Secondary Pure Red Cell Aplasia During Daratumumab/ Hyaluronidase Therapy for Multiple Myeloma
Investigators discussed the potential link between secondary pure red cell aplasia and subcutaneous daratumumab/hyaluronidase formulation for multiple myeloma.
ABSTRACT
Predominantly autoimmune in origin, severe normochromic, normocytic anemia with reticulocytopenia in the setting of the normal production of leukocytes and megakaryocytic lineages is known as pure red cell aplasia (PRCA), which is unlike aplastic anemia in which all lineages are affected due to a stem cell defect. PRCA can be primary (such as autoimmune) or acquired, which can be an acute self-limited illness or a chronic disease that may be induced by medications, including immunotherapy such as monoclonal antibodies (mAbs). Daratumumab is a mAb directed against CD38 used for the treatment of multiple myeloma and systemic amyloid light-chain amyloidosis. The intravenous formulation of daratumumab received initial FDA approval, and later approval was received for the subcutaneous formulation daratumumab and hyaluronidase-fihj. The subcutaneous version increases patient convenience and has become the preferred route of administration since its approval. We herein present the case of a patient with multiple myeloma who developed acquired DNMT3A-positive PRCA while transitioning to daratumumab/hyaluronidase after initial treatment with daratumumab.
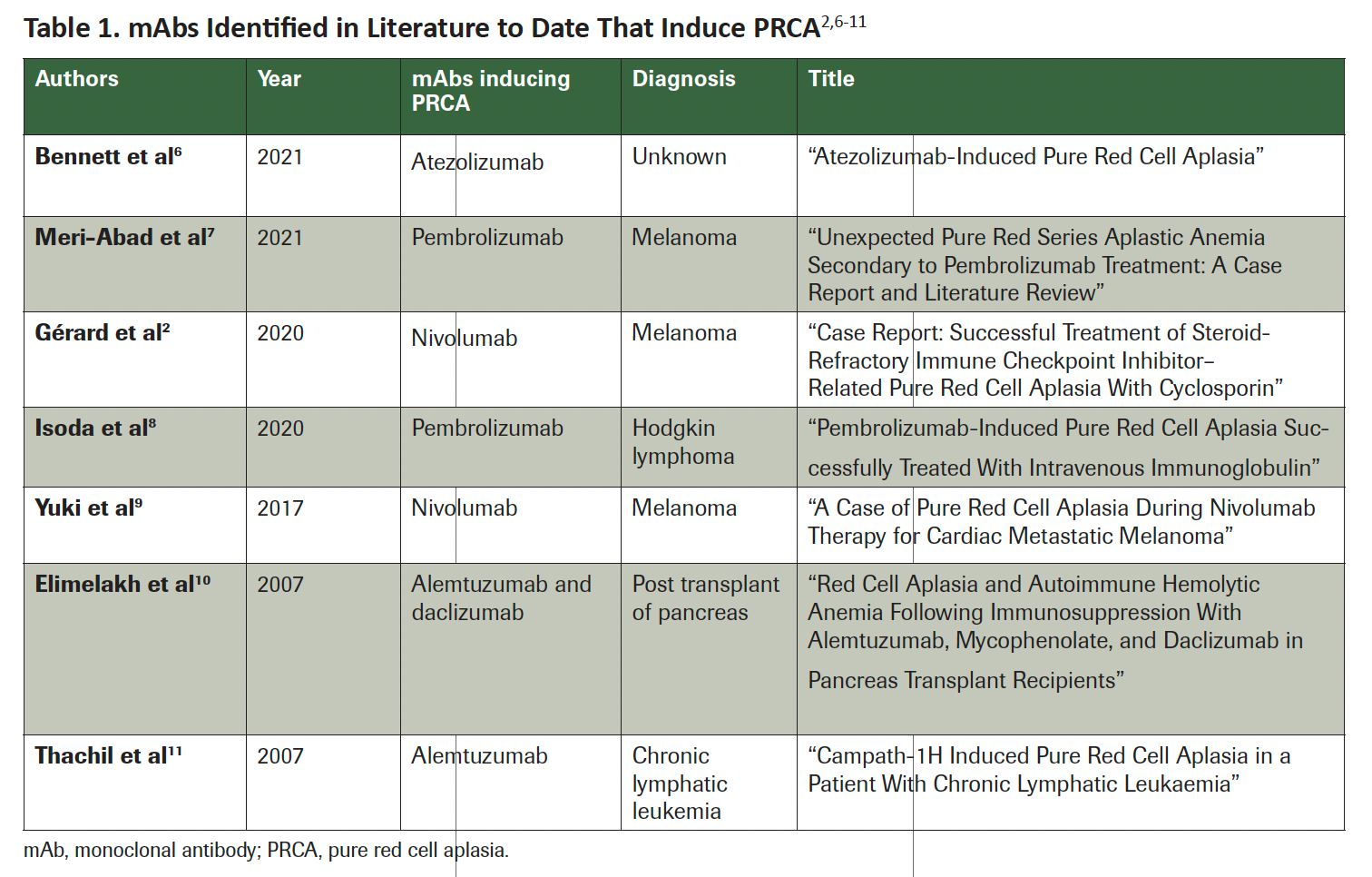
Table 1. mAbs Identified in Literature to Date That Induce PRCA2,6-11
Anemia can take many forms, and like other diseases, the acquired or secondary cases can be induced by medications, including immunotherapy with monoclonal antibodies (mAbs). Pure red cell aplasia (PRCA) is a type of anemia that is predominantly autoimmune in origin; it is characterized by the presence of severe normochromic, normocytic anemia with reticulocytopenia (< 1%) and marked reduction of erythroblasts in the bone marrow (< 5%) in the setting of normal production of leukocytic and megakaryocytic lineages.1 PRCA can be primary or secondary. It can sometimes be a transient self-limited illness or a chronic disease. Gérard et al reported a case of PRCA associated with nivolumab use for the treatment of metastatic melanoma.2 PRCA shares myeloid neoplasm–associated gene mutations, such as the DNMT3A mutation that has been described as a driver for PRCA in patients with myeloid malignancies.3 Located on chromosome 2p23.3, DNMT3A encodes DNA methyltransferase 3 alpha and is believed to be involved in de novo methylation. Additionally, DNMT3A is the most frequently mutated gene involved in clonal hematopoiesis of indeterminate potential, followed by mutations in TET2 and ASXL1. It is not yet known whether DNMT3A mutation plays a role in the development of PRCA. This mutation could be an incidental finding, especially in the older population.4
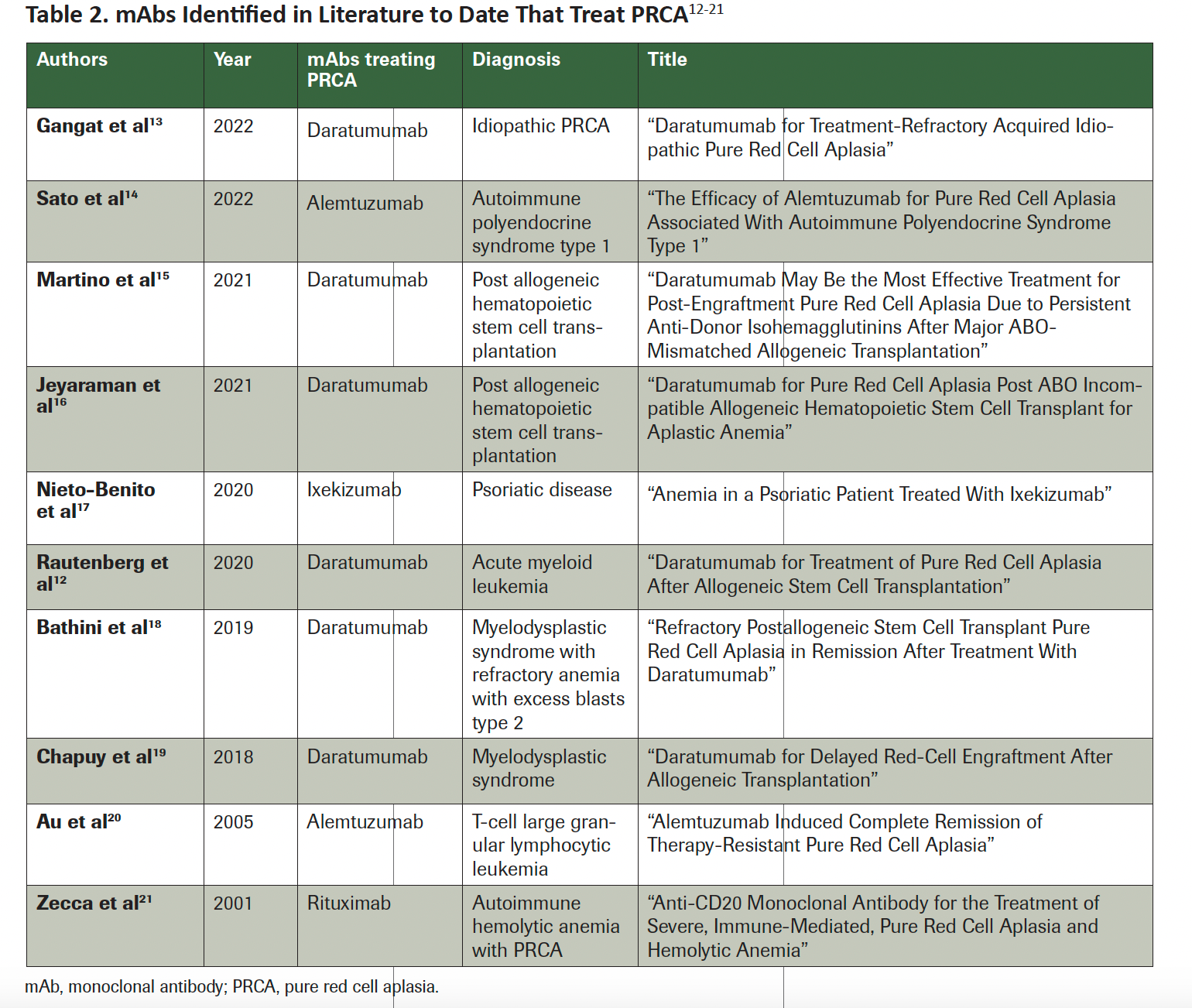
Table 2. mAbs Identified in Literature to Date That Treat PRCA12-21
CD38 is highly expressed on myeloma cells but has a relatively low expression on normal immune cells, making it an attractive target for treatment in patients with multiple myeloma (MM). Daratumumab is a humanized IgGκ mAb targeting a unique epitope of CD38 and has been approved in patients with MM and amyloid light-chain amyloidosis.5 Due to reports of PRCA acquired from the use of other types of mAbs (Table 12,6-11), cases of PRCA occurring around the time of daratumumab use should be investigated as a potential etiology. Conversely, daratumumab has been shown to be the most effective treatment for PRCA after allogeneic stem cell transplant–induced PRCA (Table 212-21).6 In this manuscript, we are discussing an older patient who developed PRCA during daratumumab-based treatment for MM.
Case Presentation
A case presentation of an 83-year old female with a history of pernicious anemia.

Serum immunofixation showed IgGλ monoclonal gammopathy. Her β2-
microglobulin level was 2.3 mg/L (range, < 2.16). Bone marrow showed less than 5% plasma cells, translocation t(11;14), and hyperdiploidy, as well as a gene expression risk stratification profile score of 22. However, no lytic lesions were present. She was diagnosed with monoclonal gammopathy of undetermined significance and was followed expectantly.
Starting in April 2018, the patient had multiple fractures, including her thoracolumbar vertebra, ribs, and jaw, for which a pathologic fracture was deemed a possibility. Hemoglobin and calcium levels were 9.8 g/dL (range, 11.5-15.5) and 11.8 mg/dL (range, 8.4-10.2), respectively, and her serum creatinine level was normal. A bone marrow biopsy in September 2018 revealed 20% involvement by clonal plasma cells and again the translocation t(11;14) that was detected in 2013, supporting the diagnosis of active MM requiring therapy.
Antimyeloma therapy was initiated in September 2019 with intravenous (IV) daratumumab at 16 mg/kg weekly, lenalidomide orally at 25 mg for 21 days on and 7 days off, and dexamethasone orally 20 mg weekly (DRd). Cycle 1 of DRd therapy was complicated by diarrhea and pancytopenia—with a white blood cell (WBC) count of 1380 K/µL (range, 3.70-11), a hemoglobin level of 7.8 g/dL (range, 11.5-15.5), and an absolute neutrophil count of 480 K/µL (range, 1.45-7.50)—requiring red blood cell transfusion and use of filgrastim. Therapy was held for 2 weeks and reintroduced with cycle 2 and cycle 3 with lenalidomide at a lower dose of 10 mg.
In December 2019, despite thromboprophylaxis with aspirin given at 81 mg daily, she experienced an acute deep venous thrombosis of the right lower extremity and was started on anticoagulation with rivaroxaban. Lenalidomide was discontinued permanently in December 2019 due to anemia, neutropenia, diarrhea, and acute deep venous thrombosis, and therapy was continued with IV daratumumab and dexamethasone. By June 2020, she achieved a very good partial response after daratumumab plus bortezomib and dexamethasone (DVd) was added to the regimen with the goal of achieving complete hematologic response. DVd therapy was given every 2 weeks due to her frailty and the multiple adverse effects (AEs) she had with the DRd regimen.
She tolerated the DVd therapy very well, and by December 2020, she achieved complete response. In January 2021, it was decided to switch from IV daratumumab to the subcutaneous (SQ) daratumumab and hyaluronidase-fihj for patient convenience; the goal was to use the SQ formulation once every 4 weeks as maintenance therapy. At the time of the transition, her WBC count was 9630 K/µL with normal differential counts, her hemoglobin level was 11.1 g/dL, and her platelet count was 239,000 (range, 150-400 K/µL).
Two weeks after the transition to the SQ formulation, her hemoglobin level dropped to 9.3 g/dL, but her platelet and WBC counts remained normal. Four weeks from the time of transition, the patient required 2 units of blood after a reticulocyte count of less than 0.2% (range, 0.6%-2.6%) and a further drop in hemoglobin level to 7.4 g/dL.
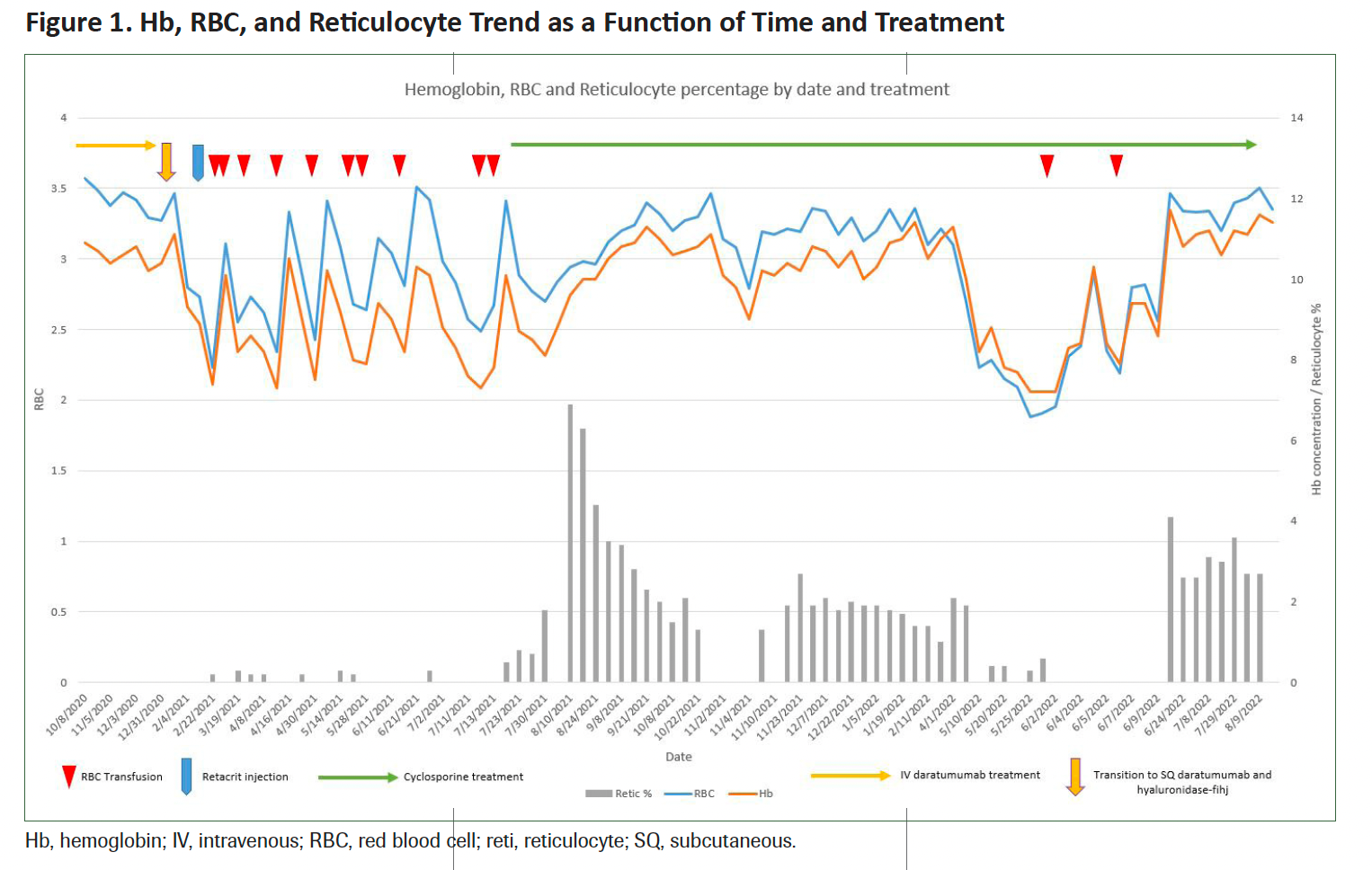
Figure 1 illustrates the trends in hemoglobin, red blood cell count, and reticulocyte levels over time, showcasing the impact of treatment. The levels of vitamin B12, iron, folate, zinc, and copper were normal. Daratumumab/hyaluronidase therapy was discontinued, and she was monitored without any further anti–plasma cell therapy.
Figure 1. Hb, RBC, and Reticulocyte Trend as a Function of Time and Treatment
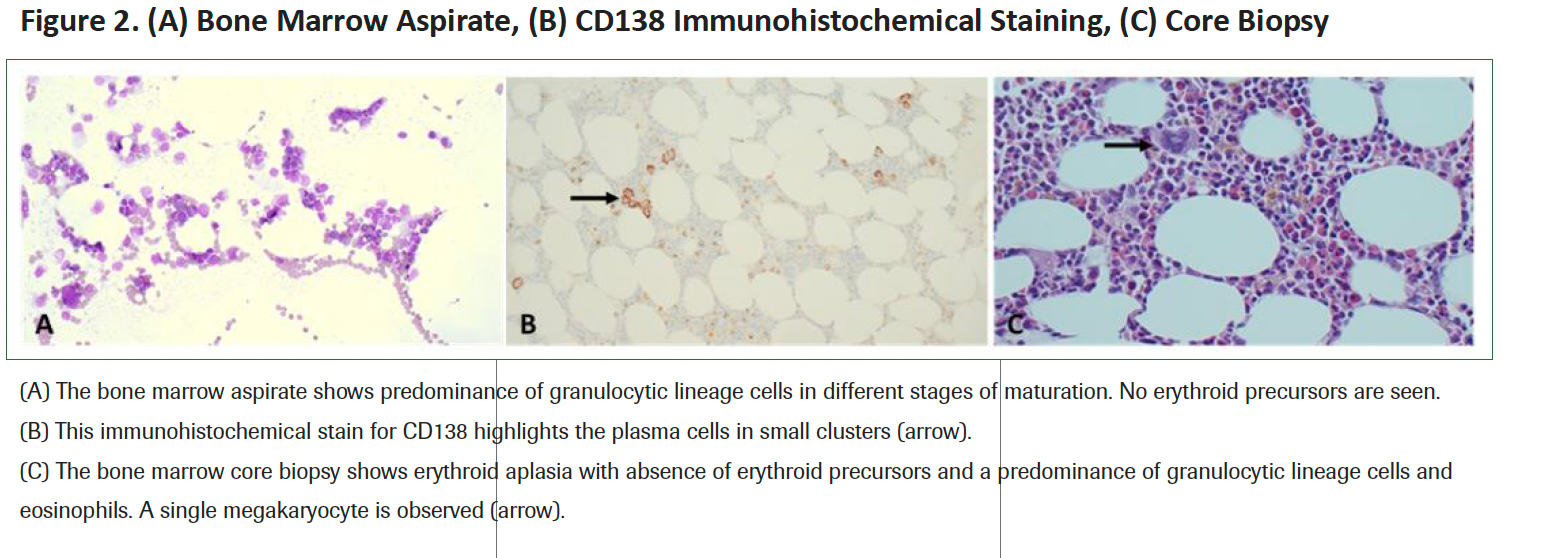
Parvovirus B19 DNA polymerase chain reaction test results were negative, and serology reported remote infection (normal immunoglobulin [IgM], elevated IgG). A bone marrow biopsy showed normal trilineage hematopoiesis, megakaryopoiesis, granulocytic maturation, and absent erythropoiesis consistent with PRCA (Figure 2). No clonal plasma cells were detected by flow cytometry, and immunohistochemistry reported rare CD38+ cells. A myeloid next-generation sequencing panel of 54 genes came back normal except for a clinically significant DNMT3A variant at splice acceptor site c.1475-1G>C.
Figure 2. (A) Bone Marrow Aspirate, (B) CD138 Immunohistochemical Staining, (C) Core Biopsy
A 60,000-unit dose of epoetin alfa injection was started weekly in May 2021. In June 2021, prednisone 1 mg/kg was added and given for 4 weeks without improvement in the patient’s hemoglobin level. Cyclosporine 100 mg twice daily was then added for the treatment of PRCA. With the dual immunosuppressive therapy of prednisone and cyclosporine, her hemoglobin level improved to 10 g/dL, making her transfusion independent by week 4. Prednisone was dose reduced by 50%, and cyclosporine continued at the same dose.
By January 2022, she remained transfusion-dependent on a reduced dose of prednisone 10 mg daily and cyclosporine 50 mg twice daily. By the end of January, the patient was instructed to reduce cyclosporine to 25 mg twice daily and to continue prednisone at 10 mg daily. In June 2022, the patient was admitted to the hospital for 10 days for new-onset atrial fibrillation with rapid ventricular response. At that time, the patient’s hemoglobin level had dropped to 7.2 g/dL. The patient had recurrent hypoproliferative anemia that required transfusion, consistent with recurrent PRCA. Hence, the cyclosporine dose was increased to 100 mg twice daily in addition to 60 mg of prednisone that was tapered to 40 mg over a 2-week period. Because her hemoglobin level rose to 11.1 g/dL in August 2022, prednisone was reduced to 20 mg and cyclosporine to 100 mg once daily, and she remained transfusion independent.
In early August 2022, the patient exhibited improvements in her anemia status. Nevertheless, despite her enhanced anemia condition and newfound independence from transfusions, she experienced a relapse later in the same month. A whole-body PET scan from September 2022 showed new fluorodeoxyglucose F18–avid myeloma lesions at T4 and the right first rib, indicative of relapsed myeloma. A restaging bone marrow aspiration/biopsy reported up to 15% λ light-chain restricted plasma, and a MM fluorescence in situ hybridization panel reconfirmed translocation t(11;14). She started venetoclax and dexamethasone in October 2022 and tolerated 1 cycle well. Unfortunately, 1 month later, the patient presented to the emergency department with acute abdominal pain. A CT scan showed pneumoperitoneum, highly suggestive of large-bowel perforation likely at the transverse colon from ischemic colitis. A subtotal colectomy with end ileostomy was performed. She was admitted to the intensive care unit for refractory septic shock that required support. Her course was complicated by multiorgan failure including shock liver and severe metabolic encephalopathy coupled with anoxic brain injury. These events led to the patient’s death.
Discussion
PRCA is an extremely rare condition that can be primary or secondary. It can be congenital, but most cases are acquired. The primary PRCA is an idiopathic autoimmune disorder in which an immune attack of erythroid precursor cells leads to underproduction anemia. Rarely, primary PRCA can be the initial presentation of a myelodysplastic syndrome.12 The patient described in this case report developed hypoproliferative anemia in the setting of DNMT3A positivity, which may have a few potential etiologies. Although PRCA can be present congenitally, such as in infancy cases of Diamond-Blackfan anemia, it may also be acquired, such as during parvovirus B19 infection. For the latter, the patient should test positive for parvovirus B19 DNA and/or IgM, which was not the case for our patient. PRCA cases have been reported in association with hepatitis A, B, and C; HIV; Epstein-Barr virus; cytomegalovirus; and SARS-CoV-2/COVID-19.3 All these serologies were negative in this patient.
Other associations of PRCA include immune disorders (autoimmune hemolytic anemia, systemic lupus erythematosus, rheumatoid arthritis, and ABO-incompatible hematopoietic stem cell transplant), hematologic neoplasms (chronic lymphocytic leukemia, large granular leukemia, Hodgkin and non-Hodgkin lymphoma, plasma cell neoplasms, chronic myeloid leukemia, primary myelofibrosis, and thymoma).
Finally, secondary PRCA can also be due to drugs (recombinant erythropoietin, mycophenolate, phenytoin, trimethoprim and sulfamethoxazole, and various mAbs) (Table 1). Drug-induced PRCA is a rare condition in which certain medications trigger immune responses, bone marrow suppression, alteration of growth factors, or autoimmune mechanisms that lead to a decrease or absence of red blood cell precursors in the bone marrow, resulting in anemia. These drugs can form immune complexes or directly interact with bone marrow cells, leading to immune-mediated destruction. Additionally, they can disrupt growth factors or trigger autoimmune reactions that target red blood cell precursors.22
The possibility of mAb-induced PRCA1 crossed our minds, prompting us to request a bone marrow aspiration and biopsy that confirmed the diagnosis.
The 7 cases outlined in Table 1 represent mAbs reported in literature that induce PRCA. At the time of writing, the literature reports no cases of SQ daratumumab/hyaluronidase causing PRCA. The exact mechanism by which SQ daratumumab and hyaluronidase-fihj might induce PRCA is not fully understood, but we suggest a few potential explanations. First, daratumumab targets CD38, which is expressed in various cell types, including immune cells.5 The immune response against CD38-expressing cells might inadvertently affect red blood cell precursors in the bone marrow, leading to PRCA. Another theory is immune-complex formation. Daratumumab could potentially form immune complexes with CD38 or other molecules in the bone marrow, leading to an immune response that damages red blood cell precursors. Lastly, the immune system—triggered by the daratumumab interaction with CD38-expressing cells—might mistakenly recognize red blood cell precursors as foreign due to their expression of CD38 or other related factors.23 This could result in an autoimmune reaction against these cells.
In contrast, the 10 cases outlined in Table 2 represent mAbs reported in the literature that treat PRCA. Interestingly, 6 of 10of the reported cases were treated with IV daratumumab, so it is with great interest that we further investigate why the patient presented here had induction of PRCA from daratumumab upon transitioning from the IV formulation to the SQ daratumumab/hyaluronidase formulation.
The transition from IV to SQ administration of daratumumab may lead to certain changes in drug exposure, pharmacokinetics, or immune interactions that could potentially contribute to the development of PRCA. The shift from IV to SQ administration might alter the interaction of daratumumab with immune cells, particularly those present in the SQ tissue. Changes in the immune cell activation or distribution could contribute to an immune response against red blood cell precursors. In addition, some patients might be more sensitive to changes in drug administration route or pharmacokinetics. Individual variability in immune responses also could contribute to the development of PRCA in response to the change in administration.24
It’s important to note that the development of PRCA after transitioning from IV to SQ daratumumab is rare. We suggest that patients undergoing this transition be closely monitored by their health care providers for any signs of AEs, including anemia or changes in blood counts. If PRCA or any other concerning AE is suspected, appropriate medical intervention and management should be pursued promptly.
Conclusion
PRCA is a primary or secondary disorder that can be induced by drugs, including mAbs. Several mAbs used for various disorders have been reported to cause PRCA. However, at the time of writing, there are no reports in the literature of PRCA induced by the SQ form of daratumumab in a patient with myeloma who tolerated IV daratumumab well. We hope this case report can raise awareness to providers that the SQ daratumumab/hyaluronidase formulation has a potential association with secondary PRCA in patients with plasma cell neoplasms.
Ludovic Saba, MD; and Kevin S. Landau, MD, contributed equally to the creation of this manuscript.
References
- Vlachaki E, Diamantidis MD Klonizakis P, Haralambidou-Vranitsa S, Ioannidou-Papagiannaki E, Klonizakis I. Pure red cell aplasia and lymphoproliferative disorders: an infrequent association. ScientificWorldJournal. 2012;2012:475313. doi:10.1100/2012/475313
- Gérard A, Romani S, Van-Obberghen E, et al. Case report: successful treatment of steroid-refractory immune checkpoint inhibitor-related pure red cell aplasia with cyclosporin. Front Oncol. 2020;10:1760. doi:10.3389/fonc.2020.01760
- Fujishima N, Kohmaru J, Koyota S, et al. Clonal hematopoiesis in adult pure red cell aplasia. Sci Rep. 2021;11(1):2253. doi:10.1038/s41598-021-81890-5
- Asada S, Kitamura T. Clonal hematopoiesis and associated diseases: a review of recent findings. Cancer Sci. 2021;112(10):3962-3971. doi:10.1111/cas.15094
- Lokhorst HM, Plesner T, Laubach JP, et al. Targeting CD38 with daratumumab monotherapy in multiple myeloma. N Engl J Med. 2015;373(13):1207-1219. doi:10.1056/NEJMoa1506348
- Rautenberg C, Kaivers J, Germing U, et al. Daratumumab for treatment of pure red cell aplasia after allogeneic stem cell transplantation. Bone Marrow Transplant. 2020;55(6):1191-1193. doi:10.1038/s41409-019-0664-4
- Thompson DF, Gales MA. Drug-induced pure red cell aplasia. Pharmacotherapy. 1996;16(6):1002-1008.
- Bennett R, Ruskova A. Atezolizumab-induced pure red cell aplasia. Br J Haematol. 2021;193(1):10. doi:10.1111/bjh.17259
- Meri-Abad M, Cunquero Tomás AJ, Jaime AB. Unexpected pure red series aplastic anemia secondary to pembrolizumab treatment: a case report and literature review. Melanoma Res. 2021;31(2):186-189. doi:10.1097/CMR.0000000000000724
- Isoda A, Miyazawa Y, Tahara K, et al. Pembrolizumab-induced pure red cell aplasia successfully treated with intravenous immunoglobulin. Intern Med. 2020;59(16):2041-2045. doi:10.2169/internalmedicine.4467-20
- Yuki A, Takenouchi T, Takatsuka S, Ishiguro T. A case of pure red cell aplasia during nivolumab therapy for cardiac metastatic melanoma. Melanoma Res. 2017;27(6):635-637. doi:10.1097/CMR.0000000000000392
- Elimelakh M, Dayton V, Park KS, et al. Red cell aplasia and autoimmune hemolytic anemia following immunosuppression with alemtuzumab, mycophenolate, and daclizumab in pancreas transplant recipients. Haematologica. 2007;92(8):1029-1036. doi:10.3324/haematol.10733
- Thachil J, Salim R. Campath-1H induced pure red cell aplasia in a patient with chronic lymphatic leukaemia. Leuk Res. 2007;31(7):1025-1026. doi:10.1016/j.leukres.2006.08.012
- van de Donk NWCJ, Richardson PG, Malavasi F. CD38 antibodies in multiple myeloma: back to the future. Blood. 2018;131(1):13-29. doi:10.1182/blood-2017-06-740944
- Gangat N, Bleeker J, Lynch D, Olteanu H, Letendre L, Tefferi A. Daratumumab for treatment-refractory acquired idiopathic pure red cell aplasia. Haematologica. 2022;107(10):2523-2526. doi:10.3324/haematol.2022.281398
- Sato M, Shino M, Yokoyama K, Ishida T, Hirao M, Kamoda Y, et al. The efficacy of alemtuzumab for pure red cell aplasia associated with autoimmune polyendocrine syndrome type 1. Article in Japanese. Rinsho Ketsueki. 2022;63(3):189-193. doi:10.11406/rinketsu.63.
- Martino R, García-Cadenas I, Esquirol A. Daratumumab may be the most effective treatment for post-engraftment pure red cell aplasia due to persistent anti-donor isohemagglutinins after major ABO-mismatched allogeneic transplantation. Bone Marrow Transplant. 2022;57(2):282-285. doi:10.1038/s41409-021-01507-3
- Jeyaraman P, Borah P, Rajput P, Dayal N, Pathak S, Naithani R. Daratumumab for pure red cell aplasia post ABO incompatible allogeneic hematopoietic stem cell transplant for aplastic anemia. Blood Cells Mol Dis. 2021;88:102464. doi:10.1016/j.bcmd.2020.102464
- Nieto-Benito LM, Mateos-Mayo A, Romero-Jiménez R, Baniandrés-Rodríguez O. Anemia in a psoriatic patient treated with ixekizumab. J Dermatol. 2020;47(5):e209-e210. doi:10.1111/1346-8138.15288
- Bathini S, Holtzman NG, Koka R, et al. Refractory postallogeneic stem cell transplant pure red cell aplasia in remission after treatment with daratumumab. Am J Hematol. 2019;94(8):E216-E219. doi:10.1002/ajh.25515
- Chapuy CI, Kaufman RM, Alyea EP, Connors JM. Daratumumab for delayed red-cell engraftment after allogeneic transplantation. N Engl J Med. 2018;379(19):1846-1850. doi:10.1056/NEJMoa1807438
- Au WY, Lam CC, Chim CS, Pang AW, Kwong YL. Alemtuzumab induced complete remission of therapy-resistant pure red cell aplasia. Leuk Res. 2005;29(10):1213-1215. doi:10.1016/j.leukres.2005.02.018
- Zecca M, De Stefano P, Nobili B, Locatelli F. Anti-CD20 monoclonal antibody for the treatment of severe, immune-mediated, pure red cell aplasia and hemolytic anemia. Blood. 2001;97(12):3995-3997. doi:10.1182/blood.v97.12.3995
Navigating AE Management for Cellular Therapy Across Hematologic Cancers
A panel of clinical pharmacists discussed strategies for mitigating toxicities across different multiple myeloma, lymphoma, and leukemia populations.
