SEAS leader tags statin-ezetimibe cancer link as most likely a matter of chance
MUNICH-Final results from the Simvastatin and Ezetimibe in Aortic Stenosis (SEAS) trial were released at the 2008 European Society of Cardiology Congress, and the lead researcher urged oncologists to continue treatment for their patients who are also on cholesterol-lowering drugs.
MUNICH-Final results from the Simvastatin and Ezetimibe in Aortic Stenosis (SEAS) trial were released at the 2008 European Society of Cardiology Congress, and the lead researcher urged oncologists to continue treatment for their patients who are also on cholesterol-lowering drugs.
In 2001, Terje Pedersen, MD, PhD, and his colleagues began enrolling 1,873 patients with aortic stenosis. The trial was designed to evaluate the risk of cardiovascular events and cardiovascular mortality among patients taking the combination lipid-lowering regimen.
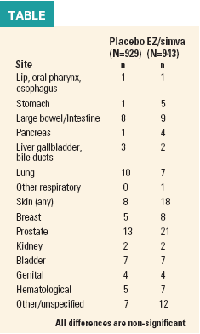
But the group unexpectedly found that the combination therapy-when compared with placebo-seemed to increase cancer risk and mortality. During a median follow- up of 52 months, there was an increase in both the incidence of cancer (101 vs 65 patients; hazard ratio, 1.55) and cancerrelated deaths [37 vs 20 deaths; hazard ratio, 1.78 (see Table)].
“It was a totally unexpected finding-and one that we think is actually quite implausible,” Dr. Pedersen, professor of medicine in the Center for Preventive Medicine at Ulleval University Hospital in Norway, told Oncology News International. “It’s difficult to believe that patients will develop a cancer within a span of four years that will lead to death-merely by taking cholesterol-lowering pills.”
Last month, U.S. Food and Drug Administration announced that it would continue to monitor a possible increased cancer risk in association with the use of simvastin (Zocor) and ezetimibe (Zetia), which are marketed in combination as Vytorin. The agency also said that it was awaiting results from the SEAS trial that would take six months to review (see “Common cholesterol drugs may be linked to escalated cancer risk,” September 2008, page 6).
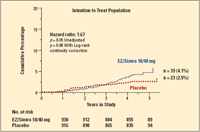
Dr. Pedersen noted that the cancer-related findings of the SEAS trial are based on small numbers and could be due to chance. He also noted that the diff erences between the treatment and placebo groups did not relate to any particular type of cancer and did not become larger with more prolonged treatment (see Figure).
However, the data have been submitted to an independent academic group for analysis, along with results from two other clinical trials that analyzed cholesterol-lowering drugs, SHARP (Study of Heart and Renal Protection) and IMPROVE-IT (Improved Reduction of Outcomes: Vytorin Effi cacy International Trial).
SEAS results have been published online in The New England Journal of Medicine (September 2, 2008). In an accompanying NEJM editorial, Thomas R. Fleming, PhD, noted that interim analysis from IMPROVE-IT and SHARP did indicate an increase in the number of cancer-related deaths associated with active vs control therapy-and a 34% increased risk of cancer-related mortality among those taking simvastatin-ezetimibe.
While Dr. Fleming noted that the interim nature of the data from the IMPROVE-IT and SHARP trials is problematic, he also called for more research to clarify the connection between simvasatin-ezetimibe and cancer, preferably through completed randomized trials.
“Such confirmation is especially important in the case of agents such as ezetimibe, for which there are safety signals of major illness or death and evidence of efficacy that is limited to documented eff ects on a biomarker,” Dr. Fleming said.
Dr. Pedersen said he believed that the cancer- related findings in the SEAS, IMPROVEIT, and SHARP trials can be explained by chance, and that cholesterol-lowering drugs do not pose an increased cancer risk.
“I’m not about to recommend that any of my patients stop taking ezetimibe based on these findings,” Dr. Pedersen said.
He added that oncologists who face concern from their patients about cholesterollowering drugs and cancer risk should not stop treatment but should explain the results from the various trials.
2 Commerce Drive
Cranbury, NJ 08512
All rights reserved.