Treatment of Acute Promyelocytic Leukemia Without Cytotoxic Chemotherapy
There has been dramatic progress in the management of acute promyelocytic leukemia during the past three decades. Important insights into the pathogenesis of the disease have come to light and effective treatment has been developed.
The introduction of all-trans retinoic acid, or ATRA, in 1985, combined with anthracycline-based chemotherapy, has transformed acute promyelocytic leukemia (APL) from a fatal disease to one that is now highly curable. With appropriate contemporary therapy, more than 90% of patients achieve complete remission, and cure rates of approximately 80% and higher response and survival rates can be expected for patients at low and intermediate risk. The introduction of arsenic trioxide, or ATO, in 1994 has provided the opportunity to minimize and even eliminate standard cytotoxic chemotherapy from initial treatment regimens without compromising the excellent outcomes achieved by anthracycline-containing protocols. APL is a unique subtype of acute myeloid leukemia that is curable with targeted therapies and potentially without exposure to conventional DNA-damaging chemotherapy. The omission of conventional cytotoxic chemotherapy may reduce long-term complications such as cardiomyopathy and therapy-related myelodysplastic syndromes. Cure rates of APL may be further increased by adopting management strategies to reduce early hemorrhagic deaths, which now appear to be the major cause of treatment failure.
Acute promyelocytic leukemia (APL) is a rare distinct subtype of acute myeloid leukemia (AML) which accounts for 10%–15% of the approximately 12,330 adults diagnosed with AML in the United States each year.[1] There has been dramatic progress in the management of APL during the past three decades. Important insights into the pathogenesis of the disease have come to light and effective treatment has been developed. APL has become highly curable and serves as a paradigm for development of effective therapies directed at a specific molecular abnormality. Modern treatment represents a departure from standard chemotherapeutic strategies applied to all other subtypes of AML, since all-trans retinoic acid (ATRA) induces differentiation of the malignant promyelocytes into mature granulocytes. The disease is characterized by distinctive morphology of blast cells,[2] an associated life-threatening and often catastrophic bleeding disorder,[3] and a specific balanced reciprocal translocation, t(15;17),[4] which results in fusion of the PML (promyelocyte) gene on chromosome 15 to the RARα (retinoic acid receptor-α) gene on chromosome 17.[5]
Prior to the introduction of ATRA as a treatment, APL was among the most fatal of AML subtypes at presentation or during induction, primarily because of an associated complex and often catastrophic bleeding disorder which is characterized by disseminated intravascular coagulation, fibrinolysis, and proteolysis.[1] However, ATRA and arsenic trioxide (ATO) have changed the natural history of this disease from one that was characterized by a high mortality rate to one that is now associated with the highest curability rate among the AML subtypes in adults. Furthermore, multiple studies have demonstrated that combination of the two targeted agents ATRA and ATO is a highly effective and potentially curative treatment. Such a strategy has become the most exciting new treatment option for APL, potentially completely eliminating exposure to conventional cytotoxic chemotherapy in many, if not most, patients. Now that the relapse rate even among high-risk patients (white blood cell counts [WBCs] > 10 × 109/L at presentation) has decreased substantially, early hemorrhagic death prior to and during induction therapy remains the major cause of treatment failure. Almost every patient with APL achieves complete remission (CR) with either ATRA plus anthracycline-based induction or ATRA + ATO–based induction, and the relapse rate once a patient is in CR is very low[6]; therefore, reducing early death has become the most important goal in the treatment of patients with APL and is likely the best strategy to apply towards the goal of curing virtually all patients with the disease. Given the unique natural history of APL, with some patients presenting with rapidly fatal bleeding before intervention is possible, clearly it will be impossible to cure every patient. However, the cure rate among patients with APL may be increased further through education of a wide variety of healthcare professionals about early recognition of the disease and introduction of ATRA and aggressive blood product support.
Evolution of Induction Therapy in APL
Prior to ATRA, induction therapy in APL was similar to that for all other subtypes of AML. Induction consisted of an anthracycline and cytarabine (also known as ara-C), which resulted in CR rates of 65%–80% among patients newly diagnosed with APL.[7-9] Historically, however, even among patients who achieved CR with initial chemotherapy, 50%–65% subsequently relapsed and only 30%–50% remained alive at 2 years-a rate somewhat higher than that often reported for patients with other subtypes of AML.[7,10] The recognition that ATRA could induce differentiation of leukemic promyelocytes into mature granulocytes had encouraged the evaluation of ATRA either as a single agent or in combination with chemotherapy, initially in relapsed and refractory disease and subsequently in patients with newly diagnosed APL. Although treatment with ATRA alone in induction results in CR rates of 72%–90%,[11-14] patients generally relapse if additional chemotherapy is not administered. Therefore, subsequent trials combined ATRA with chemotherapy. The European APL (EuroAPL) group demonstrated in a randomized study that concurrent ATRA plus chemotherapy (daunorubicin [Cerubidine] and cytarabine) resulted in a better outcome than sequential ATRA followed by chemotherapy, primarily by reducing the relapse rate at 2 years (6% vs 16%). This improvement was subsequently confirmed in other large multicenter trials.[15-18] Therefore, the concurrent administration of ATRA and anthracycline-based chemotherapy (an anthracycline plus cytarabine or an anthracycline alone, generally idarubicin [Idamycin]) is currently considered the standard of care for induction in newly diagnosed patients with APL. It is not known which anthracycline is best to administer, however. Although a retrospective analysis published only in a preliminary form has suggested that idarubicin is associated with an improved outcome compared with daunorubicin or amsacrine,[19] there is no clear evidence that one anthracycline is clearly superior to another in APL.
Omitting Cytarabine in APL Induction
Two randomized trials have been conducted to investigate the role of cytarabine; the results have been conflicting. The National Cancer Research Institute (NCRI) in the United Kingdom (formerly the Medical Research Council, or MRC) reported no differences in response, relapse, or overall survival rates in patients randomized between ATRA plus idarubicin (AIDA) and ATRA with daunorubicin and cytarabine, but there was less myelosuppression with the regimen omitting cytarabine.[20] In contrast, a randomized study by the EuroAPL group (APL 2000) showed a statistically significant increase in relapse risk (13.4% vs 29%) and decrease in overall survival (92.9% vs 83.3%) among patients in whom cytarabine was omitted from both induction and consolidation therapy.[21,22] The therapies likely were not comparable in other ways since, for example, the doses of the anthracyclines may not have been equivalent.
The cooperative groups Gruppo Italiano Malattie Ematologiche dell’Adulto (GIMEMA) and Programa Espaol de Tratamientos en Hematologa (PETHEMA) have omitted cytarabine from induction, and demonstrated that AIDA is as effective in inducing remission as a cytarabine-containing regimen, with CR rates of 89%–95% regardless of the presenting WBC.[18,23] This discrepancy may be explained by differences in the consolidation regimens (ATRA vs no ATRA in EuroAPL trial), the number of consolidation courses, specific choice of anthracycline (idarubicin in the GIMEMA, PETHEMA, and NCRI trials vs daunorubicin in the EuroAPL trial), and the cumulative doses of anthracyclines. Treatment approaches that include cytarabine have the potential benefit of reducing the likelihood of extramedullary relapse, particularly in sanctuary sites such as the central nervous system (CNS), since relatively high doses of cytarabine are well-recognized to cross the blood–brain barrier and enter the CNS. Despite some reports of an increased incidence of extramedullary relapse since ATRA was introduced into routine clinical practice, the reported incidence of CNS relapses in APL in some studies is low, ranging from 0.6% to 2%,[24-26] and these relapses are invariably associated with marrow relapse.
Emerging Strategies Combining ATRA and ATO and Minimizing or Eliminating Conventional Cytotoxic Chemotherapy
Minimizing treatment-associated toxicities observed with conventional cytotoxic chemotherapy may further improve the outcome and long-term complications of APL. Therapy-related myelodysplastic syndrome and AML in patients with APL have been reported, albeit at a low incidence; it is possible that omission of potentially leukemogenic cytotoxic chemotherapy, particularly anthracyclines, may reduce the incidence of this complication.[27-29]
Arsenic trioxide is extremely effective in patients with relapsed APL.[30,31] Several trials have investigated the role of ATO in previously untreated patients. As a single agent, ATO induces durable CR in 85%–86% of patients with untreated APL, comparable to that achieved by ATRA-based therapy.[32,33] The outcome for patients with a presenting WBC > 5 × 109/L at diagnosis, however, appears to be inferior to results for a similar patient subset treated with ATRA plus chemotherapy, with an event-free survival (EFS) of only 67% and a higher early death rate of 14.4%, mostly from hemorrhagic complications.[32] Therefore, for many patients therapy with single-agent ATO does not appear sufficient as front-line therapy. Nevertheless, its ability to induce molecular remissions as a single agent in most patients and durable remissions in many makes it the most effective single agent in the armamentarium for APL.
The combination of ATRA and ATO is synergistic in inducing differentiation and apoptosis in vitro and in accelerating tumor regression in vivo,[34,35] providing the rationale for targeted therapy of APL without exposure to cytotoxic chemotherapy. Investigators at the Shanghai Institute of Hematology reported a randomized clinical trial in which patients were to receive ATRA, ATO, or the combination of ATRA plus ATO as induction therapy.[36] All patients in CR after induction subsequently received intensive consolidation and maintenance chemotherapy. The combination treatment of ATRA and ATO resulted in the lowest rate of relapse, a faster CR rate, and a greater reduction in the number of PML/RARα transcripts, suggesting a deeper remission, with no apparent greater toxicity than each agent alone. More recently, the Australasian Leukaemia and Lymphoma Group (ALLG) reported the outcome of 124 patients with newly diagnosed APL treated with ATRA, ATO, and idarubicin for induction; two courses of consolidation with ATRA and ATO; and 2 years of maintenance with ATRA, methotrexate, and 6-mercaptopurine. With a median follow-up of 20 months, the 3-year overall survival (OS) and EFS rates were 93% and 87%, respectively.[37] This strategy showed that most patients with newly diagnosed APL can be effectively treated and likely cured with significant reduction in exposure to standard chemotherapy. Investigators at The University of Texas M.D. Anderson Cancer Center similarly demonstrated that the combination of ATRA and ATO is effective in untreated APL.[38] A high CR rate of 96% was achieved with few late relapses even when chemotherapy was eliminated during consolidation, in patients with a presenting WBC < 10 × 109/L.[37,38] Patients with presenting WBC ≥ 10 × 109/L achieved an inferior CR rate of 79%–81%, however, owing to early treatment failure from fatal hemorrhage and APL differentiation syndrome despite the addition of either gemtuzumab ozogamicin (GO) or idarubicin during induction to control the WBC.[37,38]
These studies suggest that the combination of ATRA and ATO can effectively treat and potentially cure patients with standard-risk disease (defined as those with presenting WBC < 10 × 109/L), and it has emerged as the most exciting new treatment strategy in APL. Such a strategy is an excellent alternative for patients who are considered unfit for conventional treatment and those with severe comorbidities (eg, older adults, patients with cardiac dysfunction or other severe organ dysfunction).
In patients presenting with high WBC, however, concomitant use of cytotoxic drugs such as anthracyclines in induction and high- or intermediate-dose cytarabine in either induction or consolidation appears to be critical to prevent rapid development of leukocytosis and consequent APL differentiation syndrome and relapse. Several randomized trials are being conducted to address the question of whether the outcome among patients in whom chemotherapy is completely eliminated or minimized is as favorable as conventional ATRA plus anthracycline-based approaches.
Reducing Early Death in APL
FIGURE 1
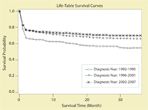
Survival of Patients With APL Over Time From 1992 to 2007FIGURE 2

Survival of Patients With APL by Age
Hemorrhage, often catastrophic in presentation and outcome, is the major cause of death during induction therapy in APL, despite the introduction of ATRA into routine clinical practice 15 years ago.[39] Delayed diagnosis and treatment, together with an elevated WBC, were significantly associated with severe bleeding and early death.[40] Multicenter cooperative group studies report a rate of approximately 5%–10% early deaths within 1 month after initiating treatment,[11,16-18,41] but several population-based observational studies report higher early death rates in unselected APL patients, with the largest study reporting a 17.3% overall early death rate despite ATRA and significantly higher in older adults at 24.2% (see Figures 1 and 2).[42-44] These findings indicate that the availability of ATRA has not eliminated the major cause of treatment failure in APL. They also emphasize the importance of immediate disease recognition and rapid intervention, since delayed treatment is associated with significant morbidity and mortality.
To implement immediate institution of the appropriate therapy in APL, however, it is critical that healthcare providers from a variety of disciplines, who may be the first to evaluate APL patients, understand that APL is a medical emergency requiring urgent initiation of ATRA at the first suspicion of the disease and before genetic confirmation, with concomitant aggressive supportive care to address the bleeding disorder. Because ATRA has been shown to rapidly reduce the severity of bleeding and decrease the amount of blood product consumption,[45] it should be started immediately at the earliest hint of the diagnosis. Although the true impact of ATRA in reducing the early death rate is not known,[11,46] it is reasonable to presume it will be beneficial given the paucity of toxicities, particularly following one to two doses of treatment. The role of ATO in reducing early death rates is less clear, and more studies are required to better define the role of ATO with or without ATRA in reducing hemorrhagic complications during induction therapy.[38,47]
Aggressive supportive measures to counteract coagulopathy need to be instituted immediately. These include fresh frozen plasma, fibrinogen, cryoprecipitate, and multiple platelet transfusions to maintain the fibrinogen concentration and platelet count above 100–150 mg/dL and 50,000/µL, respectively, and post-transfusion values should be obtained to ensure adequate repletion of coagulation factors, platelets, and fibrinogen.[48] Such replacement therapy should continue during induction therapy until disappearance of all clinical and laboratory signs of coagulopathy.
Since optimal treatment of APL is dependent on rapid access to unique specialized diagnostic tests and hospital facilities with immediate availability of ATRA and the appropriate blood products, patients with APL should be treated at specialized centers with an experienced multidisciplinary team.[48] Breccia et al reported a significantly increased early death rate in patients who initially presented to nonspecialized primary care institutions with little experience in the treatment of acute leukemia.[40] Therefore, the immediate and efficient access to experienced medical centers, to reduce the time span between the first signs and symptoms of APL and initiation of specific therapy, is critical to improve the clinical outcome.
While genetic confirmation of the specific lesion PML/RARα fusion is required to establish the definitive diagnosis of APL, test results from fluorescence in situ hybridization (FISH) and reverse transcriptase polymerase chain reaction (RT-PCR) may not be immediately available at community hospitals and require a turnaround time of 1–2 days. Recently, immunostaining with anti-PML monoclonal antibodies on dry smears of bone marrow or even peripheral blood samples has been shown to establish a rapid diagnosis in less than 4 hours, with high sensitivity and specificity.[49] This test may be particularly valuable in smaller centers lacking immediate access to a sophisticated molecular diagnostics laboratory. Such testing should not delay the initiation of ATRA if APL is suspected, however, and patients should immediately be transferred to more experienced centers with ATRA therapy, which should be started prior to patient transfer.
Consolidation Therapy
There is no consensus on the optimal type and intensity of consolidation therapy. The benefit of ATRA when combined with chemotherapy for consolidation has not been demonstrated in randomized studies. However, historical comparisons of consecutive trials by the investigators of GIMEMA[6] and PETHEMA[50] demonstrated a statistically significant reduction in the relapse rate (8.7% vs 20.1%) and a higher disease-free survival (DFS) and OS when ATRA was given with chemotherapy in consolidation.
FIGURE 3

Disease-Free Survival for High-Risk Patients With APL, According to Protocol, From the AIDA 0493 and AIDA 2000 Studies
There is no clear consensus as to which specific chemotherapy regimen is best in consolidation. Major cooperative groups have used several consolidation approaches, and their investigations suggest a benefit of cytarabine in patients with the presenting WBC > 10 × 109/L. A study by the PETHEMA group (LPA2005), in which patients with a WBC > 10 × 109/L received cytarabine combined with ATRA and idarubicin in the first and third consolidation courses, reported a favorable impact on the 3-year relapse rate (11% vs 26%, P = .03) when cytarabine was added to the consolidation.[51]
The GIMEMA group (AIDA2000) also reported a reduction in the relapse rate (49.7% vs 9.3%) when cytarabine was added to the consolidation in patients with a WBC > 10 × 109/L.[6] In the GIMEMA study, however, ATRA was also added to the consolidation therapy, which may have contributed to the improved outcome observed. In contrast, a study by the NCRI, published only in abstract form, showed no benefit of cytarabine, irrespective of the initial WBC.[20]
The majority of studies suggest a benefit of cytarabine in patients with a WBC > 10 × 109/L, possibly because of the synergistic effect of the combination of ATRA plus cytarabine.[52] The GIMEMA group recently reported the outcome of high-risk patients treated with either the AIDA 0493 protocol, which did not include intermediate-dose ara-C, or AIDA 2000, with risk-adapted post remission therapy in which such patients received intermediate-dose ara-C. The DFS among high-risk patients was 49.6% for patients treated on AIDA 0493 and 84.5% for those treated on AIDA 2000[6] (see Figure 3). It appears that cytarabine can be omitted for patients with WBC ≤ 10 × 109/L, however, and excellent outcome is preserved.
Emerging Nonchemotherapy Consolidation Regimens
To reduce toxicity and chemotherapy exposure in non–high-risk patients, several cooperative groups have investigated the role of ATRA and ATO either in place of or combined with reduced doses of cytotoxic chemotherapy in consolidation.
The North American Intergroup investigated the role of ATO combined with ATRA in consolidation in a randomized trial in which patients received 2 cycles of consolidation of ATRA plus daunorubicin, either immediately following induction therapy or preceded by two 25-day cycles of ATO plus ATRA.[53] Results from the trial demonstrated that addition of ATO as initial consolidation therapy significantly improves 3-year DFS (90% vs 70%) and OS (86% vs 81%) regardless of presenting WBC. In a phase II study, Gore et al reported a comparable outcome (DFS 90% and OS 88% at a median follow-up of 2.7 years) with a considerably reduced amount of anthracyclines combined with a single cycle (30 days) of ATO.[54] Investigators at M.D. Anderson Cancer Center completely eliminated cytotoxic chemotherapy from consolidation in all patients and reported a 3-year survival of 85% using ATRA and ATO for 28 weeks as the only post-remission therapy.[55] The ALLG administered two courses of consolidation with ATRA and ATO, and reported a 3-year OS and EFS rates of 93% and 87%, respectively.[56] The Indian APML Study Group, IAPLSG, reported a clinical outcome comparable to that achieved with conventional combination chemotherapy in patients with WBC ≤ 10 × 109/L, using single-agent treatment with ATO during induction and consolidation.[57] The majority of studies suggest a potential synergism between an ATRA and ATO combination and raise the possibility of minimizing or even eliminating chemotherapy in APL management, particularly in patients with WBC ≤ 10 × 109/L. Ongoing randomized trials by several cooperative groups comparing ATRA plus ATO to ATRA plus chemotherapy may establish a new standard of care in APL.
Maintenance Therapy
Two randomized trials demonstrated a benefit of an ATRA-based maintenance therapy. The role of maintenance therapy remains unclear, however, particularly for non–high-risk patients and for those achieving molecular remission at the end of consolidation. Maintenance therapy in non–high-risk elderly patients may even be harmful, as shown by the EuroAPL group study that reported an 11% death rate in patients with CR, mainly from sepsis attributable to myelosuppression.[41] Furthermore, introduction of ATO into first-line treatment for APL may reduce the significance of maintenance therapy.
REFERENCE GUIDE
Therapeutic Agents
Mentioned in This Article
Anthracycline
All-trans retinoic acid (ATRA)
Arsenic trioxide (ATO)
Cytarabine (ara-C)
Daunorubicin (Cerubidine)
Gemtuzumab ozogamicin
Idarubicin (Idamycin)
Tamibarotene
Brand names are listed in parentheses only if a drug is not available generically and is marketed as no more than two trademarked or registered products. More familiar alternative generic designations may also be included parenthetically.
The North American Intergroup study first reported a clinical benefit with the ATRA-based maintenance therapy for a year in terms of a reduction in relapse rate (22% vs 39%) and a higher 5-year DFS (61% vs 36%).[11,58] Similarly, the EuroAPL group confirmed the beneficial effect of adding ATRA to maintenance therapy in a randomized study of a regimen consisting of continuous low-dose 6-mercaptopurine and methotrexate in addition to intermittent ATRA.[41] The EuroAPL group recently updated the results of their previous study and reported that the clinical benefit of maintenance therapy was mainly observed in high-risk patients, while only a marginal benefit was noted in non–high-risk patients.[59] In contrast, two randomized trials by GIMEMA and the Japanese Adult Leukemia Study Group (JALSG) have reported no benefit from maintenance therapy if patients achieved molecular CR after consolidation.[60,61] However, the JALSG group trial included intensive chemotherapy and not ATRA as maintenance.
The discrepancy in these studies suggests that the benefit of maintenance treatment may depend on the specific prior induction and consolidation therapy, as well as PML/RARα status after consolidation, which has been shown to correlate with the relapse risk.[62,63] For example, both the JALSG[60] and GIMEMA[61] trials administered three cycles of consolidation therapy and used idarubicin as anthracycline for induction and consolidation chemotherapy, whereas the EuroAPL[41] and North American Intergroup[11] studies gave only two consolidation courses and used daunorubicin. Furthermore, while studies by the GIMEMA and JALSG groups were carried out in patients testing negative for PML/RARα at the end of consolidation, the North American Intergroup and EuroAPL studies did not examine molecular remission status at the end of consolidation, raising a question as to whether the observed benefit of maintenance therapy is largely attributable to responses in patients with minimal residual disease following consolidation.
These studies suggest that patients with a WBC ≤ 10 × 109/L and who achieve complete molecular remission after consolidation therapy may not benefit from the combination maintenance therapy. For high-risk patients, maintenance therapy for 1 to 2 years with intermittent ATRA and low-dose chemotherapy with 6-mercaptopurine and methotrexate are recommended. Given that there are no prospective trial data comparing 1 vs 2 years of maintenance therapy, the current recommendation is to continue maintenance therapy for 2 years unless toxicity develops.
Nonchemotherapy Maintenance Regimens
To investigate whether low-dose chemotherapy with 6-mercaptopurine and methotrexate can be safely omitted, investigators at M.D. Anderson Cancer Center used ATRA and ATO as the only postremission therapy following successful induction and reported a 3-year survival of 85%.[55] One ongoing randomized trial is exploring the role of ATRA-based nonchemotherapy maintenance. A study by the JALSG is comparing ATRA to the synthetic retinoid tamibarotene. Several trials are also being conducted exploring nonchemotherapy maintenance regimens. The IAPLSG is conducting a phase II/III study wherein patients will receive ATO as a primary induction therapy followed by either 6 or 12 months of ATO maintenance. None of these trials includes an arm without maintenance, however. Future trials should address the role of maintenance therapy, particularly when ATO is incorporated into induction and consolidation.
Future Directions in APL
Now that cure rates are so high, the treatment of APL has focused recently of exploiting the synergism between ATRA and ATO in order to minimize cytotoxic chemotherapy and prevent both short-term and long-term toxicities. Cytarabine is not needed for induction in the majority of patients. Large cumulative doses of anthracyclines may result in cardiomyopathy. Therefore, the combination of ATRA and ATO has emerged as an attractive alternative approach. An oral formulation of ATO is under development. Tamibarotene is a novel retinoid with potential advantages compared with ATRA; its advantages include lower binding to the gamma receptor, which may result in less skin toxicity.
Financial Disclosure:The authors have no significant financial interest or other relationship with the manufacturers of any products or providers of any service mentioned in this article.
References:
REFERENCES
1. Tallman MS, Altman JK. Curative strategies in acute promyelocytic leukemia. Hematology Am Soc Hematol Educ Program. 2008;391-9.
2. Bennett JM, Catovsky D, Daniel MT, et al. Proposals for the classification of the acute leukaemias. French-American-British (FAB) co-operative group. Br J Haematol. 1976;33:451-8.
3. Tallman MS, Kwaan HC. Reassessing the hemostatic disorder associated with acute promyelocytic leukemia. Blood. 1992;79:543-53.
4. Rowley JD, Golomb HM, Dougherty C. 15/17 translocation, a consistent chromosomal change in acute promyelocytic leukaemia. Lancet. 1977;1:549-50.
5. Grignani F, Ferrucci PF, Testa U, et al. The acute promyelocytic leukemia-specific PML-RAR alpha fusion protein inhibits differentiation and promotes survival of myeloid precursor cells. Cell. 1993;74:423-31.
6. Lo Coco F, Avvisati G, Vignetti M, et al. Front-line treatment of acute promyelocytic leukemia with AIDA induction followed by risk-adapted consolidation for adults younger than 61 years: results of the AIDA-2000 trial of the GIMEMA Group. Blood. 2010;116:3171-9.
7. Cunningham I, Gee TS, Reich LM, et al. Acute promyelocytic leukemia: treatment results during a decade at Memorial Hospital. Blood. 1989;73:1116-22.
8. Fenaux P, Degos L. Treatment of acute promyelocytic leukemia with all-trans retinoic acid. Leuk Res. 1991;15:655-7.
9. Head DR, Kopecky KJ, Willman C, Appelbaum FR. Treatment outcome with chemotherapy in acute promyelocytic leukemia: the Southwest Oncology Group (SWOG) experience. Leukemia. 1994;8(Suppl 2):S38-41.
10. Degos L, Dombret H, Chomienne C, et al. All-trans-retinoic acid as a differentiating agent in the treatment of acute promyelocytic leukemia. Blood. 1995;85:2643-53.
11. Tallman MS, Andersen JW, Schiffer CA, et al. All-trans-retinoic acid in acute promyelocytic leukemia. N Engl J Med. 1997;337:1021-8.
12. Chen ZX, Xue YQ, Zhang R, et al. A clinical and experimental study on all-trans retinoic acid-treated acute promyelocytic leukemia patients. Blood. 1991;78:1413-9.
13. Castaigne S, Chomienne C, Daniel MT, et al. All-trans retinoic acid as a differentiation therapy for acute promyelocytic leukemia. I. Clinical results. Blood. 1990;76:1704-9.
14. Huang ME, Ye YC, Chen SR, et al. Use of all-trans retinoic acid in the treatment of acute promyelocytic leukemia. Blood. 1988;72:567-72.
15. Lengfelder E, Reichert A, Schoch C, et al. Double induction strategy including high dose cytarabine in combination with all-trans retinoic acid: effects in patients with newly diagnosed acute promyelocytic leukemia. German AML Cooperative Group. Leukemia. 2000;14:1362-70.
16. Mandelli F, Diverio D, Avvisati G, et al. Molecular remission in PML/RAR alpha-positive acute promyelocytic leukemia by combined all-trans retinoic acid and idarubicin (AIDA) therapy. Gruppo Italiano-Malattie Ematologiche Maligne dell’Adulto and Associazione Italiana di Ematologia ed Oncologia Pediatrica Cooperative Groups. Blood. 1997;90:1014-21.
17. Asou N, Adachi K, Tamura J, et al. Analysis of prognostic factors in newly diagnosed acute promyelocytic leukemia treated with all-trans retinoic acid and chemotherapy. Japan Adult Leukemia Study Group. J Clin Oncol. 1998;16:78-85.
18. Sanz MA, Martin G, Rayon C, et al. A modified AIDA protocol with anthracycline-based consolidation results in high antileukemic efficacy and reduced toxicity in newly diagnosed PML/RARalpha-positive acute promyelocytic leukemia. PETHEMA group. Blood. 1999;94:3015-21.
19. Berman E, Little C, Kher U, et al. Prognostic analysis of patients with acute promyelocytic leukemia (abstract). Blood. 1991;78(Suppl 1):43a.
20. Burnett AK, Hills RK, Grimwade D, et al. Idarubicin and ATRA is as effective as MRC chemotherapy in patients with acute promyelocytic leukaemia with lower toxicity and resource usage: preliminary results of the MRC AML15 trial (abstract). Blood. 2007;110:589.
21. Ades L, Chevret S, Raffoux E, et al. Is cytarabine useful in the treatment of acute promyelocytic leukemia? Results of a randomized trial from the European Acute Promyelocytic Leukemia Group. J Clin Oncol. 2006;24:5703-10.
22. Ades L, Raffoux E, Chevret S, et al. Is araC required in the treatment of standard risk APL? Long term results of a randomized trial (APL 2000) from the French Belgian Swiss APL Group (abstract). Blood. 2010;116:13.
23. Avvisati G, Lo Coco F, Diverio D, et al. AIDA (all-trans retinoic acid + idarubicin) in newly diagnosed acute promyelocytic leukemia: a Gruppo Italiano Malattie Ematologiche Maligne dell’Adulto (GIMEMA) pilot study. Blood. 1996;88:1390-8.
24. Specchia G, Lo Coco F, Vignetti M, et al. Extramedullary involvement at relapse in acute promyelocytic leukemia patients treated or not with all-trans retinoic acid: a report by the Gruppo Italiano Malattie Ematologiche dell’Adulto. J Clin Oncol. 2001;19:4023-8.
25. de Botton S, Sanz MA, Chevret S, et al. Extramedullary relapse in acute promyelocytic leukemia treated with all-trans retinoic acid and chemotherapy. Leukemia. 2006;20:35-41.
26. Montesinos P, Diaz-Mediavilla J, Deben G, et al. Central nervous system involvement at first relapse in patients with acute promyelocytic leukemia treated with all-trans retinoic acid and anthracycline monochemotherapy without intrathecal prophylaxis. Haematologica. 2009;94:1242-9.
27. Andersen MK, Pedersen-Bjergaard J. Therapy-related MDS and AML in acute promyelocytic leukemia. Blood. 2002;100:1928-9.
28. Garcia-Manero G, Kantarjian HM, Kornblau S, Estey E. Therapy-related myelodysplastic syndrome or acute myelogenous leukemia in patients with acute promyelocytic leukemia (APL). Leukemia. 2002;16:1888.
29. Latagliata R, Petti MC, Fenu S, et al. Therapy-related myelodysplastic syndrome-acute myelogenous leukemia in patients treated for acute promyelocytic leukemia: an emerging problem. Blood. 2002;99:822-4.
30. Soignet SL, Maslak P, Wang ZG, et al. Complete remission after treatment of acute promyelocytic leukemia with arsenic trioxide. N Engl J Med. 1998;339:
1341-8.
31. Soignet SL, Frankel SR, Douer D, et al. United States multicenter study of arsenic trioxide in relapsed acute promyelocytic leukemia. J Clin Oncol. 2001;
19:3852-60.
32. Mathews V, George B, Lakshmi KM, et al. Single-agent arsenic trioxide in the treatment of newly diagnosed acute promyelocytic leukemia: durable remissions with minimal toxicity. Blood. 2006;107:2627-32.
33. Ghavamzadeh A, Alimoghaddam K, Ghaffari SH, et al. Treatment of acute promyelocytic leukemia with arsenic trioxide without ATRA and/or chemotherapy. Ann Oncol. 2006;17:131-4.
34. Zheng PZ, Wang KK, Zhang QY, et al. Systems analysis of transcriptome and proteome in retinoic acid/arsenic trioxide-induced cell differentiation/apoptosis of promyelocytic leukemia. Proc Natl Acad Sci U S A. 2005;102:7653-8.
35. Gianni M, Koken MH, Chelbi-Alix MK, et al. Combined arsenic and retinoic acid treatment enhances differentiation and apoptosis in arsenic-resistant NB4 cells. Blood. 1998;91:4300-10.
36. Shen ZX, Shi ZZ, Fang J, et al. All-trans retinoic acid/As2O3 combination yields a high quality remission and survival in newly diagnosed acute promyelocytic leukemia. Proc Natl Acad Sci U S A. 2004;101:5328-35.
37. Ravandi F, Estey EH, Cortes JE, et al. Phase II study of all-trans retinoic acid (ATRA), arsenic trioxide (ATO), with or without gemtuzumab ozogamicin (GO) for the frontline therapy of patients with acute promyelocytic leukemia (APL) (abstract). Blood. 2010;116:1080.
38. Estey E, Garcia-Manero G, Ferrajoli A, et al. Use of all-trans retinoic acid plus arsenic trioxide as an alternative to chemotherapy in untreated acute promyelocytic leukemia. Blood. 2006;107:3469-73.
39. de la Serna J, Montesinos P, Vellenga E, et al. Causes and prognostic factors of remission induction failure in patients with acute promyelocytic leukemia treated with all-trans retinoic acid and idarubicin. Blood. 2008;111:3395-402.
40. Breccia M, Latagliata R, Cannella L, et al. Early hemorrhagic death before starting therapy in acute promyelocytic leukemia: association with high WBC count, late diagnosis and delayed treatment initiation. Haematologica. 2010;95:853-4.
41. Fenaux P, Chastang C, Chevret S, et al. A randomized comparison of all transretinoic acid (ATRA) followed by chemotherapy and ATRA plus chemotherapy and the role of maintenance therapy in newly diagnosed acute promyelocytic leukemia. The European APL Group. Blood. 1999;94:1192-200.
42. Alizadeh AA, McClellan JS, Gotlib JR, et al. Early mortality in acute promyelocytic leukemia may be higher than previously reported (abstract). Blood. 2009;114:1015.
43. Park JH, Qiao B, Panageas KS, et al. Early death rate in acute promyelocytic leukemia remains high despite all-trans retinoic acid. Blood. 2011; Jun 8 [Epub ahead of print]
44. Micol JB, Raffoux E, Boissel N, et al. Do early events excluding patients with acute promyelocytic leukemia (APL) from trial enrollment modify treatment result evaluation? Real-life management of 100 patients referred to the university Hospital Saint-Louis Between 2000 and 2010 (abstract). Blood. 2010;116:1083.
45. Di Bona E, Avvisati G, Castaman G, et al. Early haemorrhagic morbidity and mortality during remission induction with or without all-trans retinoic acid in acute promyelocytic leukaemia. Br J Haematol. 2000;108:689-95.
46. Visani G, Gugliotta L, Tosi P, et al. All-trans retinoic acid significantly reduces the incidence of early hemorrhagic death during induction therapy of acute promyelocytic leukemia. Eur J Haematol. 2000;64:139-44.
47. Hu J, Liu YF, Wu CF, et al. Long-term efficacy and safety of all-trans retinoic acid/arsenic trioxide-based therapy in newly diagnosed acute promyelocytic leukemia. Proc Natl Acad Sci U S A. 2009;106:3342-7.
48. Sanz MA, Grimwade D, Tallman MS, et al. Management of acute promyelocytic leukemia: recommendations from an expert panel on behalf of the European LeukemiaNet. Blood. 2009;113:1875-91.
49. Dimov ND, Medeiros LJ, Kantarjian HM, et al. Rapid and reliable confirmation of acute promyelocytic leukemia by immunofluorescence staining with an antipromyelocytic leukemia antibody. Cancer. 2010;116:369-76.
50. Sanz MA, Martin G, Gonzalez M, et al. Risk-adapted treatment of acute promyelocytic leukemia with all-trans-retinoic acid and anthracycline monochemotherapy: a multicenter study by the PETHEMA group. Blood. 2004;103:1237-43.
51. Sanz MA, Montesinos P, Rayon C, et al. Risk-adapted treatment of acute promyelocytic leukemia based on all-trans retinoic acid and anthracycline with addition of cytarabine in consolidation therapy for high-risk patients: further improvements in treatment outcome. Blood. 2010;115:5137-46.
52. Flanagan SA, Meckling KA. All- trans-retinoic acid increases cytotoxicity of 1-beta-D-arabinofuranosylcytosine in NB4 cells. Cancer Chemother Pharmacol. 2003;51:363-75.
53. Powell BL, Moser B, Stock W, et al. Arsenic trioxide improves event-free and overall survival for adults with acute promyelocytic leukemia: North American Leukemia Intergroup Study C9710. Blood. 2010;116:3751-7.
54. Gore SD, Gojo I, Sekeres MA, et al. Single cycle of arsenic trioxide-based consolidation chemotherapy spares anthracycline exposure in the primary management of acute promyelocytic leukemia. J Clin Oncol. 2010;28:1047-53.
55. Ravandi F, Estey E, Jones D, et al. Effective treatment of acute promyelocytic leukemia with all-trans-retinoic acid, arsenic trioxide, and gemtuzumab ozogamicin. J Clin Oncol. 2009;27:504-10.
56. Iland H, Firkin F, Supple S, et al. Interim analysis of the APML4 trial incorporating all-trans retinoic acid (ATRA), idarubicin, and intravenous arsenic trioxide (ATO) as initial therapy in acute promyelocytic leukaemia (APL): an Australasian Leukaemia and Lymphoma Group (ALLG) study (abstract). International Oral Arsenic Union 38th Annual Scientific Meeting of the Hong Kong Society of Haematology. 2010;16:6.
57. Mathews V, George B, Chendamarai E, et al. Single-agent arsenic trioxide in the treatment of newly diagnosed acute promyelocytic leukemia: long-term follow-up data. J Clin Oncol. 2010;28:3866-71.
58. Tallman MS, Andersen JW, Schiffer CA, et al. All-trans retinoic acid in acute promyelocytic leukemia: long-term outcome and prognostic factor analysis from the North American Intergroup protocol. Blood. 2002;100:4298-302.
59. Ades L, Guerci A, Raffoux E, et al. Very long-term outcome of acute promyelocytic leukemia after treatment with all-trans retinoic acid and chemotherapy: the European APL Group experience. Blood. 2010;115:1690-6.
60. Asou N, Kishimoto Y, Kiyoi H, et al. A randomized study with or without intensified maintenance chemotherapy in patients with acute promyelocytic leukemia who have become negative for PML-RARalpha transcript after consolidation therapy: the Japan Adult Leukemia Study Group (JALSG) APL97 study. Blood. 2007;110:59-66.
61. Avvisati G, Lo-Coco F, Paoloni FP, et al. Aida 0493 protocol for newly diagnosed acute promyelocytic leukemia: Very long-term results and role of maintenance. Blood. 2011;117:4716-25.
62. Lo Coco F, Diverio D, Falini B, et al. Genetic diagnosis and molecular monitoring in the management of acute promyelocytic leukemia. Blood. 1999;94:12-22.
63. Grimwade D, Lo Coco F. Acute promyelocytic leukemia: a model for the role of molecular diagnosis and residual disease monitoring in directing treatment approach in acute myeloid leukemia. Leukemia. 2002;16:1959-73.