is an effective treatment for patch and plaque disease, owing to its direct antitumor and immunomodulatory effects.[30] IFN-α may be used alone,[31] but it offers more durable responses in combination with retinoids, PUVA,[21] or extracorporeal photopheresis.[32] A randomized trial of 82 patients showed that IFN-α plus PUVA resulted in a complete response rate of 70%, compared with 38% in patients treated with acitretin (Etretin, Soriatane) plus PUVA.[33] IFN-α may cause flu-like symptoms, psychiatric disturbances including depression, and transaminitis, myelosuppression, proteinuria, and myelopathy.
Retinoids
Oral retinoids such as isotretinoin and acitretin influence cellular differentiation and can be safely combined with other therapies such as PUVA,[34] IFN-α, and TSEBT.[35] Response rates approach 60%, although complete response rates are less than 20%.[36] Side effects include photosensitivity, xerosis, myalgias, headaches, impaired night vision, corneal opacities, teratogenicity, elevated transaminases, hyperlipidemia, and pancreatitis.
Rexinoids: Bexarotene
Oral bexarotene is approved for all stages of refractory MF and can be combined safely with other therapies.[36] Phase II and III trials with oral bexarotene administered to patients with refractory disease at dosages of 300 mg/m2/day or a higher showed 45% and 55% response rates and reductions in both skin involvement and pruritus.[37] Combining bexarotene with PUVA, photophoresis, or IFN-α can increase response rates to 69%.[38] Up to 80% of patients experience hypertriglyceridemia, increasing their risk of pancreatitis and atherosclerosis. Central hypothyroidism afflicts roughly 75% of treated patients. Therefore cholesterol and thyroid function levels require monitoring. Other side effects include headaches, mild neutropenia and transaminase elevations, skin peeling, and pruritus. Bexarotene should be discontinued prior to radiotherapy since combination therapy can exacerbate skin reactions.
Denileukin Diftitox
Denileukin diftitox is an intravenous recombinant fusion protein that combines diphtheria toxin with the receptor-binding portion of interleukin-2 (IL-2) and is approved for MF patients with refractory disease whose cells express CD25.[39] A recent study showed response rates in CD25-negative patients as well, however.[40] Binding to the IL-2 receptor results in diphtheria toxin endocytosis, protein synthesis inhibition, and cell death. Denileukin diftitox administration occurs on days 1–5 every 3 weeks for up to eight cycles. Some evidence suggests a dose-response relationship, with one phase III study showing response rates of 38% with 18 Ï g/kg/day compared with 10% with 9 Ï g/kg/day and a 68% improvement in pruritis.[39] It can be combined and have a possible synergistic effect with bexarotene[41] and various skin-directed therapies to produce long-term disease control.[36]
In the previously mentioned trial, 21% of patients withdrew because of adverse events: 60% experienced a hypersensitivity reaction, 25% a vascular leak syndrome, and 11% a thrombosis event.[39] Other toxicities include constitutional symptoms, infections, transaminase elevations, renal impairment, thyroiditis, and lymphopenia. Corticosteroid pretreatment, however, reduces the risk of acute toxicity.[42]
Histone Deacetylase (HDAC) Inhibitors
The histone deacetylase inhibitor (HDAC) vorinostat (Zolinza), also known as suberoylanilide hydroxamic acid, increases histone acetylation, leading to reduced availability of nuclear DNA to bind to transcription factors. Reduced transcription decreases intracellular protein levels, ultimately resulting in cell cycle arrest and apoptosis. Vorinostat is approved for progressive, persistent, or recurrent MF following treatment with at least two prior systemic therapies. The first published phase II trial of vorinostat showed that 400-mg daily was the best tolerated dosage.[43] In this study, 24% of patients had a partial response and nearly half reported an improvement in pruritus. A second phase II trial in refractory MF patients, stage IB–IVA, showed a 36% overall response rate and a 26-week duration of response.[44] In addition, 32% experienced pruritis relief. Some data suggest a synergistic effect with denileukin diftitox or methylation inhibitors.[36] Romidepsin (Istodax), a promising new HDAC inhibitor, produced a 34% overall response rate and a 13.7-month duration of response in 71 patients with > IIB refractory disease.[45] Romidepsin is approved for the treatment of cutaneous T-cell lymphoma refractory to at least one prior systemic therapy. Adverse effects include diarrhea, nausea, fatigue, and thrombocytopenia and possible QTc prologation. In the aforementioned study, few patients experienced > Grade 3 toxicities on vorinostat, however 5% experienced pulmonary embolism.[43]
Extracorporeal Photochemotherapy
Extracorporeal photophoresis is an immune therapy used in the treatment of erythrodermic MF and/or Szary syndrome by which leukocytes are removed by leukapheresis, combined with 8-MOP, exposed to ultraviolet A, and reinfused into the patient on two consecutive days every 2 to 4 weeks, or more frequently if the patient has extensive disease.[46,47] This process is thought to result in T-cell DNA cross-linking and ultimately apoptosis of the harvested MF T-cells. Overall response rates approximate 50%, with complete response rates approximating 15%.[36] Although typically well-tolerated, fluid shifts can result in hypotension, arrhythmias, and heart failure. Extracorporeal photophoresis has been safely combined with IFN-α, bexarotene, and TSEBT.[28,32,36]
Systemic Chemotherapy
Systemic chemotherapy is reserved for advanced or refractory MF. Response rates are typically high for single-agent or combination therapy, however responses are short-lived and can be associated with significant toxicity, including myelosuppression and bacteremia.[36,48] In advanced disease, a phase II study showed an overall response rate of 80% but there was a high toxicity profile, with 61% experiencing > grade 3 neutropenia and 40% experiencing bacteremia.[48] In addition, in a randomized trial comparing concurrent TSEBT and systemic chemotherapy vs sequential topical therapy, 23% of patients receiving chemotherapy required hospitalization.[49] No data suggest that combination chemotherapy is superior to sequential therapy, and combination therapy has increased toxicity.
Typical chemotherapeutic agents include oral agents such as methotrexate, etoposide (Toposar, VePesid) or chlorambucil which avoid the need for central venous lines and the associated risk of bacteremia, but harbor neutropenia as a risk. Pegylated liposomal doxorubicin (Doxil) is associated with a lower risk of myelosuppression and cardiotoxicity than conventional doxorubicin and in studies has resulted in an 88% overall response without any serious adverse events.[50] Gemcitabine (Gemzar), a nucleoside analogue, has demonstrated complete response rates up to 23% and overall response rates approximating 70% with limited Grade 3–4 myelosuppression.[51] Gemcitabine is associated with reversible hepatotoxicity. Pentostatin (Nipent), a purine analogue, has demonstrated a 54% response rate, but it is more effective in patients with Szary syndrome than in advanced MF.[52] Fludarabine (Fludara), a pyrimidine analogue, has not shown as much promise.
High-Dose Chemotherapy Followed by Bone Marrow Transplant
Reference Guide
Therapeutic Agents
Mentioned in This Article
Acitretin (Etretin, Soriatane)
Alemtuzumab (Campath)
Bexarotene gel (Targretin Gel)
Bortezomib (Velcade)
Carmustine
Denileukin diftitox (Ontak)
Etoposide (Toposar, VePesid)
Fludarabine (Fludara)
Gemcitabine (Gemzar)
Interferon-α
Interferon-α-2a (Alferon, Roferon-A)
Lenalidomide (Revlimid)
Mechlorethamine [nitrogen mustard]
Oral bexarotene (Targretin)
Pegylated liposomal doxorubicin
(Doxil)
Pentostatin (Nipent)
Pralatrexate (Folotyn)
Romidepsin (Istodax)
Vorinostat (Zolinza)
Zanolimumab (HuMax-CD4)
Brand names are listed in parentheses only if a drug is not available generically and is marketed as no more than two trademarked or registered products. More familiar alternative generic designations may also be included parenthetically.
While limited data exist regarding the use of high-dose chemotherapy with autologous or allogeneic transplant in the treatment of advanced, refractory MF, allogeneic transplant may be reasonable for select younger patients. Although autologous stem cell transplant results in high complete response rates, relapse within 1 year is common.[53] In contrast, allogeneic transplants may result in prolonged disease-free survival. The City of Hope Cancer Center reported on eight heavily pretreated, refractory MF patients who underwent allogeneic transplant.[11] Two patients died, however, the other six were alive without evidence of lymphoma at a median of 56 months post-transplant.
Emerging Therapies
Pralatrexate (Folotyn), a folate analogue like methotrexate, has greater cytotoxicity than methotrexate owing to a higher affinity for the reduced folate carrier and increased poly glutamation.[54] In a study involving 11 patients, there was a 45% overall response rate with several responses after only one cycle of pralatrexate. Few hematologic toxicities have been noted.[55] Cytokines, including interleukin-12, interleukin-2, show promise in treatment of MF.[29,56] In a preliminary phase II study, lenalidomide (Revlimid), an enhancer of T helper 1 cellular immunity, approved for treatment of myelodysplastic syndrome and multiple myeloma, showed promising results in three of eight patients with cutaneous T-cell lymphoma.[57] The proteosome inhibitor bortezomib (Velcade) shows promise, with response rates approaching 67% and no grade 4 toxicities in a phase II trial in 12 patients with relapsed, refractory cutaneous T-cell lymphoma or peripheral T-cell lymphoma.[58] Forodesine (also known as immucillin-H), a purine nucleoside phosphorylase inhibitor, leads to T-cell depletion and apoptosis, and it has yielded promising results with limited toxicity in patients with refractory disease.[59] Alemtuzumab (Campath), a monoclonal antibody targeting CD52, shows activity in this patient population but carries the risk of opportunistic infection.[60,61] Zanolimumab (HuMax-CD4), an anti-CD4 monoclonal antibody, results in fewer infections and in early studies was associated with a 50% response rate and a median response duration of 81 weeks.[62]
Summary
Diagnosis and management of mycosis fungoides represent a significant challenge, owing to the varyious clinical presentations of mycosis fungoides and the numerous available therapeutic modalities. Therefore, a multidisciplinary approach is essential. The past decade has brought substantial gains in the areas of diagnosis and pathologic classification which have led to development of novel, targeted, and biologic therapy. The future promises even further clinical advances that should ultimately translate into improved disease control and quality of life for patients with mycosis fungoides.
Financial Disclosure:The authors have no significant financial interest or other relationship with the manufacturers of any products or providers of any service mentioned in this article.
References:
References
1. Kim YH, Jensen RA, Watanabe GL, et al: Clinical stage Ia (limited patch and plaque) mycosis fungoides. A long- term outcome analysis. Arch Dermatol 132:1309-1313, 1996.
2. Kim YH, Chow S, Varghese A, et al: Clinical characteristics and long-term outcome of patients with generalized patch and/or plaque (T2) mycosis fungoides. Arch Dermatol 135:26-32, 1999.
3. Kim YH, Hoppe RT: Mycosis fungoides and the Sézary syndrome. Semin Oncol 26:276-289, 1999.
4. Criscione VD, Weinstock MA: Incidence of cutaneous T-cell lymphoma in the united states, 1973-2002. Arch Dermatol 143:854-859, 2007.
5. Pimpinelli N, Olsen EA, Santucci M, et al: Defining early mycosis fungoides. J Am Acad Dermatol 53:1053-1063, 2005.
6. Greene FL, Page DL, Fleming ID, et al: AJCC Cancer Staging Manual, 6th ed. New York, Springer, 2002.
7. Olsen E, Vonderheid E, Pimpinelli N, et al: Revisions to the staging and classification of mycosis fungoides and Sézary syndrome: A proposal of the International Society for Cutaneous Lymphomas (ISCL) and the Cutaneous Lymphoma Task Force of the European Organization of Research and Treatment of Cancer (EORTC). Blood 110:1713-1722, 2007.
8. Heald PW, Glusac EJ: Unilesional cutaneous T-cell lymphoma: Clinical features, therapy, and follow-up of 10 patients with a treatment-responsive mycosis fungoides variant. J Am Acad Dermatol 42:283-285, 2000.
9. Chinn DM, Chow S, Kim YH, et al: Total skin electron beam therapy with or without adjuvant topical nitrogen mustard or nitrogen mustard alone as initial treatment of T2 and T3 mycosis fungoides. Int J Radiat Oncol Biol Phys 43:951-958, 1999.
10. Jones GW, Rosenthal D, Wilson LD: Total skin electron radiation for patients with erythrodermic cutaneous T-cell lymphoma (mycosis fungoides and the Sézary syndrome). Cancer 85:1985-1995, 1999.
11. Molina A, Zain J, Arber DA, et al: Durable clinical, cytogenetic, and molecular remissions after allogeneic hematopoietic cell transplantation for refractory Sézary syndrome and mycosis fungoides. J Clin Oncol 23:6163-6171, 2005.
12. Kim YH, Martinez G, Varghese A, et al: Topical nitrogen mustard in the management of mycosis fungoides: Update of the Stanford experience. Arch Dermatol 139:165-173, 2003.
13. Vonderheid EC, Tan ET, Kantor AF, et al: Long-term efficacy, curative potential, and carcinogenicity of topical mechlorethamine chemotherapy in cutaneous T cell lymphoma. J Am Acad Dermatol 20:416-428, 1989.
14. Licata AG, Wilson LD, Braverman IM, et al: Malignant melanoma and other second cutaneous malignancies in cutaneous T-cell lymphoma. The influence of additional therapy after total skin electron beam radiation. Arch Dermatol ,131:432-435 1995.
15. Zackheim HS, Epstein EH, Jr., Crain WR: Topical carmustine (BCNU) for cutaneous T cell lymphoma: A 15-year experience in 143 patients. J Am Acad Dermatol 22:802-810, 1990.
16. Zackheim HS, Kashani-Sabet M, Amin S: Topical corticosteroids for mycosis fungoides. Experience in 79 patients. Arch Dermatol 134:949-954, 1998.
17. Breneman D, Duvic M, Kuzel T, et al: Phase 1 and 2 trial of bexarotene gel for skin-directed treatment of patients with cutaneous T-cell lymphoma. Arch Dermatol 138:325-332, 2002.
18. Heald P, Mehlmauer M, Martin AG, et al: Topical bexarotene therapy for patients with refractory or persistent early-stage cutaneous T-cell lymphoma: Results of the phase III clinical trial. J Am Acad Dermatol 49:801-815, 2003.
19. Diederen PV, van Weelden H, Sanders CJ, et al: Narrowband UVB and psoralen-UVA in the treatment of early-stage mycosis fungoides: A retrospective study. J Am Acad Dermatol 48:215-219, 2003.
20. Herrmann JJ, Roenigk HH, Jr., Hurria A, et al: Treatment of mycosis fungoides with photochemotherapy (PUVA): Long-term follow-up. J Am Acad Dermatol 33:234-242, 1995.
21. Chiarion-Sileni V, Bononi A, Fornasa CV, et al: Phase II trial of interferon-alpha-2a plus psolaren with ultraviolet light A in patients with cutaneous T-cell lymphoma. Cancer 95:569-575, 2002.
22. Wilson LD, Kacinski BM, Jones GW: Local superficial radiotherapy in the management of minimal stage IA cutaneous T-cell lymphoma (mycosis fungoides). Int J Radiat Oncol Biol Phys 40:109-115, 1998.
23. Micaily B, Miyamoto C, Kantor G, et al: Radiotherapy for unilesional mycosis fungoides. Int J Radiat Oncol Biol Phys 42:361-364, 1998.
24. Wong R, Jones G, Farrar N, et al: Local superficial radiotherapy (LSRT) for newly diagnosed stage ia patch-plaque mycosis fungoides (MF) with only one, two, or three presenting lesions. Int J Radiat Oncol Biol Phys 57 Suppl 1:S289-S290, 2003.
25. Jones GW, Hoppe RT, Glatstein E: Electron beam treatment for cutaneous T-cell lymphoma. Hematol Oncol Clin North Am 9:1057-1076, 1995.
26. Quiros PA, Kacinski BM, Wilson LD: Extent of skin involvement as a prognostic indicator of disease free and overall survival of patients with T3 cutaneous T-cell lymphoma treated with total skin electron beam radiation therapy. Cancer 77:1912-1917, 1996.
27. Wilson LD, Quiros PA, Kolenik SA, et al: Additional courses of total skin electron beam therapy in the treatment of patients with recurrent cutaneous T-cell lymphoma. J Am Acad Dermatol 35:69-73, 1996.
28. Wilson LD, Jones GW, Kim D, et al: Experience with total skin electron beam therapy in combination with extracorporeal photopheresis in the management of patients with erythrodermic (T4) mycosis fungoides. J Am Acad Dermatol 43:54-60, 2000.
29. Rook AH, Kuzel TM, Olsen EA: Cytokine therapy of cutaneous T-cell lymphoma: Interferons, interleukin-12, and interleukin-2. Hematol Oncol Clin North Am 17:1435-1448, 2003.
30. Ross C, Tingsgaard P, Jorgensen H, et al: Interferon treatment of cutaneous T-cell lymphoma. Eur J Haematol 51:63-72, 1993.
31. Jumbou O, N’Guyen JM, Tessier MH, et al: Long-term follow-up in 51 patients with mycosis fungoides and Sézary syndrome treated by interferon-alfa. Br J Dermatol 1999;140:427-431.
32. Wollina U, Looks A, Meyer J, et al: Treatment of stage II cutaneous T-cell lymphoma with interferon alfa-2a and extracorporeal photochemotherapy: A prospective controlled trial. J Am Acad Dermatol 44:253-260, 2001.
33. Stadler R, Otte HG, Luger T, et al: Prospective randomized multicenter clinical trial on the use of interferon -2a plus acitretin versus interferon-2a plus PUVA in patients with cutaneous T-cell lymphoma stages I and II. Blood 92:3578-3581, 1998.
34. Thomsen K, Hammar H, Molin L, et al: Retinoids plus PUVA (RePUVA) and PUVA in mycosis fungoides, plaque stage. A report from the Scandinavian Mycosis Fungoides Group. Acta Derm Venereol 69:536-538, 1989.
35. Jones G, McLean J, Rosenthal D, et al: Combined treatment with oral etretinate and electron beam therapy in patients with cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome). J Am Acad Dermatol 26:960-967, 1992.
36. Duvic M: Systemic monotherapy vs combination therapy for CTCL: Rationale and future strategies. Oncology (Williston Park) 21:33-40, 2007.
37. Duvic M, Martin AG, Kim Y, et al: Phase 2 and 3 clinical trial of oral bexarotene (targretin capsules) for the treatment of refractory or persistent early-stage cutaneous T- cell lymphoma. Arch Dermatol 137:581-593, 2001.
38. Talpur R, Ward S, Apisarnthanarax N, et al: Optimizing bexarotene therapy for cutaneous T-cell lymphoma. J Am Acad Dermatol 47:672-684, 2002.
39. Olsen E, Duvic M, Frankel A, et al: Pivotal phase III trial of two dose levels of denileukin diftitox for the treatment of cutaneous T-cell lymphoma. J Clin Oncol 19:376-388, 2001.
40. Negro-Vilar A: Efficacy and safety of denileutoxin difititox (DD) in a phase III, double -blind, placebo controlled study of CD25+ patients with cutaneous T-cell lymphoma (CTCL). J Clin Oncol 25:447s, 2007.
41. Foss F, Demierre MF, DiVenuti G: A phase-1 trial of bexarotene and denileukin diftitox in patients with relapsed or refractory cutaneous T-cell lymphoma. Blood 106:454-457, 2005.
42. Foss FM, Bacha P, Osann KE, et al: Biological correlates of acute hypersensitivity events with DAB(389)IL-2 (denileukin diftitox, Ontak) in cutaneous T-cell lymphoma: Decreased frequency and severity with steroid premedication. Clin Lymphoma 1:298-302, 2001.
43. Duvic M, Talpur R, Ni X, et al: Phase 2 trial of oral vorinostat (suberoylanilide hydroxamic acid, SAHA) for refractory cutaneous T-cell lymphoma (CTCL). Blood 109:31-39, 2007.
44. Olsen EA, Kim YH, Kuzel TM, et al: Phase IIb multicenter trial of vorinostat in patients with persistent, progressive, or treatment refractory cutaneous T-cell lymphoma. J Clin Oncol 25:3109-3115, 2007.
45. Piekarz RL, Frye R, Turner M, et al: Phase II multi-institutional trial of the histone deacetylase inhibitor romedepsin as monotherapy for patients with cutaneous T-cell lymphoma. J Clin Oncol 27:5410-5417, 2009.
46. Lim HW, Edelson RL: Photopheresis for the treatment of cutaneous T-cell lymphoma. Hematol Oncol Clin North Am 9:1117-1126, 1995.
47. Heald P, Rook A, Perez M, et al: Treatment of erythrodermic cutaneous T-cell lymphoma with extracorporeal photochemotherapy. J Am Acad Dermatol 27:427-433, 1992.
48. Akpek G, Koh HK, Bogen S, et al: Chemotherapy with etoposide, vincristine, doxorubicin, bolus cyclophosphamide, and oral prednisone in patients with refractory cutaneous T-cell lymphoma. Cancer 86:1368-1376, 1999.
49. Kaye FJ, Bunn PA, Jr., Steinberg SM, et al: A randomized trial comparing combination electron-beam radiation and chemotherapy with topical therapy in the initial treatment of mycosis fungoides. N Engl J Med 321:1784-1790, 1989.
50. Wollina U, Dummer R, Brockmeyer NH, et al: Multicenter study of pegylated liposomal doxorubicin in patients with cutaneous T-cell lymphoma. Cancer 98:993-1001, 2003.
51. Marchi E, Alinari L, Tani M, et al: Gemcitabine as frontline treatment for cutaneous T-cell lymphoma: Phase II study of 32 patients. Cancer 104:2437-2441 2005.
52. Tsimberidou AM, Giles F, Duvic M, et al: Phase II study of pentostatin in advanced T-cell lymphoid malignancies: Update of an M.D. Anderson Cancer Center series. Cancer 100:342-349, 2004.
53. Russell-Jones R, Child F, Olavarria E, et al: Autologous peripheral blood stem cell transplantation in tumor-stage mycosis fungoides: Predictors of disease-free survival. Ann N Y Acad Sci 941:147-154, 2001.
54. Wang ES, O’Connor O, She Y, et al: Activity of a novel anti-folate (PDX, 10-propargyl 10-deazaaminopterin) against human lymphoma is superior to methotrexate and correlates with tumor RFC-1 gene expression. Leuk Lymphoma 44:1027-1035, 2003.
55. Horwitz S, Duvic M, Kim Y, et al: Low-dose pralatrexate (PDX) is active in cutaneous T-cell lymphomas: Preliminary results of a multi-center dose finding trial (abstract 261). Ann Oncol 19(suppl 4):iv162, 2008.
56. Querfeld C, Rosen ST, Guitart J, et al: Phase II trial of subcutaneous injections of human recombinant interleukin-2 for the treatment of mycosis fungoides and Sézary syndrome. J Am Acad Dermatol 56:580-583, 2007.
57. Querfeld C, Kuzel TM, Guitart J, et al: Preliminary results of phase II study of CC-5013 (lenalidomide, Revlimid) in patients with cutaneous T-cell lymphoma (abstract 3351). Blood 106:936A-937A, 2005.
58. Zinzani PL, Musuraca G, Tani M, et al: Phase II trial of proteasome inhibitor bortezomib in patients with relapsed or refractory cutaneous T-cell lymphoma. J Clin Oncol 25:4293-4297, 2007.
59. Duvic M, Foss F, Olsen E, et al: Response to oral forodesine in refractory cutaneous T-cell lymphoma: Interim results of a phase I/II study (abstract 122). Blood 110: 2007.
60. Ruuls SR, Lammerts van Bueren JJ, van de Winkel JG, et al: Novel human antibody therapeutics: The age of the Umabs. Biotechnol J 3:1157-1171, 2008.
61. Bernengo MG, Quaglino P, Comessatti A, et al: Low-dose intermittent alemtuzumab in the treatment of Sézary syndrome: Clinical and immunologic findings in 14 patients. Haematologica 92:784-794, 2007.
62. Kim YH, Duvic M, Obitz E, et al: Clinical efficacy of zanolimumab (HuMax-CD4): Two phase 2 studies in refractory cutaneous T-cell lymphoma. Blood 109:4655-4662, 2007.
63. Smith BD, Wilson LD: Management of mycosis fungoides. Part 1. Diagnosis, staging, and prognosis. Oncology (Williston Park) 17:1281-1288, 2003.
64. Smith BD, Wilson LD: Management of mycosis fungoides: Part 2. Treatment. Oncology (Williston Park) 17:1419-1428, 2003.