PET Finds Responders to Preop Chemotherapy for Esophagogastric Junction Ca
Early metabolic imaging with positron emission tomography (PET) identifies responders to neoadjuvant chemotherapy for advanced adenocarcinoma of the esophagogastric junction
WASHINGTONEarly metabolic imaging with positron emission tomography (PET) identifies responders to neoadjuvant chemotherapy for advanced adenocarcinoma of the esophagogastric junction, German researchers reported at the Society of Nuclear Medicine (SNM) meeting (abstract 93).
"This is the first study to apply PET results from early metabolic response assessment to clinical decision making in the treatment of common solid tumors," said Ken Herrmann, MD, of the Department of Nuclear Medicine at the Technical University in Munich. The study was initiated by Florian Lordick, MD, of the Department of Surgery.
Dr. Herrmann noted that "the outcome for metabolic responders turned out to be remarkably favorable, compared to metabolic nonresponders."
Who Will Benefit?
The study, part of the MUNICON trial, was designed to determine whether PET could discern between those patients who would benefit from neoadjuvant chemotherapy and those who would not respond and therefore lose valuable time and suffer side effects with no corresponding benefit. Researchers used 18-fluorodeoxyglucose (FDG) with PET to make their decisions.
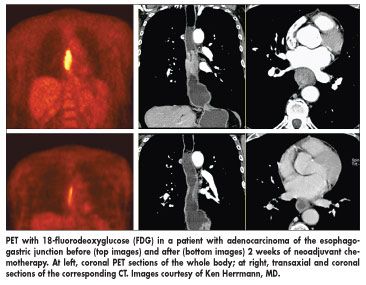
Between May 2002 and August 2005, 111 patients were enrolled in the study. Eligible patients had potentially R0 resectable adenocarcinoma of the esophagogastric junction determined by endoscopic ultrasound, CT, and FDG-PET.
Patients with a greater than 35% decrease of tumor standardized uptake value (SUV) after 2 weeks of neoadjuvant platin/5-fluorouracil-based chemotherapy, compared with baseline, were defined as metabolic responders. They continued on chemotherapy for 12 weeks before surgery.
Patients with less than a 35% SUV decrease were classified as metabolic nonresponders. Chemotherapy was stopped after the initial 2 weeks for nonresponders, and they proceeded to immediate surgery. Follow-up CT scans and endoscopy were performed every 3 months in the first year and every 6 months thereafter.
110 Assessable Patients
Of the 111 patients enrolled, 110 were assessable for metabolic response. The overall response rate after 2 weeks of neoadjuvant chemotherapy was 49%. A total of 104 patients underwent resection (subtotal esophagectomy 70%, extended gastrectomy 30%).
Major histologic remissions (less than 10% residual tumor) were observed in 58% of metabolic responders vs none for the metabolic nonresponders (P < .001). R0 resection rate was 96% in responders and 74% in nonresponders, indicating a significant difference.
After a median follow-up of 2.3 years, metabolic responders have obtained a significantly better median event-free survival of 29.7 months vs 14.1 months for metabolic nonresponders (P < .002), Dr. Herrmann said. Median overall survival for metabolic responders was not reached vs 25.8 months for metabolic nonresponders (P < .015).