A Comparison of FDA and EMA Drug Approval: Implications for Drug Development and Cost of Care
The development and approval of oncology therapeutics has been facilitated by the cooperation and coordination of regulatory practices between the EMA and the FDA. However, there continue to be important differences between the decisions of the two agencies.
Lynn J. Howie, MD, MA

Bradford R. Hirsch, MD, MBA

Amy P. Abernethy, MD, PhD

Table 1: Brief Timeline of Harmonization Efforts Between the FDA and the EMA
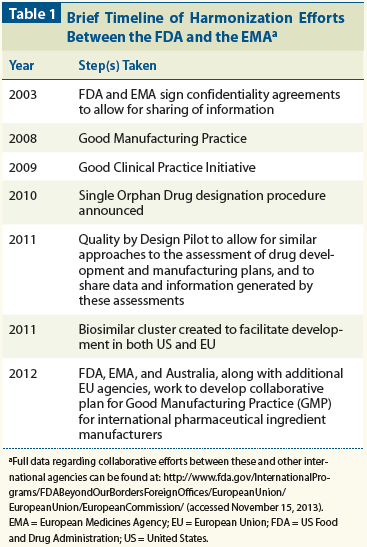
Table 2: Comparison of FDA and EMA Approval Dates for Selected Agents Approved by the FDA in 2012
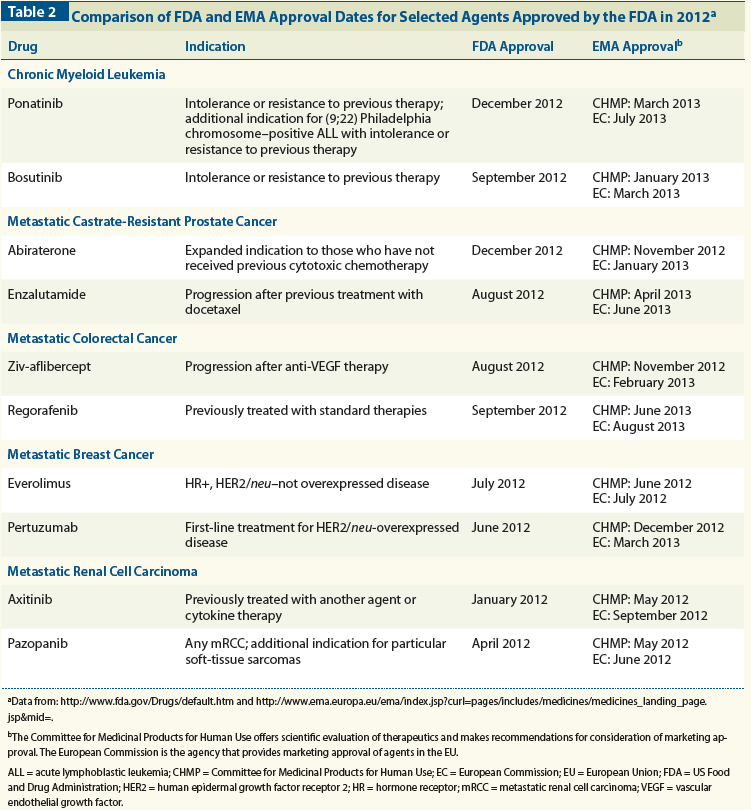
Table 3: Examples of Drug Labeling Differences for Oncology Therapeutics Approved in 2012
The cost of cancer care has been steadily increasing and is untenable. The medical portion of cancer care was estimated to cost $99 billion in the United States (US) and €51 billion in the European Union (EU) in 2009.[1,2] The high cost of medical cancer treatment is due, in part, to the significant costs of bringing new therapeutics to market. In order to reduce the costs of development and facilitate approval of meaningful treatments, efforts have been made to align and streamline the regulatory environment within which drug efficacy and safety are evaluated. This is crucial for the introduction of more affordable new therapeutics, particularly in areas such as oncology, where there have been significant advances in the understanding of tumor biology and molecular targets. Staggering drug prices negatively impact both patient care and overall healthcare costs.
Over the past decade, there have been efforts to harmonize the regulatory processes of the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA) (Table 1). Here we discuss the similarities and differences between these two agencies in several key areas of concern for oncology drug development:
• The time to drug approval.
• The wording of drug indications.
• The development of follow-on biologic agents (biosimilars).
• The use of patient-reported outcomes in the approval process.
A better understanding of the regulatory environments in the US and the EU, as well as efforts to align the approaches of their respective drug approval agencies, can guide resource allocation, facilitate advances in cancer care, and ultimately optimize patient care. Hopefully such actions will also lower drug costs.
Time to Drug Approval and Implications for Marketing and Prescribing
There is concern that regulation hampers drug development and slows approvals beyond what is required, because of costs incurred by pharmaceutical companies in their efforts to meet clinical research requirements that may be excessive and duplicative.[3] Accelerated approval pathways advanced by both the FDA and the EMA are among the reform efforts frequently taken advantage of by pharmaceutical companies’ oncology drug development programs.[4] The goal of the accelerated pathways is to shorten the time from application to approval for therapies that treat diseases whose morbidity and mortality are significant, and for which there are few effective alternatives. Section 901 of the Safety Innovations Act (FDASIA) of 2012 permits use of surrogate endpoints such as progression-free survival (PFS) rather than overall survival (OS) within the accelerated pathway. This allows a demonstration of efficacy within a shorter period of time.[5] For agents to be considered for accelerated approval, compelling clinical trials must meet the surrogate endpoint, and following approval, confirmatory phase IV trial data must be provided to verify the clinical benefit.[5] For example, regorafenib was approved by the FDA for the treatment of metastatic colorectal cancer just 3 months after it became eligible for accelerated approval.[6]
Despite similarities in the pathways and assessments used for accelerated approval, there are differences between the two agencies in time to approval and in the marketing of therapeutics. Some have argued that the EMA’s processes allow for quicker approval than those of the FDA,[7] but closer review demonstrates that the FDA typically approves drugs more quickly than the EMA-both new medicines generally and cancer therapeutics specifically.[8,9] This demonstrates a key difference between the authority of the two agencies. Dates on which the two agencies approved a sample of agents used in common cancer diagnoses are found in Table 2. While the FDA both scientifically evaluates new agents and issues marketing approval decisions for therapeutics, the scope of the EMA’s Committee for Medicinal Products for Human Use (CHMP) is limited to the scientific evaluation of therapeutics. Based on the CHMP assessment, the EMA makes recommendations to the European Commission (EC) for marketing approval of the drug. This decoupling of the scientific approval process and the marketing approval process leads to additional delays. Because patients do not have access to the treatment, and pharmaceutical companies cannot realize revenues until the drug can be marketed and prescribed, there are practical patient care and financial differences between the two approaches.
Some of the differences between the US and the EU in the actual use of drugs arise because each EU member state has its own system for determining which therapeutics will be approved by that member state. For example, in the United Kingdom (UK), a new therapeutic can apply for a license through the Medicines and Healthcare products Regulatory Agency (MHRA), or go through the EMA. If the agent is approved by the EMA, it then must be approved by the MHRA in order to be marketed in the UK. In addition, the National Institute for Health and Care Excellence (NICE) must then evaluate the agent for both efficacy and cost concerns in order to determine whether the agent that has been approved for sale by the MHRA will actually be purchased by the National Health Service (NHS).[10,11]
Furthermore, there have been differences between the US and the EU with regard to the labeling language that describes therapeutic indications. A study comparing the approval sequence and label wording of the FDA and EMA demonstrated that approximately half of all drug approval decisions and label wordings were the same. However, 20% of drugs were approved by one agency and not the other, and 28% of drug approvals had variations in the wording of the label, with the second agency to approve an agent being more likely to have a restrictive indication.[12] Several examples of these discrepancies are found in Table 3.
While significant steps have been taken to harmonize and align regulatory practices for drug approval and marketing, there remain differences in decisions made by the FDA and the EMA that may result in differences in patient access to cancer therapies. However, differences between agencies with regard to time to approval appear to be shortening, and alignment is increasing as a result of these ongoing and deliberate efforts.[12]
Biosimilars: Different Approaches on the Two Sides of the Atlantic
The development of follow-on biologic agents (biosimilars) is an area where the FDA and the EMA have differing track records. Biosimilars promise cost savings and expanded access to biologic therapies, somewhat analogous to the advantages offered by generic drugs.[13] However, because the development and manufacturing of biologic agents are very different from those for small-molecule agents, the standards put forth in legislation for generic drug development are inadequate for biosimilars; revised approaches must accommodate the complexities of developing biologic drugs that rely on living microorganism, plant, or animal cells for production.[14] The EU first identified and defined a regulatory strategy for biosimilars in 2004. Since that time, multiple biosimilars have become available in the EU, including cancer supportive care agents, such as erythropoietin analogues and granulocyte colony-stimulating factors.[15,16]
The Biologics Price Competition and Innovation Act of 2009, enacted under the 2010 Patient Protection and Affordable Care Act (ACA), was the first step in the development of a biosimilar regulatory process in the US.[17] This act delineated an accelerated approval approach for biologic products deemed to be interchangeable with their predecessor biologic agent. The act defines biosimilars as agents that demonstrate no significant differences in safety, efficacy, or potency compared with the original agent. It goes on to identify a standard of interchangeability according to which the biosimilar is expected to produce the same clinical response as the reference product.
The approval of tbo-filgrastim (Teva Pharmaceuticals) may serve as a test case for determining the US market’s acceptance of biosimilars. Tbo-filgrastim was approved by the EMA in 2008 under its biosimilar pathway; however, it was subsequently approved in the US under a full Biologics License Application in August 2012, since the biosimilar approval pathway was not yet finalized. Tbo-filgrastim did not enter the US market until November 10, 2013, due to a lawsuit and subsequent settlement between Teva and Amgen (the manufacturer of filgrastim and pegfilgrastim) over claims of patent infringement.[18] Tbo-filgrastim does not carry biosimilar approval in the US; however, it has demonstrated noninferiority to filgrastim in multiple clinical trials, with similar adverse effects.[19] While Teva has not been able to make a claim of interchangeability at this point, use of this agent by US clinicians and healthcare payers may serve as a test case of biosimilar uptake in the future.
The impact of biosimilars on the cost of cancer care will not be as dramatic as that of the switch to generic agents. Because of the high development costs of biosimilars, it is anticipated that they will be marketed at a 20% to 30% discount off the price of the innovator product. For example, the dose price of tbo-filgrastim is approximately 30% lower than that of filgrastim (~$300/dose for filgrastim vs ~$210/dose for tbo-filgrastim).[20] There is concern that biosimilars will not achieve market share, since innovators will likely lower their prices to remain competitive and thereby undermine the business case for developing competing agents. The key will be the complexity of the regulatory processes, since the more costly the approach, the smaller the pricing differential and the less incentive there is to enter the market. Safety and efficacy remain paramount. Included in the ACA legislation are reimbursement incentives to switch to biosimilars; these may compel physician prescribing to favor these agents, since their use would reduce the risk of significant loss of revenue.[21]
Patient-Reported Outcomes
Both the EMA and the FDA recognize the value of patient-reported outcomes (PROs) as important patient-centered endpoints when determining the efficacy of therapies and considering them for approval. The EMA began drafting recommendations regarding the use of PROs in 2004-for the Efficacy Working Party of the CHMP; these recommendations were adopted in June 2005. The FDA followed with draft guidance in 2006. Both these documents highlight the importance of PROs in considerations of therapeutic efficacy, but they are divergent in their approach.
The initial documents from the EMA indicated that the agency’s primary concern was to acknowledge the importance of improvement in patient-reported health-related quality of life (HRQOL).[22] The PRO guidance from the EMA centers on multiple domains for generalized assessment of HRQOL, while the FDA focuses on symptom-specific measures. The approval documents for pazopanib demonstrate this difference. While the EMA incorporates the HRQOL data from the phase III trials of pazopanib in their assessment, there is no mention of this endpoint in the FDA document.[23,24] The FDA is also more explicit in its requirements for the development and use of PROs as an outcome in a clinical trial.[25] This trend toward more specific requirements has intensified, with recent documents from the FDA favoring symptom-specific assessments over global HRQOL assessments.[26] Moreover, the FDA usually sees PROs as a supplemental endpoint for the demonstration of clinical efficacy rather than as part of a primary assessment.
In general, the FDA’s guidelines regarding PRO development and validation are more stringent than those of the EMA, although both agencies note the role and value of these measures in bringing the patient’s perspective to the assessment of therapeutics. The November 2011 approval of ruxolitinib heralded an important transition for the FDA.[27] Ruxolitinib was the first drug to get symptom data on its approved label since the FDA first issued its draft PRO guidance in 2006; successful approval required early planning regarding the PRO component of the drug development plan, including working with patients to identify the most important and bothersome symptom concerns (eg, night sweats, itch, abdominal pain), and collaboration with the FDA on the development and administration of a new PRO instrument designed specifically for this drug development program (the modified Myelofibrosis Symptom Assessment Form v2.0, administered daily using an electronic interface). It will be interesting to see whether there is more harmonization between EMA and FDA expectations around PROs over time.
Discussion
The development and approval of oncology therapeutics has been facilitated by the cooperation and coordination of regulatory practices between the EMA and the FDA. This cooperation has resulted in harmonized practices and requirements that have helped decrease regulatory burden and made it possible for drug development to occur with fewer obstacles. However, there continue to be important differences between the decisions of the two agencies, particularly with regard to decision-making timing and outcomes. Also, despite their having similar procedures and processes, there remain significant cultural and economic differences between the US and the EU-and within the EU, between its various member states.[28]
Efforts to align practices, regardless of approval results, can lead to decreased costs associated with drug development and can provide a more streamlined and predictable process for evaluation of the efficacy and safety of new therapeutics. Where procedural differences occur, we can learn which factors lead to the best outcomes for patients with regard to treatment efficacy and minimization of adverse effects. Ultimately, using mutually agreed upon processes will lower barriers to drug development and eliminate redundant processes that affect both the availability of safe and effective therapeutics to patients and the manageability of the cost of these therapeutics to the populations served.
Financial Disclosure:Dr. Abernethy has been involved in industry-funded research for Alexion, Amgen, Biovex, Celgene, DARA, Dendreon, Helsinn, MiCo, and Pfizer; she has pending industry-funded research involvements with Bristol-Myers Squibb, Genentech, GlaxoSmithKline, Insys, and Kanglaite. Dr. Abernethy serves as a consultant to Bristol-Myers Squibb, Novartis, and Pfizer; she serves on the corporate board of directors of Advoset, AthenaHealth, and Orange Leaf Associates, LLC; she is president of the American Academy of Hospice and Palliative Medicine; and she has a pending appointment as co-chair of the scientific advisory committee of Bristol-Myers Squibb. Drs. Howie and Hirsch have no significant financial interest in or other relationship with the manufacturer of any product or provider of any service mentioned in this article.
[[{"type":"media","view_mode":"media_crop","fid":"17952","attributes":{"alt":"","class":"media-image media-image-left","id":"media_crop_8529149987953","media_crop_h":"0","media_crop_image_style":"-1","media_crop_instance":"1178","media_crop_rotate":"0","media_crop_scale_h":"0","media_crop_scale_w":"0","media_crop_w":"0","media_crop_x":"0","media_crop_y":"0","style":"float: left;","title":" ","typeof":"foaf:Image"}}]]The content of ONCOLOGY’s Practice & Policy department represents a joint venture between the editors of ONCOLOGY and the Community Oncology Alliance. Articles featured in Practice & Policy are supplied by the COA as part of its mission to educate oncologists about the policy issues that affect the nation's cancer care delivery system. Practice & Policy features reflect the views of their authors and not necessarily those of the COA.
References:
1. Mariotto AB, Yabroff KR, Shao Y, et al. Projections of the cost of cancer care in the United States: 2010-2020. J Natl Cancer Inst. 2011;103:117-28. Erratum in: J Natl Cancer Inst. 2011;103:699.
2. Luengo-Fernandez R, Leal J, Gray A, Sullivan R. Economic burden of cancer across the European Union: a population-based cost analysis. Lancet Oncol. 2013;14:1165-74.
3. The FDA and slower cures. Wall Street Journal. February 28, 2011.
4. FDA launches 7 initiatives with European drug regulators. Available from: http://www.cancernetwork.com/articles/fda-launches-7-initiatives-european-drug-regulators. Accessed November 14, 2013.
5. Fast track, breakthrough therapy, accelerated approval and priority review. Available from: http://www.fda.gov/forconsumers/byaudience/forpatientadvocates/speedingaccesstoimportantnewtherapies/ucm128291.htm#accelerated. Accessed November 15, 2013.
6. Regorafenib. Available from: http://www.fiercebiotech.com/tags/regorafenib. Accessed November 14, 2013.
7. Zakaria F. Finding a strategy for growth. Washington Post. January 24, 2011.
8. Roberts SA, Allen JD, Sigal EV. Despite criticism of the FDA review process, new cancer drugs reach patients sooner in the united states than in Europe. Health Aff (Millwood). 2011;7:1375-81.
9. Downing NS, Aminawung JA, Shah ND. Regulatory review of novel therapeutics-comparison of three regulatory agencies. N Engl J Med. 2012;366:2284-93.
10. NICE and the NHS: where NICE guidance applies. Available from: http://www.nice.org.uk/aboutnice/whatwedo/niceandthenhs/nice_and_the_nhs.jsp. Accessed November 15, 2013.
11. How we regulate: what principles inform the decisions. Available from: http://www.mhra.gov.uk/Howweregulate/Whatprinciplesinformthedecisions/. Accessed November 15, 2013.
12. Trotta F, Leufkens HG, Schellens JH, et al. Evaluation of oncology drugs at the European Medicines Agency and US Food and Drug Administration: when differences have an impact on clinical practice. J Clin Oncol. 2011;29:2266-72.
13. Hirsch BR, Lyman GH. Will biosimilars gain momentum? J Natl Compr Canc Netw. 2013;11:1291-7.
14. Guidance for industry. Biosimilars: questions and answers regarding the implementation of the Biologics Price Competition and Innovation Act of 2009. Available from: http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatory Information/Guidances/UCM273001.pdf. Accessed November 15, 2013.
15. Gascon P. Presently available biosimilars in hematology-oncology: G-CSF. Target Oncol. 2012;7(suppl 1):S29-S34.
16. Aapro M, Cornes P, Sun D, Abraham I. Comparative cost efficiency across the European G5 countries of originators and a biosimilar erythropoiesis-stimulating agent to manage chemotherapy-induced anemia in patients with cancer. Ther Adv Med Oncol. 2012;4:95-105.
17. Development and approval processes: biologics. Available from: http:\\www.fda.gov/Drugs/DevelopmentApprovalProcess/HowDrugsare DevelopedandApproved/ApprovalApplications/TherapeuticBiologicApplications/Biosimilars/default.htm. Accessed November 15, 2013.
18. Teva announces FDA grants approval for tbo-filgrastim for the treatment of chemotherapy-induced neutropenia. Available from: http://www.tevapharm.com/Media/News/Pages/2012/1730014.aspx. Accessed November 16, 2013.
19. Crawford J, Armitage J, Balducci L, et al. Myeloid growth factors. J Natl Compr Canc Netw. 2013;11:1266-90.
20. Tbo-Filgrastim. Presented by Teva Pharmaceuticals at the AJMC Patient Centered Oncology Care Conference; November 14, 2013; Baltimore.
21. Megerlin F, Lopert R, Taymor K, Trouvin JH. Biosimilars and the European experience: implications for the United States. Health Aff (Millwood). 2013;32:1803-10.
22. Reflection paper on the regulatory guidance for the use of health-related quality of life (HRQL) measures in the evaluation of medicinal products. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2009/09/WC500003637.pdf. Accessed November 20, 2013.
23. Approved drugs: pazopanib. Available from: http://www.fda.gov/drugs/informationondrugs/approveddrugs/ucm302090.htm. Accessed November 16, 2013.
24. Pazopanib: EPAR product information. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_Product_Information/human/001141/WC500094272.pdf. Accessed November 16, 2013.
25. Bottomley A, Jones D, Claasens L. Patient-reported outcomes: assessment and current perspectives of the guidelines of the Food and Drug Administration and the reflection paper of the European Medicines Agency. Eur J Cancer. 2009;45:347-53.
26. Sutter S. Patient-reported outcomes tools should be symptom specific, FDA says. The Pink Sheet. November 11, 2013.
27. Moran N. Incyte comes of age with JAK inhibitor approval. Nature Biotechnol. 2012;30:3-5.
28. Pignatti F. Implementing a national cancer clinical trials system for the 21st century: workshop #2, Institute of Medicine. February 11, 2013. [video] Available from: http://www.iom.edu/Activities/Disease/NCPF/2013-FEB-11/Day%202/Session%206/34-Pignatti.aspx. Accessed November 20, 2013.