Management of Sexual Dysfunction After Prostate Brachytherapy
Erectile dysfunction is a common sequela following potentiallycurative local treatment for early-stage carcinoma of the prostategland. With larger studies and longer follow-up, it is clear that erectiledysfunction following prostate brachytherapy is more common thanpreviously reported, with a myriad of previously unrecognized sexualsymptoms. Approximately 50% of patients develop erectile dysfunctionwithin 5 years of implantation. Several factors including preimplantpotency, patient age, the use of supplemental external-beam irradiation,radiation dose to the prostate gland, radiation dose to the bulb ofthe penis, and diabetes mellitus appear to exacerbate brachytherapyrelatederectile dysfunction. The majority of patients with brachytherapy-induced erectile dysfunction respond favorably to sildenafil citrate(Viagra). Despite reports questioning the potency-sparing advantageassociated with brachytherapy, recent elucidations of brachytherapyrelatederectile dysfunction may result in refinement of treatmenttechniques, an increased likelihood of potency preservation, andultimately, improved quality of life.
ABSTRACT: Erectile dysfunction is a common sequela following potentially curative local treatment for early-stage carcinoma of the prostate gland. With larger studies and longer follow-up, it is clear that erectile dysfunction following prostate brachytherapy is more common than previously reported, with a myriad of previously unrecognized sexual symptoms. Approximately 50% of patients develop erectile dysfunction within 5 years of implantation. Several factors including preimplant potency, patient age, the use of supplemental external-beam irradiation, radiation dose to the prostate gland, radiation dose to the bulb of the penis, and diabetes mellitus appear to exacerbate brachytherapyrelated erectile dysfunction. The majority of patients with brachytherapy- induced erectile dysfunction respond favorably to sildenafil citrate (Viagra). Despite reports questioning the potency-sparing advantage associated with brachytherapy, recent elucidations of brachytherapyrelated erectile dysfunction may result in refinement of treatment techniques, an increased likelihood of potency preservation, and ultimately, improved quality of life.
Over the past decade, prostate brachytherapy has been used increasingly as definitive treatment for early-stage carcinoma of the prostate gland, with the majority of the literature on brachytherapy reporting biochemical results as favorable as those in the most positive radical prostatectomy and externalbeam radiation therapy series.[1-4] Because of a lack of definitive evidence supporting the efficacy of one local treatment approach over another, quality-of-life (QOL) parameters have assumed greater importance. It has been widely asserted that preservation of potency is more likely following brachytherapy, but longer follow-up has raised substantial doubts about brachytherapy's potency- sparing advantage.[5,6] In addition, brachytherapy results in a myriad of previously unrecognized effects on sexual function.[7,8]
Erectile dysfunction has been estimated to affect up to 30 million American men and is a common sequela of potentially curative local treatment for early-stage carcinoma of the prostate gland.[9] The National Institutes of Health (NIH) Consensus Conference defined erectile dysfunction as "the inability to attain and/or maintain penile erection sufficient for satisfactory sexual performance" and recommended the development of reliable methods for assessing the symptoms of erectile dysfunction and evaluating treatment outcomes.[9] Erectile dysfunction has a deleterious effect on quality of life, including diminished physical and emotional well-being, marital discord, and loss of self-esteem.[9-12]
Following brachytherapy, the use of either palladium-103 or iodine-125 with or without supplemental external- beam irradiation resulted in erectile dysfunction in 6% to 61% of cases.[5-7,13-22] With additional follow- up, continued deterioration in erectile function is expected due to the normal aging process.[23] The documentation of sexual function following prostate brachytherapy may help to elucidate the etiology of erectile dysfunction, refine treatment techniques, and lead to an increase in the likelihood of potency preservation.
QOL Instruments
Investigators are increasingly evaluating postimplant QOL with survey instruments far more detailed than previously published scales such as those of the Radiation Therapy Oncology Group (RTOG).[24] These more detailed instruments were created in an attempt to enable more uniform reporting of lifestyle effects and facilitate comparisons between different modalities. Although admirable in their intent, such efforts culminate in scores of, for example, urinary bother, functional well-being, and expanded prostate cancer index composite (EPIC) scores that are so nebulous, they are almost meaningless to patients or investigators other than QOL aficionados.[ 25-28] To make matters worse, none of the current QOL instruments adequately capture brachytherapyrelated sexual dysfunction. In fact, a variety of sexual symptoms other than erectile dysfunction occur following prostate brachytherapy.
FIGURE 1
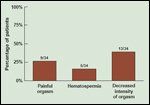
Survey of Sexual Function
Although QOL experts have been working to homogenize QOL parameters for comparison between treatment modalities, Merrick and colleagues have detailed specific sexual changes related to brachytherapy by means of an in-depth survey of sexual function that combines the specific erectile questions of the International Index of Erectile Function (IIEF) with a number of more detailed topics. Hematospermia, orgasmalgia (pain at the time of orgasm), and alteration in the intensity of orgasm were reported in the first year postimplant by 26%, 15%, and 38% of patients, respectively (Figure 1).[7]
Orgasmalgia normally occurs within the first 6 to 12 months following implantation and is probably related to inflammation of the terminal portion of the ejaculatory ducts and the urethra.[8] Hematospermia occurs as both an early and late phenomenon, is probably due to radiation- induced capillary fragility, and appears to be of no clinical significance.[8] These side effects are of limited duration in most patients.
Prostate brachytherapy does affect sexual function and, unfortunately, standard survey tools do not address these changes. In order for QOL instruments to accurately reflect sexual function following brachytherapy, new validated instruments that address brachytherapy-specific symptomatology will have to be developed.
Assessment of Sexual Potency After Prostate Brachytherapy
TABLE 1

Specific Erectile Function Questions of the International Index of Erectile Function
The wide range of erectile dysfunction after prostate brachytherapy may result from differences in follow-up and may be distorted by the method of data collection. Litwin et al reported that physician ratings of patient symptoms do not correlate well with patient self-assessment of QOL.[29] To date, only one brachytherapy potency study has exclusively used a patient-administered validated instrument.[6] The IIEF comprises 15 questions in five domains (erectile function, orgasmic function, sexual desire, intercourse satisfaction, and overall satisfaction) and has been validated as a sensitive and specific tool for the evaluation of male sexual function.[30,31] Merrick et al used the specific erectile questions of the IIEF (Table 1), with potency defined as an IIEF score ≥ 11.[6,7]
TABLE 2

Mount Sinai Four-Tiered System for Grading Erectile Function
In contrast, Stock and colleagues at Mount Sinai School of Medicine formulated a four-tiered system in which erectile function was graded on a scale of 0 to 3, with the score primarily assigned by the physician during an interview with the patient (Table 2).[5,14] The Mount Sinai scoring system is currently being used in two prospective randomized prostate brachytherapy trials.[32] The remainder of the literature on potency following brachytherapy has either used an unvalidated patient questionnaire administered via telephone interview[ 17] or has defined potency by either a subjective physician interpretation of erections sufficient for vaginal penetration[18-20,22] or without any defining criteria.[13,21]
Potency Preservation
TABLE 3

Potency Preservation After Permanent Prostate Brachytherapy
After brachytherapy, wide ranges of erectile dysfunction have been re- ported, possibly the result of differences in patient follow-up, definitions of erectile dysfunction, and mode of data collection (Table 3).[5-7,13,14,16-22] In studies of QOL outcomes, information should ideally be obtained by direct patient surveys.[29] Early investigators uniformly reported high potency rates with 1 to 3 years of follow- up.[13,16,19]
Using a validated patient-administered QOL instrument, Merrick and colleagues reported a 39% 6-year rate of potency preservation for patients undergoing brachytherapy with or without supplemental external-beam irradiation and/or hormonal manipulation; among patients who did not receive radiation, the potency preservation rate was 52%.[6] This finding is comparable to prior 5- and 6- year potency results from Mount Sinai and Memorial Sloan-Kettering for patients undergoing brachytherapy as a monotherapeutic approach.[5,16]
Recently, Potters et al reported a 76% rate of potency preservation at 5 years for patients undergoing monotherapeutic brachytherapy.[22] In addition, these investigators found that 80% of brachytherapy-induced erectile dysfunction was apparent by 24 months,[22] whereas two other studies reported median times to the development of brachytherapy-induced erectile dysfunction of 17 and 6 months, respectively.[6,16]
Timing of Impotence
Using more reliable tools and longer follow-up, two factors regarding postimplant erectile dysfunction have become apparent. First, there is a significant incidence of immediate postimplant impotence that is presumably related to needle trauma. Merrick and colleagues reported that 6 of 34 patients developed severe erectile dysfunction (IIEF < 6) in the immediate postimplant period.[7] Long-term potency among patients experiencing immediate postimplant erectile dysfunction has not been detailed, but our impression is that many will improve spontaneously over time.
FIGURE 2
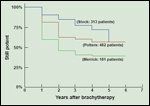
Overall Potency Following Brachytherapy-Based Treatment of Prostate Cancer
The second important factor that has become clear is that erectile dysfunction increases with time after treatment. Series with longer followup have uniformly reported lower rates of potency preservation (see Figure 2).[5,6,22]
Potency preservation rates following all treatment approaches are significantly lower when patientadministered questionnaires rather than physician interview are used to collect data. Moreover, the use of patient-administered questionnaires following radical prostatectomy has resulted in dramatically differing results.[ 33-36] Three studies that used patient-administered questionnaires reported a 7% to 44% rate of poten- cy preservation following radical prostatectomy.[34-36]
In contrast, a study in 64 patients that used a validated patient-admin istered questionnaire and a definition of potency as "the ability to engage in unassisted intercourse with or without sildenafil citrate [Viagra]" reported that 86% were potent 18 months following radical prostatectomy.[ 33] Only two patients were exclusively dependent on sildenafil. However, a possible selection bias may have been present, because only 38% of the 59 patients who agreed to participate in the study returned all four questionnaires.
Factors Affecting Potency
The mechanism of brachytherapy- induced impotence has not been definitively proven. The etiology, however, likely represents a multifactorial process including neurogenic compromise, vascular insufficiency, local trauma, and psychogenic causes.[37]
Preimplant Erectile Function
FIGURE 3

Potency Status After Brachytherapy for Prostate Cancer
-Factors that probably predispose to implant-related impotence. EB = external-beam irradiation; EF = erectile function; RT = radiotherapy; Adapted from Stock et al,[5] Merrick et al,[6] and Potters et al.[22]
In multivariate analysis, Stock and colleagues reported that pretreatment erectile function was the strongest predictor of posttreatment erectile dysfunction.[5] At 6 years, 70% of patients with normal preimplant erectile function maintained potency, compared with only 34% of patients with preimplant erections considered sufficient for intercourse but suboptimal (Figure 3).[5,6,22] Using the IIEF instrument, Merrick and colleagues also reported that preimplant erectile function was predictive of 6-year potency preservation (50% for normal erections vs 13% for preimplant erections considered suboptimal).[6]
Patient Age
Patient age may influence the likelihood of brachytherapy-induced erectile dysfunction. Potters and colleagues reported that patients < 60, 60 to 69, and ≥ 70 years old, respectively, had a 78%, 58%, and 50% potency preservation rate at 5 years following brachytherapy (P = .024, Figure 3).[22] Merrick and colleagues reported that patient age was significant in univariate but not multivariate analysis,[6] whereas Stock and colleagues found no relationship between patient age and impotence in either univariate or multivariate analyses.[5]
Supplemental External-Beam Radiation Therapy
The addition of supplemental external- beam radiation therapy appears to increase the incidence of brachytherapy- induced erectile dysfunction. Merrick and colleagues reported that the addition of supplemental external- beam irradiation to prostate brachytherapy decreased the 6-year actuarial rate of potency preservation from 52% to 26% (P < .001).[6] Potters et al reported a statistically insignificant reduction in potency when external-beam radiation was added to brachytherapy (5-year actuarial rate of potency preservation of 76% vs 56%, P = .08, Figure 3).[22]
Neoadjuvant Hormonal Manipulation
Studies investigating the relationship between neoadjuvant hormonal manipulation and potency preservation have reported mixed results. With a median follow-up of 34 months, Potters and colleagues reported the use of neoadjuvant hormonal manipulation predicted for posttreatment erectile dysfunction.[22] In patients undergoing monotherapeutic brachytherapy, 76% maintained potency, whereas when neoadjuvant hormonal manipulation was followed by brachytherapy without supplemental external-beam irradiation, the potency preservation rate was 52%.
Two other studies have failed to reveal a relationship between hormonal therapy and erectile dysfunction.[ 5,6] On univariate analysis, Stock et al reported a deleterious effect with the use of neoadjuvant hormonal therapy (potency preservation rates of 54% vs 61%, P = .04).[5] However, on multivariate analysis, neoadjuvant hormonal manipulation was not statistically significant (P = .12). Merrick et al reported that the use of hormonal manipulation did not significantly affect the ultimate rate of potency preservation (37% vs 46%, P = .836).[6]
Choice of Isotope
Of the published brachytherapy potency studies, only two have evaluated the influence of choice of isotope (Pd-103 vs I-125) on potency preservation.[ 5,6] With 6-year follow-up, neither study was able to discern a difference in potency preservation based on isotope. An ongoing prospective study comparing Pd-103 with I-125 in patients with low-risk prostate cancer (PSA ≤ 10 ng/mL, Gleason score 5/6, and clinical stage T1b/2a) will provide invaluable information regarding the ultimate effect of choice of isotope on potency preservation.[38]
Other Clinical and Treatment Parameters
Of the multitude of additional clinical and treatment parameters evaluated, one study also implicated a history of diabetes mellitus in the development of brachytherapy-induced erectile dysfunction.[6] At 2 and 6 years, patients with and without diabetes had potency preservation rates of 41.4% and 49.0% vs 0% and 0%, respectively (P = .017).[6]
Mechanism of Brachytherapy- Induced Erectile Dysfunction
Radiation-related impotence likely represents a multifactorial process.[ 37] In the case of brachytherapy, emerging evidence suggets that erectile dysfunction is related to technique and may be minimized by careful attention to source placement.
Radiation Dose to the Prostate Gland
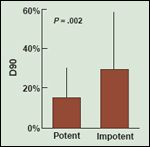
Conflicting results have been reported regarding the relationship between radiation dose and the development of postbrachytherapy erectile dysfunction. Stock and colleagues reported in multivariate analysis that a D90 (the radiation dose delivered to 90% of the prostate gland) greater than 160 Gy for I-125 and a D90 greater than 100 Gy for Pd-103 was predictive of brachytherapy- induced erectile dysfunction.[5] Although significantly different potency preservation rates were reported, the absolute differences were minimal (58% vs 64%, P = .02).
Merrick and colleagues found no correlation between the radiation dose delivered to the prostate gland and brachytherapy-induced erectile dysfunction.[ 6] Evaluated dosimetric parameters included the D90 and the volume of the prostate gland receiving 100%, 150%, and 200% of the prescription dose.
Neurovascular Bundles
Following radical prostatectomy, erectile dysfunction has been correlated with surgical trauma to the neurovascular bundles. Excessive radiation to this site represents a potential mechanism of brachytherapy- induced erectile dysfunction.[39] DiBiase and colleagues reported that radiation doses to the neurovascular bundles typically range from 130% to 226% of the prescription dose.[40]
FIGURE 4
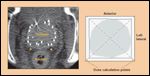
Brachytherapy Dose to Neurovascular Bundles
Merrick and colleagues evaluated the radiation dose to the neurovascular bundles by means of a twodimensional geometric model derived from day 0 computed tomography (CT) scan-based dosimetry (see Figure 4).[7,41] Because the position of the neurovascular bundles varies somewhat from patient to patient, represents a plexus and not a well-localized structure, and is not identifiable on CT scan, the radiation dose delivered to this region was determined by both point-dose measurements and dose-surface histograms.
With a median follow-up of 37 months, there was no relationship between dose to the neurovascular bundles (mean: 217% ± 55% of prescription dose) and the development of brachytherapy-induced erectile dysfunction.[7,41] Although initial reports have been negative, it is possible that, with longer follow-up, dose to the neurovascular bundles may be found to contribute to brachytherapy- related sexual dysfunction.
Penile Erectile Bodies
FIGURE 5

Penile and Erectile Tissue Anatomy
The penile erectile bodies-the paired corpora cavernosa and the midline corpus spongiosum-are potential targets for structure-specific radiation doses associated with erectile dysfunction. Followed posteriorly into the perineum, the corpora cavernosa separate and form the crura of the penis, which are attached to the inferior pubic rami. Between the two crura, the corpus spongiosum enlarges to form the penis bulb, which is attached superiorly to the inferior surface of the urogenital diaphragm (Figure 5). The penile bulb is best visualized on T2-weighted magnetic resonance images (MRIs) and appears as an oval-shaped hyperdense midline structure located approximately 10- to 15-mm inferior to the apex of the prostate gland.
Investigators at the University of California at San Francisco reported that in both a laboratory rat model and in clinical studies, the externalbeam radiation dose to the bulb of the penis correlated directly with posttreatment erectile dysfunction.[42,43] They reported a higher incidence of erectile dysfunction with increasing radiation doses to the proximal penis and concluded that external-beam radiation either directly or indirectly damaged the vascular supply of the erectile tissue as well as the nerves that supply the cavernosa smooth muscles. In addition, a histologic examination of the proximal penile shaft specimens revealed that the number of nitric oxide synthase-containing nerve fibers per corpus cavernosum was significantly decreased in the irradiated groups.
In a retrospective evaluation of 21 patients who received three-dimensional (3D) conformal external-beam radiation therapy, posttreatment potency was strongly associated with a dose of less than 40 Gy to 70% of the penis bulb (D70).[43] In addition, in patients with no change in erectile function, the median dose to 50% (D50) and 95% (D95) of the bulb of the penis was 48.5 Gy and 14.0 Gy, respectively.
FIGURE 6
Brachytherapy Dose to the Bulb of the Penis FIGURE 7

Excessive Penile Bulb Brachytherapy Doses FIGURE 8
Sagittal View of Two Brachytherapy Implants for Prostate Cancer
Merrick et al also reported that prostate brachytherapy-induced impotence was highly correlated with the radiation dose to the penis bulb.[44,45] With day 0 CT-based dosimetric evaluation, the radiation dose delivered to 50% of the penis bulb (D50) was less than 50 Gy, with a D95 of less than 20 Gy for the majority of patients who maintained potency (Figure 6). In the case of brachytherapy, suboptimal placement (either due to poor planning or poor implementation) of periapical radiation sources results in excessive radiation doses to the bulb (Figures 7 and 8). Detailed day 0 dosimetric evaluation failed to discern a significant correlation between radiation dose to the prostate gland and the bulb of the penis, further suggesting that the dose to the bulb is due to suboptimal technique or planning.[44]
With mounting evidence that penile- specific radiation doses affect potency status, more detailed dosimetric studies are in order. For instance, radiation doses to the corpora cavernosa may be more important than those to the penile bulb because the corpora cavernosa are true erectile tissue whereas the corpus spongiosum is believed to play little role in the maintenance of erectile rigidity.[ 46,47] Studies evaluating the relationship between postbrachytherapy erectile dysfunction and radiation doses to specific anatomic structures are ongoing. It is likely that a better appreciation of penile dosimetry will lead to modifications in seed placement strategies to maintain high tumor control rates while decreasing the likelihood of postimplant impotence.
Management of Brachytherapy- Induced Erectile Dysfunction
Although long-term impotence rates following brachytherapy are higher than previously believed, patients with radiation-related erectile dysfunction typically respond well to conventional erectile aids.[48] Mulhall has reported that the lack of erectile activity may be deleterious to erectile function, and as such, patients should be encouraged to develop regular erections with or without sexual relations.[46] This recommendation is based on the concept of cavernosal oxygenation. In the absence of routine penile erections, the corporal smooth muscle experiences chronic hypoxia with a resultant loss of elasticity and distensibility, which may lead to a venous leak.[49] The early institution of oral therapy in patients without spontaneous erections or semierections may help preserve or restore erectile function.[46]
FIGURE 9
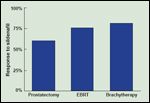
Effect of Sildenafil
As shown in Figure 9, the majority of patients with either brachytherapy-, prostatectomy-, or external beam-induced erectile dysfunction respond favorably to sildenafil citrate.[ 50-52] In a brachytherapy population, the 6-year actuarial rate of potency preservation was 92% when potent patients were grouped with erectile dysfunction patients who used sildenafil.[6] The postimplant response to sildenafil was highly dependent on preimplant potency.[6] Among patients with normal erections and suboptimal erections sufficient for vaginal penetration, 95% and 70% responded favorably to sildenafil (P = .006).
Potters et al reported that 83% of patients who underwent prostate brachytherapy (as either monotherapy or in conjunction with supplemental external-beam irradiation) responded favorably to sildenafil, whereas only 46% of patients who received neoadjuvant hormonal manipulation responded to pharmacologic support.[22] Radiation-induced erectile dysfunction also normally responds to vasoactive agents and penile implants.[53,54]
Conclusions
Brachytherapy-induced erectile dysfunction is more common than previously reported. Some but not all studies have reported that preimplant potency, patient age, diabetes mellitus, the use of supplemental external- beam radiation, radiation doses to the prostate gland, and radiation doses to the bulb of the penis are predictive of the development of posttreatment erectile dysfunction. The majority of patients with brachytherapy- induced erectile dysfunction respond favorably to sildenafil citrate.
In order to obtain the most reliable quality-of-life information and for intraobserver consistency, physicians should use validated patientadministered QOL instruments. The continued documentation and elucidation of the etiology of erectile dysfunction following permanent prostate brachytherapy may result in refinement in treatment techniques with improved potency preservation, improved treatments, and ultimately improved QOL outcomes.
Disclosures:
The author(s) have no significant financial interest or other relationship with the manufacturers of any products or providers of any service mentioned in this article.
References:
1.
Merrick GS, Butler WM, Lief JH, et al: Isbrachytherapy comparable with radical prostatectomyand external-beam radiation for clinicallylocalized prostate cancer? Tech Urol7:12-19, 2001.
2.
Blasko JC, Grimm PD, Sylvester JE: Palladium-103 brachytherapy for prostate carcinoma.Int J Radiat Oncol Biol Phys 46:839-850,2000.
3.
Merrick GS, Butler WM, Galbreath RW,et al: Five-year biochemical outcome followingpermanent interstitial brachytherapy forclinical T1-T3 prostate cancer. Int J RadiatOncol Biol Phys 51:41-48, 2001.
4.
Grimm PD, Blasko JC, Sylvester JE, et al:10-year biochemical (prostate-specific antigen)control of prostate carcinoma with (125)Ibrachytherapy. Int J Radiat Oncol Biol Phys51:32-40, 2001.
5.
Stock RG, Kao J, Stone NN: Penile erectilefunction after permanent radioactive seedimplantation for treatment of prostate cancer. JUrol 165:436-439, 2001.
6.
Merrick GS, Butler WM, Galbreath RW,et al: Erectile function after permanent prostatebrachytherapy. Int J Radiat Oncol Biol Phys52:893-902, 2002.
7.
Merrick GS, Wallner K, Butler WM, et al:Short-term sexual function after prostatebrachytherapy. Int J Cancer 96:313-319, 2001.
8.
Wallner K, Blasko J, Dattoli MJ (eds):Prostate Brachytherapy Made Complicated,2nd ed, pp 16.1-16.30. Seattle, SmartMedicinePress, 2002.
9.
NIH Consensus Conference: NIH ConsensusDevelopment Panel on Impotence: Impotence.JAMA 270:83-90, 1993.
10.
Day D, Ambegaonkar A, Harriot K, etal: A new tool for predicting erectile dysfunction.Adv Ther 18:131-139, 2001.
11.
Burnett AL: Erectile dysfunction: A practicalapproach for primary care. Geriatrics53:34-48, 1998.
12.
Laumann EO, Paik A, Rosen RC: Sexualdysfunction in the United States: Prevalenceand predictions. JAMA 281:537-544, 1999.
13.
Stone NN, Stock RG: Brachytherapy forprostate cancer: Real-time three-dimensionalinteractive seed implantation. Tech Urol 1:72-80, 1995.
14.
Stock RG, Stone NN, Iannuzzi C: Sexualpotency following interactive ultrasound-guidedbrachytherapy for prostate cancer. Int J RadiatOncol Biol Phys 35:267-272, 1996.
15.
Ragde H, Blasko JC, Grimm PD, et al:Interstitial iodine-125 radiation without adjuvanttherapy in the treatment of clinically localizedprostate cancer. Cancer 80:442-453,1997.
16.
Zelefsky MJ, Wallner KE, Ling CC, etal: Comparison of the 5-year outcome and morbidityof three-dimensional conformal radiotherapyversus transperineal permanentiodine-125 implantation for early-stage prostatecancer. J Clin Oncol 17:517-522, 1999.
17.
Chaikin DC, Broderick GA, Malloy TA,et al: Erectile dysfunction following minimallyinvasive treatments for prostate cancer. Urology48:100-104, 1996.
18.
Dattoli M, Wallner K, Sorace R, et al:103Pd brachytherapy and external beam irradiationfor clinically localized, high-risk prostaticcarcinoma. Int J Radiat Oncol Biol Phys35:875-879, 1996.
19.
Wallner K, Roy J, Harrison L: Tumorcontrol and morbidity following transperinealiodine 125 implantation for stage T1/T2 prostaticcarcinoma. J Clin Oncol 14:449-453, 1996.
20.
Zeitlin SI, Sherman J, Raboy A, et al:High dose combination radiotherapy for thetreatment of localized prostate cancer. J Urol160:91-96, 1998.
21.
Sharkey J, Chovnick S, Behar R, et al:Outpatient ultrasound-guided palladium 103brachytherapy for localized adenocarcinomaof the prostate: A preliminary report of 434patients. Urology 51:796-803, 1998.
22.
Potters L, Torre T, Fearn PA, et al: Potencyafter permanent prostate brachytherapyfor localized prostate cancer. Int J Radiat OncolBiol Phys 50:1235-1242, 2001.
23.
Feldman HA, Goldstein I, HatzichriftouDG, et al: Impotence and its medical and psychosocialcorrelates: Results of the MassachusettsMale Aging Study. J Urol 151:54-61, 1994.
24.
Lawton CA, Won M, Pilepich MV: Longtermtreatment sequelae following externalbeamirradiation for adenocarcinoma of theprostate: Analysis of RTOG studies 7506 and7706. Int J Radiat Oncol Biol Phys 21:935-939, 1991.
25.
Krupski T, Petroni GR, Bissonette EA,et al: Quality-of-life comparison of radical prostatectomyand interstitial brachytherapy in thetreatment of clinically localized prostate cancer.Urology 55:736-742, 2000.
26.
Brandeis JM, Litwin MS, Burnison CM,et al: Quality of life outcomes after brachytherapyfor early stage prostate cancer. J Urol163:851-857, 2000.
27.
Lim AJ, Brandon AH, Fiedler J, et al:Quality of life: Radical prostatectomy versusradiation therapy for prostate cancer. J Urol154:1420-1425, 1995.
28.
Wei JT, Dunn RL, Litwin MS, et al:Development and validation of the expandedprostate cancer index composite (EPIC) forcomprehensive assessment of health-relatedquality of life in men with prostate cancer.Urology 56:899-905, 2000.
29.
Litwin MS, Lubeck DP, Henning JM, etal: Differences in urologist and patient assessmentsof health related quality of life in menwith prostate cancer: Results of the CaPSUREdatabase. J Urol 159:1988-1992, 1998.
30.
Blander DS, Sanchez-Ortiz RF, BroderickGA: Sex inventories: Can questionnairesreplace erectile dysfunction testing? Urology54:719-723, 1999.
31.
Rosen RC, Riley A, Wagner G, et al: Theinternational index of erectile function (IIEF):A multidimensional scale for assessment oferectile dysfunction. Urology 49:822-830,1997.
32.
Wallner K, Merrick G, True L, et al: I-125 versus Pd-103 for low risk prostate cancer:Preliminary urinary functional outcomes froma prospective randomized trial. J BrachytherInt 16:151-155, 2000.
33.
Walsh PC, Marschke P, Ricker D, et al:Patient-reported urinary continence and sexualfunction after anatomic radical prostatectomy.Urology 55:58-61, 2000.
34.
Talcott JA, Rieker P, Clark JA, et al:Patient-reported symptoms after primary therapyfor early prostate cancer: Results of a prospectivecohort study. J Clin Oncol 16:275-283,1998.
35.
Talcott JA, Rieker P, Propert KJ, et al:Patient-reported impotence and incontinenceaftger nerve-sparing radical prostatectomy. JNatl Cancer Inst 89:1117-1123, 1997.
36.
Stanford JL, Feng Z, Hamilton AS, et al:Urinary and sexual function after radical prostatectomyfor clinically localized prostate cancer:The Prostate Cancer Outcomes Study.JAMA 283:354-360, 2000.
37.
Zelefsky MJ, Eid JF: Elucidating theetiology of erectile dysfunction after definitivetherapy for prostate cancer. Int J Radiat OncolBiol Phys 40:129-133, 1998.
38.
Wallner K, Merrick GM, True L, et al: I-125 versus Pd-103 for low-risk prostate cancer:Morbidity outcomes from a prospectiverandomized multicenter trial. Cancer J 8:67-73, 2002.
39.
Lepor H, Gregerman M, Crosby R, et al:Precise localization of the autonomic nervesfrom the pelvic plexus to the corpora cavernosa:A detailed anatomical study of the adultmale pelvis. J Urol 133:207-212, 1985.
40.
DiBiase SJ, Wallner K, Tralins K, et al:Brachytherapy radiation doses to the neurovascularbundles. Int J Radiat Oncol Biol Phys46:1301-1307, 2000.
41.
Merrick GS, Butler WM, Dorsey AT, etal: A comparison of the radiation dose to theneurovascular bundles in men with and withoutprostate brachytherapy induced erectiledysfunction. Int J Radiat Oncol Biol Phys48:1069-1074, 2000.
42.
Carrier S, Hricak H, Lee S, et al: Radiation-induced decrease in nitric oxide synthasecontainingnerves in the rat penis. Radiology195:95-99, 1995.
43
Fisch BM, Pickett B, Weinberg V, et al:Dose of radiation received by the bulb of thepenis correlates with risk of impotence afterthree-dimensional conformal radiotherapy forprostate cancer. Urology 57:955-959, 2001.
44.
Merrick GS, Wallner K, Butler WM, etal: A comparison of radiation dose to the bulbof the penis in men with and without prostatebrachytherapy induced erectile dysfunction. IntJ Radiat Oncol Biol Phys 50:597-604, 2001.
45.
Merrick GS, Wallner K, Benson M, et al:Penile bulb imaging. Int J Radiat Oncol BiolPhys 53:928-933, 2002.
46.
Mulhall JP: Minimizing radiation-inducederectile dysfunction. J Brachyther Int17:221-227, 2001.
47.
Mulhall JP, Yonover P, Sethi A, et al:Radiation exposure to the corporeal bodies during3-dimensional conformal radiation therapyfor prostate cancer. J Urol 167:539-542, 2002.
48.
Perez MA, Meyerowitz BE, LieskovskyG, et al: Quality of life and sexuality followingradical prostatectomy in patients with prostatecancer who use or do not use erectile aids.Urology 50:740-746, 1997.
49.
Moreland RB: Is there a role of hypoxemiain penile fibrosis: A viewpoint presentedto the Society for the Study of Impotence. Int JImpot Res 10:113-120, 1998.
50.
Merrick GS, Butler WM, Lief JH, et al:Efficacy of sildenafil citrate in prostate brachytherapypatients with erectile dysfunction. Urology53:1112-1116, 1999.
51.
Lowentritt BH, Scardino PT, Miles BJ,et al: Sildenafil citrate after radical retropubicprostatectomy. J Urol 162:1614-1617, 1999.
52.
Zelefsky MJ, McKee AB, Lee H, et al:Efficacy of oral sildenafil in patients with erectiledysfunction after radiotherapy for carcinomaof the prostate. Urology 53:775-778, 1999.
53.
Pierce LJ, Whittington R, Hanno PM, etal: Pharmacologic erection with intracavernosalinjection for men with sexual dysfunctionfollowing irradiation: A preliminaryreport. Int J Radiat Oncol Biol Phys 21:1311-1314, 1991.
54.
Dubocq FM, Bianco FJ Jr, Maralani SJ,et al: Outcome analysis of penile implant surgeryafter external beam radiation for prostatecancer. J Urol 158:1787-1790, 1997.
Prolaris in Practice: Guiding ADT Benefits, Clinical Application, and Expert Insights From ACRO 2025
April 15th 2025Steven E. Finkelstein, MD, DABR, FACRO discuses how Prolaris distinguishes itself from other genomic biomarker platforms by providing uniquely actionable clinical information that quantifies the absolute benefit of androgen deprivation therapy when added to radiation therapy, offering clinicians a more precise tool for personalizing prostate cancer treatment strategies.