Successful Treatment of Melanoma Metastatic to the Left Atrium Using External Beam Radiation Therapy
The successful treatment of a patient with primary nasal melanoma metastatic to the lung, pulmonary vein, and left atrium using radiation therapy is described. The patient was effectively treated with a conventional external beam radiation fractionation scheme (rather than a more commonly used hypofractioned regimen) that was utilized to minimize risk of arterial embolus of the tumor or rupture of a vessel wall. A post-treatment CT demonstrated a significant decrease in the caliber of the right pulmonary vein and tumor thrombus. The patient never developed cardiac valvular dysfunction or acute life-threatening massive embolism of tumor from the atrium. Unfortunately, the patient experienced clinical decline secondary to the massive progression of intra-abdominal disease and subsequently died from multiple liver metastases and liver failure. Numerous studies and this case report demonstrate that radiation therapy can be very effective in the treatment of malignant melanoma, especially when only small volumes of disease need to be treated and adequate total doses are used. Therefore, radiation therapy appears to play an important yet underutilized role in the treatment of metastatic melanomas.
The successful treatment of a patient with primary nasal melanoma metastatic to the lung, pulmonary vein, and left atrium using radiation therapy is described. The patient was effectively treated with a conventional external beam radiation fractionation scheme (rather than a more commonly used hypofractioned regimen) that was utilized to minimize risk of arterial embolus of the tumor or rupture of a vessel wall. A post-treatment CT demonstrated a significant decrease in the caliber of the right pulmonary vein and tumor thrombus. The patient never developed cardiac valvular dysfunction or acute life-threatening massive embolism of tumor from the atrium. Unfortunately, the patient experienced clinical decline secondary to the massive progression of intra-abdominal disease and subsequently died from multiple liver metastases and liver failure. Numerous studies and this case report demonstrate that radiation therapy can be very effective in the treatment of malignant melanoma, especially when only small volumes of disease need to be treated and adequate total doses are used. Therefore, radiation therapy appears to play an important yet underutilized role in the treatment of metastatic melanomas.
The incidence of melanoma has increased significantly in the past two decades, while mortality has remained stable or has decreased.[1,2] As the incidence of melanoma increases, so does the incentive to discover novel and more efficacious methods of treatment. Effective palliative therapies should not be overlooked, however. Delaney et al recently reported that 23% of melanoma patients will have an indication for radiotherapy during their illness.[3] Radiation therapy is applied to less than 3% of all cases, largely because of the long-held notion that melanomas are universally resistant to radiation therapy.[4]
Radiation therapy has a well-established role for patients with incurable, metastatic melanoma. The major indications include treatment of dermal, subcutaneous, lymph node, bone, and brain metastases and spinal cord compression. The effects of palliative radiation are often easily seen because only relatively slight tumor shrinkage is required for symptomatic relief. Symptoms of mass effect, pressure, and bleeding from metastases in various sites benefit from palliative radiation. Seegenschmiedt et al found median survival in stage IV melanoma treated with radiotherapy was only 7 months.[5] While cardiac extension into the right atrium of tumors that invade the vena cava, such as Wilms tumor and renal cell carcinoma, is fairly common, and metastatic melanoma to the pericardium or myocardium is occasionally seen, extension of any tumor from the pulmonary vein into the heart is extremely rare. We report on a patient with stage IV melanoma with a bulky right lung lesion extending along the pulmonary vein into the left atrium palliatively treated with external beam radiation therapy.
FIGURE 1
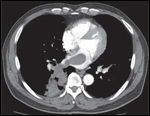
Computed tomography scan of patient’s chest when he was first diagnosed with intracardiac extension of disease.
Case Presentation
In 2005, a 63-year-old Filipino American male without previous disease or family history of cancer was diagnosed with a primary nasal cavity melanoma and underwent resection and postoperative radiation therapy that consisted of 6,600 cGy to the nasal cavity via a multifield 3D conformal technique completed in September 2005. The patient was asymptomatic until December 2007, when he contracted what was thought to be a lower respiratory infection that subsequently failed to respond to antibiotic therapy. In February 2008 the patient underwent a chest CT scan which demonstrated multiple pulmonary nodules, including a large right infrahilar mass in the right lower lobe that abutted the mediastinum and extended through the pulmonary vein into the left atrium. The tumor thrombus was approximately the diameter of the aortic arch and at least 4-5 cm in length, occupying a large portion of the left atrium (Figure 1).
Computed tomography (CT)-guided lung biopsy confirmed metastatic melanoma. Subsequent magnetic resonance imaging (MRI) showed multiple small (7 mm and 3 mm) enhancing lesions in the supratentorial and infratentorial brain consistent with metastatic disease. Review of systems revealed only cough of 2 months’ duration and intermittent hemoptysis. The patient denied any weight loss, decrease in energy, shortness of breath, or neurologic symptomatology. The only finding appreciated on physical examination was diminished breath sounds in the right lung base.
FIGURE 2
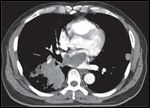
Computed tomography scan of chest 4 weeks after prior CT scan showing progression of intracardiac and pulmonary disease prior to treatment.
At that time it was believed that the malignancy in the patient’s pulmonary vein and left atrium would ultimately cause the patient to die before his asymptomatic brain metastases would become manifest. He was referred to a university medical center cardiothoracic surgery service, but was deemed to be inoperable and referred back to our service.
The lesion was found to be increasing in size and threatening the tricuspid valve (Figure 2). Palliative radiotherapy was offered owing to the inoperability of the tumor, and because systemic chemotherapy was not likely to yield a significant response. Palliative radiation therapy is typically given with hypofractionated regimens of 400 cGy × 5, 300 cGy × 10, or 250 cGy × 15 fractions to minimize patients’ time on treatment, given their limited survival time. In addition, because of the broad shoulder on the cell-survival curve, larger fraction sizes have been considered optimal for patients with malignant melanoma.
There also was concern about abrupt tumor death with larger fraction sizes resulting in an arterial embolus of tumor or rupture of vessel wall with severe acute sequelae in a patient who at that time had an excellent performance status. Therefore, a conventional external beam radiation fractionation scheme (180 cGy per fraction to 4500 cGy) was planned to minimize risk and achieve the palliative goal. A four-field box technique was used with 18 MV X-rays because of the central location of the tumor. Only the immediately perihilar and atrial volumes were targeted, to spare the normal lung and esophagus. No attempt was made to include the entire bulky mass in the more peripheral lung volume. Daily cone beam CT was used for localization.
FIGURE 3
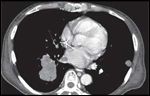
Computed tomography scan of patient’s chest 8 weeks after treatment, showing decrease in intracardiac and extracardiac disease.
The patient began treatment in March 2008 and tolerated the treatment remarkably well, experiencing only mild dysphagia and a slight cough. Interestingly, a repeat brain MRI scan prior to radiation therapy showed that several previously noted brain metastases had resolved spontaneously and other small new lesions were now seen. Also, during the course of therapy, the patient developed multiple small, tender, subcutaneous nodules on the trunk and extremities that would resolve spontaneously over several days. These were worrisome as they suggested episodes of tumor or thrombotic emboli, although no biopsies were performed. The patient was started on aspirin alone for thromboembolism prophylaxis, because full anticoagulation with Coumadin (warfarin) or low-molecular-weight heparin would place him at increased risk of intracranial hemorrhage. A CT scan performed in July 2008 after completion of treatment in early May demonstrated a significant decrease in the caliber of the right pulmonary vein and tumor thrombus (Figure 3).
The patient later developed definite progressive symptomatic lesions in the CNS consistent with metastatic disease. Palliative brain irradiation (3,250 Gy in 13 fractions) from late June 2008 to mid July 2008 was well tolerated. He never developed cardiac valvular dysfunction or acute life-threatening massive embolism of tumor from the atrium. Unfortunately, shortly after completion of brain irradiation, the patient experienced clinical decline secondary to massive progression of intra-abdominal disease. In late July 2008, he was admitted to the hospital for progressive abdominal pain and ascites; decline secondary to massive liver metastases caused his death in mid August 2008.
Discussion
In the 1930s, when the model of grading tumor radiosensitivity by histologic type obtained recognition, malignant melanoma was quickly labeled as radioresistant.[6] While early retrospective data proposed little responsiveness to radiation delivered at a low dose per fraction, it is now well documented that melanoma cells are radioresponsive irrespective of fractionation schedule if sufficient total doses of radiation are given.[7-9] Furthermore, recent radiobiological studies have shown great heterogeneity of malignant melanoma tumor cells, which may account for the extensive differences of radiosensitivity found in human xenografts.[10,11]
The spectrum of clinical responses and in vitro assays of cultured cells illustrates that melanomas have a wide range of sensitivities to radiation. Generally speaking, melanomas tend to be less sensitive to radiation than common epithelial carcinomas.[12] Nevertheless, this level of increased resistance does not validate the reputation for radioresistance often ascribed to melanoma in accepted texts. The resolution of this discrepancy has come from in vitro studies demonstrating that melanoma cells are moderately less susceptible to radiation only at lower doses.[12] These results suggested that using greater doses per fraction would result in superior outcomes. This belief has been supported by several clinical studies that have used higher doses per fraction (≥ 4 Gy) and achieved high response rates in an assortment of primary and metastatic sites in patients receiving regional irradiation.[13-15] However, in the only prospective randomized trial (Radiation Therapy Oncology Group 83-05) that compared a greater fraction size (800–3,200 cGy; biologically effective dose [BED] acute 57.6 Gy, BED late 117 Gy) with conventional fraction size (250–5,000 cGy; BED acute 62.5 Gy, BED late 92 Gy ), no significant difference in either complete response or partial response was detected.[16]
Studies by Overgaard and Sause have found that the total radiation therapy dose may be the best prognostic factor for metastatic melanoma.[17,18] A recent Australian study of two palliative regimens of whole-brain radiotherapy doses for metastatic melanoma also demonstrated improved intracranial control for patients treated at 2 Gy per fraction twice daily to 40 Gy, compared with 4-Gy fractions given once daily to 20 Gy.[19] These findings demonstrate that if the proper total dosages are employed, the term radioresistant need not apply to malignant melanoma and that larger fraction sizes may not be necessary.
For patients with resectable local-regional disease, Ang and colleagues from M. D. Anderson Cancer Center achieved excellent complete and partial response rates with six fractions of 6 Gy given twice per week.[20] The study demonstrated an overall 5-year local-regional control rate for patients with high-risk disease (melanomas at least 1.5 mm thick, clinically detectable nodal disease or patients with previously resected disease) of 88%, surpassing results from previous trials of a similar nature. However, a review of adjuvant treatment of melanoma by Chang and colleagues from the University of Florida showed no difference between 6-Gy fractions to 30 Gy and 2-Gy fractions to a median of 60 Gy, with similarly impressive local/regional control rates[21] Patients with incurable disease and large axillary or ilioinguinal nodal metastases (but with a satisfactory life expectancy) can be effectively treated with a lower dose per fraction (eg, 50 Gy in 25 fractions) to decrease the risk of neuropathy or limb edema.[20]
The size of the metastatic lesion has been shown to be predictive of response, as lesions of 1 cc volume or less respond almost universally to radiation.[22] Furthermore, a study by Overgaard et al showed that tumors < 3 cm in size and > 5 cm in size had 79% and 21% complete response rates, respectively. Likewise, in the RTOG study, lesions > 5 cm had a complete response rate of only 20%. A study by Bentzen et al showed that the radiation sensitivity of melanoma cells varies within the same individual according to the site of biopsy, depending on whether the neoplasm is from the primary site, regional nodes, or distant metastases.[23] The study showed that metastatic deposits were by and large less responsive to radiation than their primary lesions, signifying that tumor development may result in the evolution of a radiation-resistant tumor phenotype.
When put in the perspective of other palliative uses of radiation therapy, the response of radiated malignant melanomas that have metastasized to the brain or bone is virtually identical to the response observed following treatment of other types of disseminated neoplasms that are generally regarded as radiosensitive.[12,24] The most frequent sites of metastatic melanoma are skin, subcutaneous tissue, and lymph nodes, comprising 50% of metastases.[20] The lung is the next most frequent site of occurrence, followed by the liver, brain, and bone.[25]
Seegenschmiedt et al analyzed prognostic factors and survival in patients with metastatic melanoma treated with external beam radiotherapy. The investigators found that in patients with soft tissue, skin, and/or lymph node metastases (UICC [International Union Against Cancer] stage IV), the median survival time was 7 months.[5] Patients with solitary metastases had a median survival time of 8 months, while those with multiple metastases had a median survival time of 4 months.[5] Finally, patients treated for bone metastases had a median survival time of 6.6 months, compared with 5.3 months for those treated for CNS metastases and 3.6 months for patients treated for pulmonary metastases.[5] Our patient’s survival time was similar to these described outcomes.
After the results of the randomized RTOG study are grouped with the findings of elective radiation therapy, it becomes apparent that malignant melanomas are not resistant to radiation therapy. Furthermore, the results of these studies clearly demonstrate that radiation therapy can be very effective, especially when only small volumes of disease need to be treated and adequate total doses are used. Therefore, radiation therapy appears to play an important yet underutilized role in the treatment of metastatic melanomas. Regardless of the effectiveness of radiation therapy to control disease locally, increases in survival will necessitate use of more successful systemic agents. Only then will the effects of refined local treatment improve our ability to increase survival and quality of life for patients with melanoma.
Financial Disclosure:The authors have no significant financial interest or other relationship with the manufacturers of any products or providers of any service mentioned in this article.
References:
References
1. Tsao H, Atkins MB, Sober AJ: Management of cutaneous melanoma. N Engl J Med 351: 998-1012, 2004.
2. Baade P, Coory M: Trends in melanoma mortality in Australia: 1950-2002 and their implications for melanoma control. Aust N Z Public Health 29:383-386, 2005.
3. Delaney G, Barton M, Jacob S: Estimation of an optimal radiotherapy utilization rate for melanoma: A review of the evidence. Cancer 100:1293-1301, 2004.
4. Urist MU, Karnell LH: The national cancer data base-Report on melanoma. Cancer 74:782-788, 1994.
5. Seegenschmiedt MH, Keilholz L, Altendorf-Hoffman A, et al: Palliative radiotherapy for recurrent and metastatic malignant melanoma: prognostic factors for tumor response and long-term outcome: A 20-year experience. Int J Radiat Oncol Biol Phys 44:607-618, 1999.
6. Paterson R: Classification of tumors in relation to radiosensitivity. Br J Radiol 6:218-233, 1933.
7. Overgaard J: The role of radiotherapy in recurrent and metastatic malignant melanoma: A clinical-radiobiologic study. Int J Radiat Oncol Biol Phys 12:867-872, 1986.
8. Habernalz HJ, Fischer JJ: Radiation therapy of malignant melanoma: Experience with high individual treatment doses. Cancer 38:2258-2262, 1976.
9. Sause WT, Cooper JS, Rush S, et al: Fraction size in external beam radiation therapy in the treatment of melanoma. Int J Radiat Oncol Biol Phys 20:429-432, 1991.
10. Rofstad EK: Local tumor control following single dose irradiation of human melanoma xenografts: Relationship of cellular radiosensitivity and influence of an immune response by athymic mouse. Cancer Res 49:3163-3168, 1998.
11. Rofstad EK: Radiation biology of malignant melanoma. Cancer Res 25:1-10, 1986.
12. Carella RJ, Gelber R, Hendrickson F, et al: Value of radiation therapy in the management of patient with cerebral metastases from malignant melanoma; R.T.O.G. Brain metastases study I & II. Cancer 45:679-683, 1980.
13. Katz HR: The results of different fractionation schemes in the palliative irradiation of metastatic melanoma. Int J Radiat Oncol Biol Phys 7:907-911, 1981.
14. Ho RC: Medical management of stage IV malignant melanoma: medical issues. Cancer 15;75(2 Suppl): 735-741, 1995.
15. Habermaltz HJ: Irradiation of malignant melanoma: experience in the past and present. Int J Radiat Oncol Biol Phys 7:131-133, 1981.
16. Sause WT, Cooper JS, Rush S, et al: Fraction size in external beam radiotherapy in the treatment of melanoma. Int J Radiat Oncol Biol Phys 20:429-432, 1991.
17. Overgaard J, Overgaard M, Hansen PV, et al: Some factors of importance in the radiation treatment of malignant melanoma. Radiother Oncol 5:183-192, 1986.
18. Barranaco SC, Romsdahl MM, Humphrey RM: The radiation response of human malignant melanoma cells grown in vitro. Cancer Res 31: 830-833, 1971.
19. Graham PH, Bucci J, Browne L: Randomized comparison of whole brain radiotherapy, 20 Gy in four daily fractions versus 40 Gy in 20 twice-daily fractions, for Brain Metastases. Corrected Proof. . Int J Radiat Oncol Biol Phys 15 October 2009 (ePub ahead of print)
20. Ang KK, Peters LJ, Weber RS, et al: Postoperative radiotherapy for cutaneous melanoma of the head and neck region. Int J Radiat Oncol Biol Phys 30:795-798, 1994.
21. Change DT, Amdur RJ, Morris CG, et al: Adjuvant radiotherapy for cutaneous melanoma: Comparing hypofractionation to conventional fractionation. Int J Radiat Oncol Biol Phys 66:1051-1055, 2006.
22. Overgaard J: Radiation therapy of malignant melanoma. Int J Radiat Oncol Biol Phys 6:41-44, 1980.
23. Bentzen SM, Overgaard J, Thames HD, et al: Clinical radiobiology of malignant melanoma. Radiother Oncol 16:169-182, 1989.
24. Ang KK, Byers RM, Peters LJ, et al: Regional radiotherapy as an alternative to nodal dissection for high risk cutaneous malignant melanoma of the head and neck. Arch Otolaryngol Head Neck Surg 116:169, 1990.
25. Habernalz HJ, Fischer JJ: Radiation therapy of malignant melanoma: Experience with high individual treatment doses. Cancer 38:2258-2262, 1976.