Management of Xerostomia Related to Radiotherapy for Head and Neck Cancer
Xerostomia is a permanent and devastating sequela of head and neckirradiation, and its consequences are numerous. Pharmaceutical therapyattempts to preserve or salvage salivary gland function through systemicadministration of various protective compounds, most commonlyamifostine (Ethyol) or pilocarpine. When these agents are ineffective orthe side effects too bothersome, patients often resort to palliative care, forexample, with tap water, saline, bicarbonate solutions, mouthwashes, orsaliva substitutes. A promising surgical option is the Seikaly-Jha procedure,a method of preserving a single submandibular gland by surgicallytransferring it to the submental space before radiotherapy. Improved radiationtechniques, including intensity-modulated radiotherapy andtomotherapy, allow more selective delivery of radiation to defined targetsin the head and neck, preserving normal tissue and the salivary glands.Acupuncture may be another option for patients with xerostomia. All ofthese therapies need to be further studied to establish the most effectiveprotocol to present to patients before radiotherapy has begun.
Xerostomia is a permanent and devastating sequela of head and neck irradiation, and its consequences are numerous. Pharmaceutical therapy attempts to preserve or salvage salivary gland function through systemic administration of various protective compounds, most commonly amifostine (Ethyol) or pilocarpine. When these agents are ineffective or the side effects too bothersome, patients often resort to palliative care, for example, with tap water, saline, bicarbonate solutions, mouthwashes, or saliva substitutes. A promising surgical option is the Seikaly-Jha procedure, a method of preserving a single submandibular gland by surgically transferring it to the submental space before radiotherapy. Improved radiation techniques, including intensity-modulated radiotherapy and tomotherapy, allow more selective delivery of radiation to defined targets in the head and neck, preserving normal tissue and the salivary glands. Acupuncture may be another option for patients with xerostomia. All of these therapies need to be further studied to establish the most effective protocol to present to patients before radiotherapy has begun.
An often severe form of xerostomia results from iatrogenic salivary gland destruction caused by therapeutic doses of irradiation given to treat head and neck cancer. Salivary tissue is sensitive to external-beam irradiation, and xerostomia often develops when the glands are included in the treatment fields.[1] Classical standard radiation portals almost universally include the major salivary glands within the treatment fields because of their proximity to primary disease, or to at-risk nodal basins in the neck.[2] The magnitude of damage depends both on the volume of tissue irradiated and the dose of radiation delivered.[3] The usual radiation dosage required in the treatment of head and neck cancer exceeds 50 Gy, resulting in a high likelihood of impairment of irradiated salivary tissue and the development of xerostomia.[4,5] Xerostomia is a permanent and devastating sequela of head and neck irradiation, and its consequences are numerous, affecting most aspects of the patient's life.[6,7] Xerostomia impairs mastication, deglutition, and gustation and alters oral microbial flora, leading to the development of caries. The oral mucosa becomes dry, cracked, and painful. Patients may develop a low tolerance for dental prostheses because of tissue friability and lack of lubrication.[7,8] Speech is altered and swallowing is impaired, as movement of a bolus from the mouth to the pharynx is slowed.[9] Food choices are sometimes altered, leading to nutritional deficiencies.[10] Sleep disturbances are also common, as patients wake up to moisten their dry mouth or to relieve the polyuria experienced because of polydipsia.[1] Since saliva also facilitates formation of speech patterns, its loss hinders speaking and communicating. Together these conditions can impair the physiological and psychological well-being of the patient. These severe consequences have prompted researchers to actively search for ways to minimize the xerostomia experienced by head and neck cancer patients. To this end, physicians have several options to consider in this setting before, during, and after radiotherapy. Pharmaceutical Therapy Pharmaceutical therapy attempts to preserve or salvage salivary gland function through systemic administration of various protective compounds. The two most commonly used agents are amifostine (Ethyol) and pilocarpine hydrochloride.[11] Amifostine
Amifostine is a thiol-containing compound that has been recognized for decades for its radioprotective potential. Its mechanism of radioprotection is through the scavenging of radiation-induced free radicals.[1] Amifostine has shown some efficacy in clinical trials at reducing short-term and long-term xerostomia after radiotherapy.[ 11-13] The first large-scale randomized study to report the successful clinical use of amifostine was recently published by Brizel and associates.[11] Patients who were pretreated daily with amifostine had a significantly lower incidence of acute xerostomia (grade ≥ 2) than those who received radiotherapy alone. Furthermore, patients pretreated with amifostine who developed xerostomia did not do so until higher cumulative doses of irradiation had been delivered. Moderate to severe chronic xerostomia was significantly less prevalent with the use of amifostine. In addition, patients on amifostine produced larger volumes of saliva than those who did not receive the drug. Longitudinal posttreatment patient self-assessment showed fewer symptoms among patients who received amifostine. Likewise, 1 year after the completion of treatment, chronic xerostomia occurred significantly less frequently in patients who received amifostine compared with those who did not. Amifostine's main disadvantages are the need for daily intravenous infusions, prohibitive price, and associated systemic effects, which result in treatment discontinuation in 21% of patients.[11] Nausea, vomiting, hypotension, and allergic reactions are the most common side effects. Brizel and colleagues[11] examined tumor recurrence patterns among patients who received amifostine vs those who did not and found that amifostine did not compromise the antitumor efficacy of radiotherapy. Eighteen-month actuarial local-regional control rates were 65% vs 68% with and without amifostine, respectively. Overall survival was better in patients receiving amifostine than in those who did not, although this difference was not statistically significant.[11] Researchers are continuing to examine whether an administration route more convenient than daily IV injection is possible and associated with less toxicity. In addition, attempts are being made to better understand the role of this drug in treatment schemes that use modified fractionation schedules and/or concurrent chemotherapy. Pilocarpine
Pilocarpine hydrochloride is a direct- acting cholinergic parasympathomimetic agent. It acts through direct stimulation of muscarinic receptors and can have broad, widely distributed effects on smooth muscle and exocrine tissues. Pilocarpine produces increased smooth muscle tone of the gastrointestinal and genitourinary systems, eye, and respiratory tract. Exocrine glands of the lacrimal, gastric, intestinal, respiratory tract, and salivary systems are also affected.[14] The benefits from pilocarpine may arise from the hyperstimulation of small residual volumes of unirradiated parotid gland. The usefulness of pilocarpine when the entirety of both parotids have been irradiated to high doses is unclear.[15,16] Results with pilocarpine have been inconsistent, but several investigations[ 15,17,18] have demonstrated its effectiveness at reducing the rate of xerostomia when used after radiotherapy. These placebo-controlled, randomized trials found that approximately 50% of patients given 5 to 10 mg three times daily noted an improvement in the sensation of oral dryness. Warde and colleagues examined the use of pilocarpine during and after radiotherapy for head and neck cancer in 130 patients in a phase III placebo-controlled trial.[19] These patients began taking pilocarpine on the first day of radiotherapy and continued its use until 1 month after the completion of radiotherapy. Their study found no beneficial effect of pilocarpine for xerostomia when examining a patient-completed linear analog scale measuring the acute toxicity of radiotherapy or a patient questionnaire measuring quality of life at 1, 3, and 6 months after radiotherapy completion. These findings are at variance with nonrandomized data from Zimmerman and associates.[20] In the latter study, patients also began pilocarpine on the first day of radiotherapy and continued its use until 3 months following radiotherapy. This study found that the use of pilocarpine was associated with significantly less subjective xerostomia than that reported by a similar cohort of patients who had not received pilocarpine (P < .01). Unpleasant cholinergic side effects occur in approximately half of the patients using this drug.[14] Adverse reactions can include gastrointestinal effects (nausea, vomiting, epigastric distress, abdominal cramping, diarrhea), genitourinary effects (bladder tightness, urinary frequency), central nervous system effects (headache, syncope, tremors), cardiovascular effects (flushing, diaphoresis, hypotension, hypertension, bradycardia, arrhythmias). Pilocarpine therapy must be used judiciously if cardiac failure, bronchial asthma, urinary tract obstruction, peptic ulcer, gastrointestinal spasm, hyperthyroidism, or Parkinson's disease are present.[14] Despite these severe side effects and mixed results, pilocarpine continues to be used by patients today. In many instances, patients require lifelong treatment with this drug.[14] Therefore, these patients may experience a lifetime of extremely bothersome and possibly hazardous consequences in an attempt to relieve their xerostomia. Cevimeline
Cevimeline (Evoxac) is another muscarinic agonist often used for the treatment of radiation-induced xerostomia. Several studies have reported on this drug's effect on xerostomia attributable to Sjgren's syndrome; however, there have been no published trials of the drug for radiation-induced xerostomia. Fife and colleagues conducted a 6- week, randomized, double-blind, placebo- controlled study in 75 patients with Sjgren's syndrome[21]; 76% of the patients in the 30 mg tid group reported a global improvement in their dry mouth symptoms, compared to 35% of the patients in the placebo group. This difference was statistically significant at P = .0043. There was no evidence that patients in the 60-mgtid group had better global evaluation scores than the patients in the 30-mgtid group. Although both dosages of cevimeline provided symptomatic improvement, 60 mg three times daily was associated with an increase in the occurrence of adverse events, particularly gastrointestinal tract disorders A 12-week, randomized, doubleblind, placebo-controlled study was conducted in 197 patients with Sjgren's syndrome by Petrone and associates.[22] Statistically significant global improvement in the symptoms of dry mouth (P = .0004) was seen for the 30-mg-tid group compared to placebo, but not for the 15-mg group, compared to placebo. Like pilocarpine, cevimeline has numerous side effects due to its parasympathomimetic properties. These may include headache, visual disturbance, lacrimation, sweating, respiratory distress, gastrointestinal spasm, nausea, vomiting, diarrhea, atrioventricular block, tachycardia, bradycardia, hypotension, hypertension, shock, mental confusion, cardiac arrhythmia, and tremors.[23] Again, in order for patients to gain any relief from their xerostomia, many have to endure numerous and at times debilitating consequences. Palliative Care When pharmaceutical agents have proven to be ineffective or the side effects become too bothersome, patients often resort to palliative care to treat their xerostomia. Patients may choose to treat with household products such as tap water, saline, or bicarbonate solutions.[24] The solutions simply provide the patient with a method of continually moistening the oral tissues. In addition, sugarless lozenges and gums are often used and have been found to provide significant relief in some patients. A study by Markovic and colleagues found the use of a sorbitol-sweetened gum was associated with statistically significant stimulated whole mouth and parotid salivary flow rate increases, compared to unstimulated whole mouth and parotid salivary flow rates.[25] Commercially available mouthwashes may provide some relief by acting as a moistening agent for the oral tissues. In addition, several mouthwashes have been further developed to provide antimicrobial proteins that reduce the incidence of caries in these at-risk patients.[26] An increasingly popular option is the use of saliva substitutes, which have been developed to provide a more lasting sensation of oral wetness than the above mentioned therapies. The substitutes usually contain a polymer based on carboxymethylcellulose, which is used as a thickening agent to provide lubrication to the oral tissues.[24] Other commercially available saliva substitutes contain natural mucins-primarily porcine gastric mucin or bovine submandibular mucin[ 27]-to better simulate human saliva. Research is currently being directed at developing artificial saliva that not only provides lubrication and oral moistness, but like natural human salivation, also protects against microorganisms.[24] Surgery A surgical option that continues to show promise is a method of preserving a single submandibular gland by surgically transferring it to the submental space before radiotherapy.[ 28,29] The surgery is referred to as the Seikaly-Jha procedure (SJP) of submandibular gland transfer.[2] Current thought is that sparing the submandibular gland is preferable to sparing the parotid gland, because it is responsible for most of the resting state salivary volume.[2] The parotid glands normally contribute about 20% of the total volume of unstimulated saliva, while the submandibular salivary glands contribute 65% and the sublingual salivary glands contribute 7% to 8%.[30,31] At the high flow rates produced during eating, the parotid becomes the dominant gland, contributing 50% of salivary volume.[ 30] Several studies have demonstrated that the unstimulated saliva flow (resting state) is more important in the subjective symptoms of xerostomia and in preservation of oral homeostasis.[2] The transferred submandibular gland is shielded from radiation after surgery, without changing the standard radiation prescription to the primary disease areas or the neck nodes.[29] The initial outcomes of a phase II trial of this procedure were encouraging and resulted in the prevention of xerostomia in 81% of the patients.[32] The long-term results of this trial continue to be encouraging.[ 2] Xerostomia was prevented in 83% of patients in the surgical group. These results are clinically and statistically significant when contrasted with the development of xerostomia in all the patients in the nonsurgical group. The procedure appears to be oncologically sound and safe, as there were no changes in the pattern of disease recurrence or recurrences in the submental space. A direct comparison of the SJP and chemoprevention outcomes is difficult based on the literature because of the variable methods of reporting.[2] Radiotherapy Improved radiation techniques allow minimization of the dose received by the salivary glands. Three-dimen- sional radiation delivery techniques such as intensity-modulation radiotherapy (IMRT)[33] and tomotherapy[ 34] permit more selective delivery of radiation to defined targets in the head and neck, preserving normal tissue and the salivary glands.[35]
One study by Dawson and associates found parotid gland preservation to be possible when the total dose to the gland was less than 26 Gy, but the local recurrence rate was disappointingly high (21%).[36] Other studies have continued to find IMRT to be effective at reducing the severity of xerostomia with excellent tumor control rates.[37-42] Munter and associates[37] reported that with their use of IMRT, it was possible in 16 of 18 patients to reduce the dose for at least one parotid gland to ≤ 26 Gy. In seven of these patients, protection of both parotid glands was possible. In their study, no recurrent disease adjacent to the protected parotid glands was observed, and only three patients had grade 2 xerostomia. A study by Bussels and colleagues[ 38] examined the treatment of patients with parotid-sparing radiotherapy by omitting irradiation to the junctional nodes contralateral to the tumor. This technique proved to be safe, with no recurrences developing in the spared area and reduced patient-reported xerostomia. Chao and associates[39] found an exponential relationship between saliva flow reduction and mean parotid dose for each gland. More specifically, stimulated saliva flow at 6 months after treatment was reduced exponentially, for each gland independently, at a rate of approximately 4% per Gy of mean parotid dose. Eisbruch and associates[42] describe a "threshold effect" in that glands receiving a lower mean dose than 26 Gy showed a time-related recovery, whereas most glands receiving a higher dose produced no measurable salivary output and did not recover over time. This study showed the functional recovery of the glands that received low to moderate doses to continue during the second year following radiotherapy. The results of this study suggest that the effort to spare the major salivary glands can gain meaningful improvement of xerostomia over time. The results of IMRT are expected to continue to improve with further development and standardization of the technologies and increased familiarity of radiation oncologists with the new treatment protocols. We hope that xerostomia may decrease apace. Acupuncture Acupuncture may be an option for patients with xerostomia secondary to radiation therapy for head and neck malignancy. The mechanism by which acupuncture or electrical stimulation relieves xerostomia is elusive.[43] Consideration of the Eastern philosophy from which acupuncture emerges supports the representation that xerostomia is due to a blockage of qi (pronounced "chee"). Thus, acupuncture removes that block and restores the host to "balance," or homeostasis. This is clearly difficult to translate to a Western construct. Western practitioners believe parasympathetic mediation to be responsible in some part, although the plethora of points and stimulation techniques referred to below make that claim tenuous and difficult to prove.
Nevertheless, there is an emerging body of literature supporting acupuncture for symptom relief in patients with xerostomia secondary to head and neck irradiation. Although the most reliable acupuncture technique is currently under study, the studies discussed below have all shown promise. Clinical Trials
- Swedish Data- The first recent descriptions of large trial data of acupuncture for xerostomia were authored by Blom and colleagues.[44] These authors initially conducted a trial in 38 patients. Acupuncture was delivered to 5 to 8 of 28 points shown in the manuscript. When analyzed over the entire acupuncture population, all patients had increased flow rates at the 6-month follow-up evaluation compared to baseline. Patients continuing with acupuncture regimens subsequently had higher stimulated and unstimulated flow rates at 3 years compared to patients who had not continued undergoing acupuncture.[45]
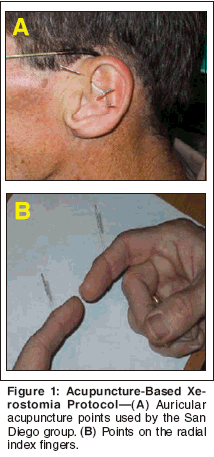
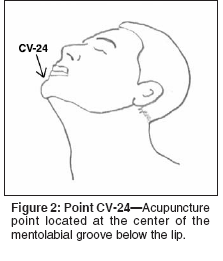
- San Diego Data- A study published by San Diego investigators uses a much simpler auricular-based xerostomia protocol.[46] This technique involves needling three points in the bilateral ears (Figure 1A), and a single point in the distal radial aspect of the index finger (Figure 1B). Median palliation using a subjective questionnaire (Xerostomia Inventory [XI][47]) was 9 points (range: 0-25 points), and 35 patients (70%) noted an improvement of 10% or better over baseline XI values. Using a cutoff of 10 points or greater on the XI, 24 patients (48%) received benefit. Duration of effect for 13 patients (26%) exceeded 3 months. An unpublished practice analysis recently evaluated the addition of the acupuncture point CV-24 (Chengjiang), a point located at the center of the mentolabial groove directly below the lip (Figure 2). Patients treated with the previously reported acupuncture regimen[46] with the addition of an acupuncture needle at CV-24, experienced enhanced salivation in many cases.
- Ontario Data- In 2003, a group from McMaster University published results of a phase I/II study with transcutaneous electrical nerve stimulation using a proprietary device (CODETRON).[48] For the 37 who completed therapy, statistically increased salivation using a visual analog scale was reported at both 3 and 6 months of follow-up. Furthermore, statistically increased basal and stimulated salivation was noted at both follow- up durations.
- Ongoing Trials- Prospective, randomized trials of acupuncture for xerostomia after head and neck radiotherapy are ongoing in France and in a collaborative program between Emory University and Memorial Sloan- Kettering Cancer Center. With the results of these and other ongoing studies, the technique and understanding of the use of acupuncture for xerostomia are expected to make great strides.
Conclusions In what used to be an inevitable consequence of radiotherapy for head and neck cancer, xerostomia is becoming a treatable and, at times, preventable side effect. The above therapies all show significant promise; however, it is obvious that xerostomia continues to be a debilitating consequence for many patients. In order to offer these patients an improved quality of life, all of the above therapies need to be further studied in order to establish the most effective protocol to present to patients before any radiotherapy has begun. It is likely that several of the above therapies may be used to minimize damage to the salivary glands and maximize saliva after radiotherapy. Once these techniques become established, xerostomia may be less bothersome to these patients who have already faced a very troublesome diagnosis.
Disclosures:
The authors have no significant financial interest or other relationship with the manufacturers of any products or providers of any service mentioned in this article. Dr. Johnstone is a Georgia Cancer Coalition Distinguished Cancer Scholar.
References:
1. Taylor SE, Miller EG: Preemptive pharmacologic intervention in radiation-induced salivary dysfunction. Proc Soc Exper Biol Med 221:14-26, 1999.
2. Seikaly H, Jha N, Harris JR, et al: Longterm outcomes of submandibular gland transfer for prevention of postradiation xerostomia. Arch Otolaryngol Head Neck Surg 130:956- 961, 2004.
3. Emami B, Lyman J, Brown A, et al: Tolerance of normal tissue to therapeutic irradiation. Int J Radiat Oncol Biol Phys 21:109-122, 1991.
4. Wescott WB, Mira JG, Starcke EN, et al: Alterations in whole saliva flow rate induced by fractionated radiotherapy. AJR Am J Roentgenol 130:145-149, 1978.
5. Mossman K, Shatzman A, Chencharick J: Long-term effects of radiotherapy on taste and salivary function in man. Int J Radiat Oncol Biol Phys 8:991-997, 1982.
6. Wijers OB, Levendag PC, Braaksma MM, et al: Patients with head and neck cancer cured by radiation therapy: A survey of the dry mouth syndrome in long-term survivors. Head Neck 24:737-747, 2002.
7. Fox PC, van der Ven PF, Sonies BC, et al: Xerostomia: Evaluation of a symptom with increasing significance. J Am Dent Assoc 110:519-525, 1985.
8. Vissink A, Panders AK, Gravenmade EJ, et al: The causes and consequences of hyposalivation. Ear Nose Throat J 67:166-168, 173-176, 1988.
9. Hughes PJ, Scott PM, Kew J, et al: Dysphagia in treated nasopharyngeal cancer. Head Neck 22:393-397, 2000.
10. Chencharick JD, Mossman KL: Nutritional consequences of the radiotherapy of head and neck cancer. Cancer 51:811-815, 1983.
11. Brizel DM, Wasserman TH, Henke M, et al: Phase III randomized trial of amifostine as a radioprotector in head and neck cancer. J Clin Oncol 18:3339-3345, 2000.
12. Antonadou D, Pepelassi M, Synodinou M, et al: Prophylactic use of amifostine to prevent radiochemotherapy-induced mucositis and xerostomia in head-and-neck cancer. Int J Radiat Oncol Biol Phys 52:739-747, 2002.
13. Bohuslavizki KH, Klutmann S, Bleckmann C, et al: Salivary gland protection by amifostine in high-dose radioiodine therapy of differentiated thyroid cancer. Strahlenther Onkol 175:57-61, 1999.
14. Hamlar DD, Schuller DE, Gahbauer RA, et al: Determination of the efficacy of topical oral pilocarpine for postirradiation xerostomia in patients with head and neck carcinoma [1994 annual meeting paper]. Laryngoscope 106:972- 976, 1996.
15. Johnson JT, Ferretti GA, Nethery WJ, et al: Oral pilocarpine for post-irradiation xerostomia in patients with head and neck cancer. N Engl J Med 329:390-395, 1993.
16. LeVeque FG, Montgomery M, Potter D, et al: A multicenter, randomized, double-blind, placebo-controlled, dose-titration study of oral pilocarpine for treatment of radiation-induced xerostomia in head and neck cancer patients. J Clin Oncol 11:1124-1131, 1993.
17. Horiot JC, Lipinski F, Schraub S, et al: Post-radiationsevere xerostomia relieved by pilocarpine: A prospective French cooperative study. Radiother Oncol 55:233-239, 2000.
18. Rieke JW, Hafermann MD, Johnson JT, et al: Oral pilocarpine for radiation-induced xerostomia: Integrated efficacy and safety results from two prospective randomized clinical trials. Int J Radiat Oncol Biol Phys 31:661- 669, 1995.
19. Warde P, O’Sullivan B, Aslandis J, et al: A phase III placebo-controlled trial of oral pilocarpine in patients undergoing radiotherapy for head and neck cancer. Int J Radiat Oncol Biol Phys 54: 9-13, 2002.
20. Zimmerman RP, Mark RJ, Tran LM, et al: Concomitant pilocarpine during head and neck irradiation is associated with decreased posttreatment xerostomia. Int J Radiat Oncol Biol Phy 37:571-575, 1997.
21. Fife RS, Chase WF, Dore RK, et al: Cevimeline for the treatment of xerostomia in patients with Sjogren syndrome: A randomized trial. Arch Intern Med 162:1293-1300, 2002.
22. Petrone D, Condemi JJ, Fife RS, et al: A double-blind, randomized, placebo-controlled study of cevimeline in Sjogren’s syndrome patients with xerostomia and keratoconjunctivitis sicca. Arthritis Rheum 46:748-754, 2002.
23. Mosby’s Drug Consult. St. Louis, Mosby, 2005.
24. Nieuw Amerongen AV, Veerman EC: Current therapies for xerostomia and salivary gland hypofunction associated with cancer thera pies. Support Care Cancer 11:226-231, 2003.
25. Markovic N, Abelson DC, Mandel ID: Sorbitol gum in xerostomics: The effect on dental plaque pH and salivary flow rates. Gerodontology 7:71-75, 1988.
26. Epstein JB, Emerton S, Le ND, et al: A double-blind crossover trial of Oral Balance gel and Biotene toothpaste versus placebo in patients with xerostomia following radiation therapy. Oral Oncol 35:132-137, 1999.
27. Davies AN: A comparison of artificial saliva and chewing gum in the management of xerostomia in patients with advanced cancer. Palliat Med 14:197-203, 2000.
28. Seikaly H, Jha N, McGaw T, et al: Submandibular gland transfer: A new method of preventing radiation-induced xerostomia. Laryngoscope 111:347-352, 2001.
29. Liu R, Seikaly H, Jha N: Anatomic study of submandibular gland transfer in an attempt to prevent postradiation xerostomia. J Otolaryngol 31:76-79, 2002.
30. Dawes C: Factors influencing saliva flow rate and composition in Edgar WM (ed): Saliva and Oral Health, pp 27-41. London, British Dental Association, 1996.
31. Scheneyer L: Source of resting total mixed saliva of man. J Appl Physiol 9:79-81, 1956.
32. Jha N, Seikaly H, McGaw T, et al: Submandibular salivary gland transfer prevents radiation- induced xerostomia. Int J Radiat Oncol Biol Phys 46:7-11, 2000.
33. Eisbruch A, Dawson LA, Kim HM, et al: Conformal and intensity modulated irradiation of head and neck cancer: The potential for improved target irradiation, salivary gland function, and quality of life. Acta Otorhinolaryngol Belg 53:271-275, 1999.
34. Mackie TR, Balog J, Ruchala K, et al: Tomotherapy. Semin Radiat Oncol 9:108-117, 1999.
35. Eisbruch A, Kim HM, Terrell JE, et al: Xerostomia and its predictors following parotidsparing irradiation of head-and-neck cancer. Int J Radiat Oncol Biol Phys 50:695-704, 2001.
36. Dawson LA, Anzai Y, Marsh L, et al: Patterns of local-regional recurrence following parotid-sparing conformal and segmental intensity- modulated radiotherapy for head and neck cancer. Int J Radiat Oncol Biol Phys 46:1117-1126, 2000.
37. Munter MW, Karger CP, Hoffner SG, et al: Evaluation of salivary gland function after treatment of head-and-neck tumors with intensity- modulated radiotherapy by quantitative pertechnetate scintigraphy. Int J Radiat Oncol Biol Phys 58:175-184, 2004.
38. Bussels B, Maes A, Hermans R, et al: Recurrences after conformal parotid-sparing radiotherapy for head and neck cancer. Radiother Oncol 72:119-127, 2004.
39. Chao KS, Deasy JO, Markman J, et al: A prospective study of salivary function sparing in patients with head-and-neck cancers receiving intensity-modulated or three-dimensional radiation therapy: Initial results. Int J Radiat Oncol Biol Phys 49:907-916, 2001.
40. Lin A, Kim HM, Terrell JE, et al: Quality of life after parotid-sparing IMRT for headand- neck cancer: a prospective longitudinal study. Int J Radiat Oncol Biol Phys 57:61-70, 2003.
41. Chao KS, Majhail N, Huang CJ, et al: Intensity-modulated radiation therapy reduces late salivary toxicity without compromising tumor control in patients with oropharyngeal carcinoma: A comparison with conventional techniques. Radiother Oncol 61:275-280, 2001.
42. Eisbruch A, Kim HM, Terrell JE, et al: Xerostomia and its predictors following parotid- sparing irradiation of head-and-neck cancer. Int J Radiat Oncol Biol Phys 50:695-704, 2001.
43. Johnstone PAS, Riffenburgh RH, Niemtzow RC: Acupuncture for xerostomia: Clinical update. Cancer 94:1151-1156, 2002.
44. Blom M, Davidson I, Fernberg JO, et al: Acupuncture treatment of patients with radiation- induced xerostomia. Eur J Cancer B Oral Oncol 32B:182-190, 1996.
45. Blom M, Lundeberg T: Long-term follow- up of patients treated with acupuncture for xerostomia and the influence of additional treatment. Oral Dis 6:15-24, 2000.
46. Johnstone PAS, Peng YP, May BC, et al: Acupuncture for pilocarpine-resistant xerostomia following radiotherapy for head and neck malignancies. Int J Radiat Oncol Biol Phys 50:353-357, 2001.
47. Thomson WM, Chalmers JM, Spencer AJ, et al: The Xerostomia Inventory: A multiitem approach to measuring dry mouth. Community Dent Health 16:12-17, 1999.
48. Wong RKW, Jones GE, Sagar SM, et al: A phase I-II study in the use of acupuncturelike transcutaneous nerve stimulation in the treatment of radiation-induced xerostomia in head-and-neck cancer patients treated with radical radiotherapy. Int J Radiat Oncol Biol Phys 57:472-480, 2003.
Oncology Peer Review On-The-Go: Cancer-Related Fatigue Outcome Measures in Integrative Oncology
September 20th 2022Authors Dori Beeler, PhD; Shelley Wang, MD, MPH; and Viraj A. Master, MD, PhD, spoke with CancerNetwork® about a review article on cancer-related fatigue published in the journal ONCOLOGY®.
Oncology Peer Review On-The-Go: Cancer-Related Fatigue Outcome Measures in Integrative Oncology
September 20th 2022Authors Dori Beeler, PhD; Shelley Wang, MD, MPH; and Viraj A. Master, MD, PhD, spoke with CancerNetwork® about a review article on cancer-related fatigue published in the journal ONCOLOGY®.
2 Commerce Drive
Cranbury, NJ 08512
All rights reserved.