Hereditary Renal Tumor Syndromes and the Use of mTOR Inhibitors
A 47-year-old woman with a history of drug-resistant epilepsy during childhood presented to the emergency department with sudden dyspnea and chest pain. Upon admission, her oxygen saturation was 88%.
The Case
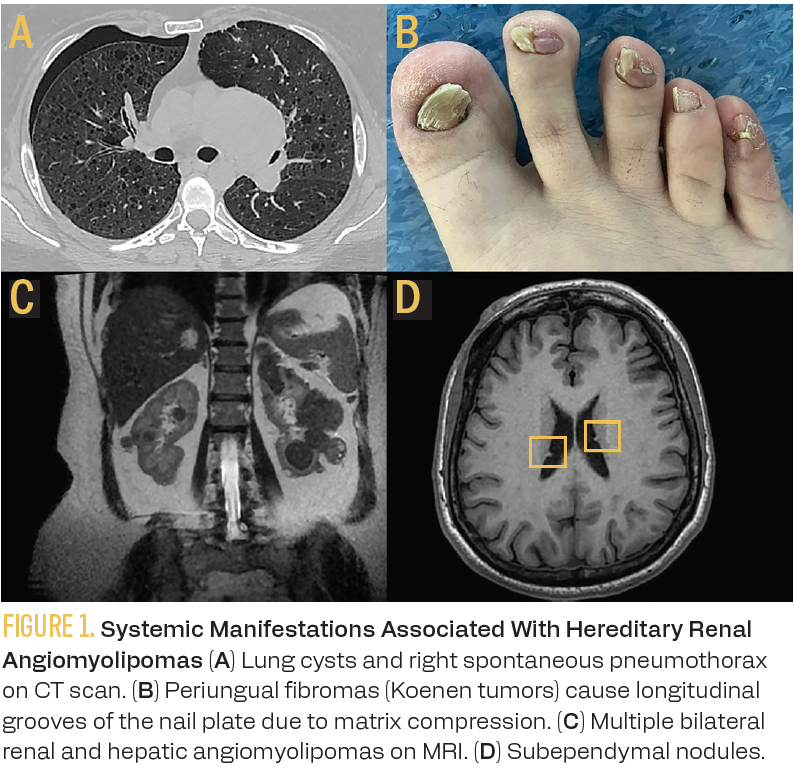
A 47-year-old woman with a history of drug-resistant epilepsy during childhood presented to the emergency department with sudden dyspnea and chest pain. Upon admission, her oxygen saturation was 88%. A chest CT scan revealed pulmonary cystic lesions consistent with lymphangioleiomyomatosis and a right spontaneous pneumothorax (Figure 1A), which resolved with the placement of a chest tube. Physical examination revealed a hypopigmented macule on the skin of the lumbar region, facial angiofibromas, and periungual fibromas (Figure 1B). An abdominal MRI documented multiple bilateral renal tumors that were hypointense on T2-weighted imaging and showed a black boundary artifact, suggestive of fat-poor angiomyolipomas (AMLs) (Figure 1C). Subsequent percutaneous biopsy of the largest renal tumor confirmed the diagnosis of angiomyolipoma (positive for HMB-45 on immunohistochemistry). The brain MRI revealed subependymal nodules (Figure 1D). The pulmonary function tests showed a mild obstructive pattern. Germline genetic testing confirmed the suspected diagnosis, and the patient started oral systemic treatment with everolimus (Afinitor) 10 mg once daily, along with dexamethasone rinses for prophylaxis.
Figures 1A-D
Which of the following hereditary cancer syndromes is associated with renal angiomyolipomas and the systemic findings presented by this patient?

It is estimated that 6.4% of non–clear cell renal carcinoma cases are associated with hereditary cancer syndromes.1 The National Comprehensive Cancer Network guidelines recommend genetic cancer risk assessment for individuals with a diagnosis of RCC aged 46 years and younger, with multifocal or bilateral tumors, with 1 or more first- or second-degree relatives with RCC, or meeting specific histologies and clinical features of a hereditary kidney cancer syndrome.2 Therefore, oncologists must be mindful of potential hereditary kidney cancer syndromes and refer patients for genetic counseling. Genodermatoses are hereditary diseases characterized by distinctive skin lesions often accompanied by multiorgan effects. While these are often benign tumors, some of them predispose individuals to cancer.3,4
BHD is one of the main differential diagnoses for patients presenting with skin hamartomas, pulmonary cysts, pneumothorax, and renal tumors. These findings overlap with clinical features of other genodermatoses, especially TSC. The key findings to distinguish these 2 entities are the histologic findings of skin and renal tumors, as well as ruling out cardiac or central nervous system (CNS) manifestations that would be more common with TSC. BHD is caused by germline pathogenic variants (PVs) in FLCN, which encodes folliculin.5 Unlike in this patient, the characteristic skin lesions of BHD are fibrofolliculomas and trichodiscomas, benign hair follicle tumors. However, BHD can also present less frequently with angiofibromas.6 The histology of BHD renal neoplasms is diverse, the most common being chromophobe, followed by oncocytoma and, more rarely, hybrid chromophobe-oncocytoma.7 Although 84% of individuals with BHD have lung cysts on a CT scan, and up to 38% have a history of spontaneous pneumothorax, the skin findings, histology of renal tumors, and history of CNS manifestations of this patient do not align with the diagnosis of BHD.8 Therefore, answer A is incorrect.
CS is part of the PHTS spectrum, a group of diseases associated with germline PVs in PTEN, a tumor suppressor gene that encodes a phosphatase antagonizing cell cycle progression by intervening in the PI3K-AKT pathway.9 The most common mucocutaneous characteristics of CS are multiple facial trichilemmomas and oral papillomas.10 PTEN PVs have been associated with an increased risk of breast cancer in women, endometrial cancer, thyroid cancer, colon polyps, and familial kidney cancer.11 The histopathological spectrum of renal neoplasms reported in individuals with CS is predominantly chromophobe and papillary RCC.12 Although this patient has CNS involvement, she does not exhibit the classic manifestations associated with PHTS, such as Lhermitte-Duclos disease (dysplastic gangliocytoma of the cerebellum), macrocephaly, or autism spectrum disorder.13 Thus, answer B is incorrect.
HLRCC is caused by heterozygous germline PVs in FH which encodes an enzyme that catalyzes the conversion of fumarate to malate in the tricarboxylic acid cycle.14 The main manifestations of HLRCC are cutaneous leiomyomas, uterine fibroids, and type 2 papillary RCC.15 Renal tumors associated with germline PVs in FH are of the papillary subtype, are unifocal, and have a high potential for developing early metastasis. The lifetime risk of developing renal cancer in carriers of PVs in FH is estimated to be 5.8% to 11.9%.16 This patient did not have cutaneous or uterine leiomyomas and her renal tumors are more consistent with AML rather than papillary RCC, ruling out clinical suspicion for HLRCC. Therefore, answer C is incorrect.
HPRC is caused by germline PVs in MET that result in a gain of function of the kinase domain of c-MET (hepatocyte growth factor receptor), functioning as a driver in the tumorigenesis of papillary renal cancer.17 Clinicians should suspect that an individual might be a carrier of a PV in MET in cases of papillary RCC type 1, bilateral/multifocal tumors, and a family history of papillary RCC.18 To date, no associations have been made with extrarenal manifestations in HPRC.19 Thus, answer D is incorrect.
The patient fulfills the clinical diagnosis of TSC with multisystem involvement affecting the skin (facial angiofibromas, periungual fibromas, hypomelanotic spots); CNS (epilepsy, subependymal nodules); lungs (lymphangioleiomyomatosis [LAM] causing pneumothorax); and kidneys (AMLs).20 Answer E is correct.
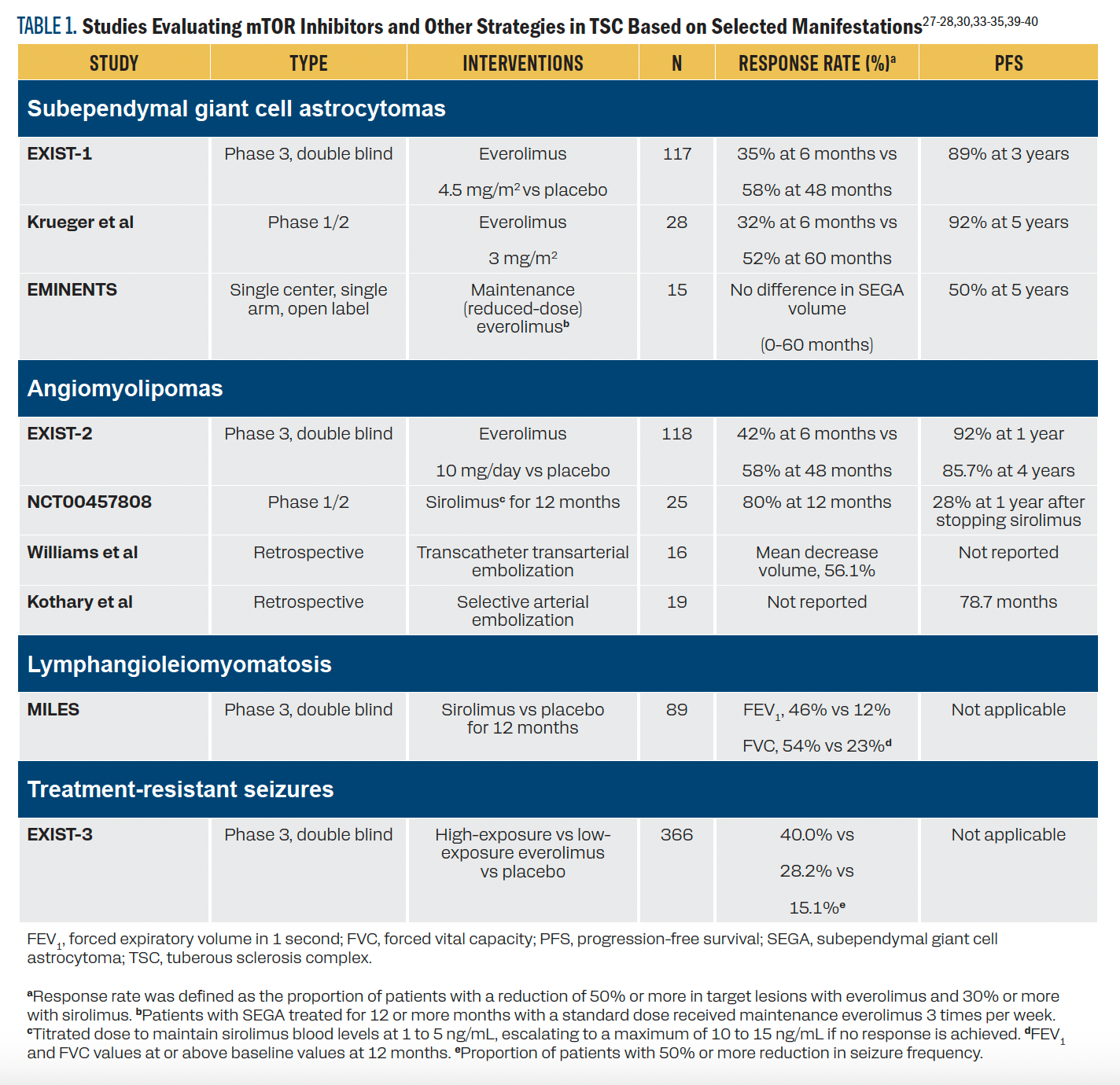
Tuberous sclerosis complex is an autosomal dominant inherited disease associated with PVs in TSC1 (encoding hamartin) and more frequently TSC2 (encoding tuberin). The mTOR pathway aims to activate ribosomal complexes, allowing cell proliferation, while the TSC1-TSC2 heterodimer inhibits mTOR, controlling cell cycle progression.21 This finding provided the rationale for the use of mTOR inhibitors for the treatment of patients with TSC-associated tumors (Table 1).
TABLE 1. Studies Evaluating mTOR Inhibitors and Other Strategies in TSC Based on Selected Manifestations27-28,30,33-35,39-40
Although AMLs are often benign tumors, these patients require multidisciplinary management, ideally at a genitourinary-oncology clinic. These tumors can follow an indolent course, as shown in patients who received a placebo in earlier clinical trials. In light of this, active surveillance (AS) is an important strategy to discuss. The International TSC Diagnostic Criteria and Surveillance and Management Recommendations, updated in 2021, recommend MRI as the initial screening tool for renal AMLs because up to one-third may be fat-poor and not adequately identified with ultrasound.
For patients who are asymptomatic with AMLs less than 3 cm with a slow growth rate (typically less than 5 mm/year), it is reasonable to observe with annual MRI, monitoring blood pressure for potential secondary hypertension, as well as tracking proteinuria, hematuria, and glomerular filtration rate.22,23
In a meta-analysis involving individuals with predominantly sporadic AMLs, diverse AS protocols showed a low rate of spontaneous bleeding (2.3% to 3.1%) and the need for active treatment ranged from 2.2% to 13.1%.24 In a single-center cohort, independent risk factors for AS discontinuation included AML size 4 cm or more (HR, 11.23; 95% CI, 3.41-37.03) and being symptomatic at diagnosis (HR, 3.74; 95% CI, 1.41-9.90).25 When determining the eligibility of individuals for AS, it is important to consider that TSC-associated AMLs (approximately 10% of all cases), in comparison with sporadic cases, may have a faster growth rate and are more likely to require active treatment.26
Local treatment options for renal AMLs include selective arterial embolization or surgical resection, particularly when complications such as secondary retroperitoneal bleeding (Wunderlich syndrome), pain, or rapid growth arise. In settings where access to mTOR inhibitors is limited, and for patients who are not candidates for mTOR inhibitors, selective arterial embolization could be considered for the management of renal AMLs.27,28 Patients with TSC are at a high risk of chronic kidney disease due to the replacement of normal renal parenchyma with multiple bilateral AMLs and renal cysts.23 Therefore, if the multidisciplinary consensus agrees on surgical treatment, it should, whenever possible, adhere to the principles of nephron-sparing surgery29 with the aim of delaying the need for renal replacement therapy.
The effectiveness of systemic treatment with mTOR inhibitors for TSC-associated manifestations has been demonstrated in several clinical trials. The phase 3 EXIST-2 trial (NCT00790400) included individuals 18 years or older with TSC or sporadic lymphangioleiomyomatosis and renal AMLs. The response rate at 24 weeks was 42% with everolimus vs 0% with placebo (P < .0001). The median time to response with everolimus was 2.9 months. The median time to AML progression was not reached with everolimus and 11.4 months with placebo. Interestingly, VEGF-D levels decreased with everolimus treatment and correlated with the reduction in AML size.30 In the extended phase of the trial where crossover was permitted, the overall AML response rate was 58%, with a duration of response ranging from 3.0 to 55.5 months.31
Even though it is more commonly used for TSC-associated lymphangioleiomyomatosis, sirolimus administered for 1 year has also demonstrated effectiveness in reducing the volume of AMLs. In an open-label phase 2 trial (NCT00457808), at 12 months, 16 of the 20 patients had at least a 30% reduction in AML volume. However, at 6 and 12 months after stopping sirolimus, the mean AML volume had increased to 76.8% ± 27.5% (P < .001) and 85.9% ± 28.5% of the respective baseline volumes, highlighting the need for continuous treatment with everolimus.32
Although the EXIST-1 trial (NCT00789828) was a phase 3, double-blind study focused on patients with subependymal giant cell astrocytoma (SEGA), 38% had AMLs. Its primary end point was SEGA response rate defined as volume reduction of 50% or more. In the initial analysis, with a median follow-up of 9.7 months, the SEGA response rate with everolimus was 35% vs 0% with placebo (P < .0001). Exploratory analysis also showed greater response rates in skin lesions (42% vs 11%) and AMLs (53% vs 0%).33 In the extended follow-up, the median exposure to everolimus was 47.1 months. With longer follow-up, the overall SEGA response rate was 58%, and the 3-year progression-free survival was 89%. The overall response rate in AMLs also increased over time, reaching 73%, with a median duration of response of 42.3 months and no renal hemorrhage events.34
Lymphangioleiomyomatosis affects 30% to 40% of women with TSC and is associated with a risk of pneumothorax and progressive decline in lung function. The phase 3 MILES trial (NCT00414648) demonstrated a significant benefit of giving sirolimus for 1 year, stabilizing lung function compared with placebo (forced expiratory volume in 1-second slope 1 ± 2 mL/month vs –12 ± 2 mL/month with placebo, P < .001).35
Epilepsy is the most common neurological manifestation, with a higher risk of drug-resistant epilepsy compared with non-TSC-associated seizure disorders (60% vs 30% to 40%). EXIST-3 is the largest phase 3 clinical trial (NCT01713946) evaluating the use of everolimus for drug-resistant focal-onset seizures in patients with TSC. It demonstrated a lower frequency of seizures in patients treated with high everolimus exposure (14.9% vs 29.3% vs 39.6% with placebo, low [3 to 7 ng/mL], and high exposure [9 to 15 ng/mL], respectively; P <.001).36
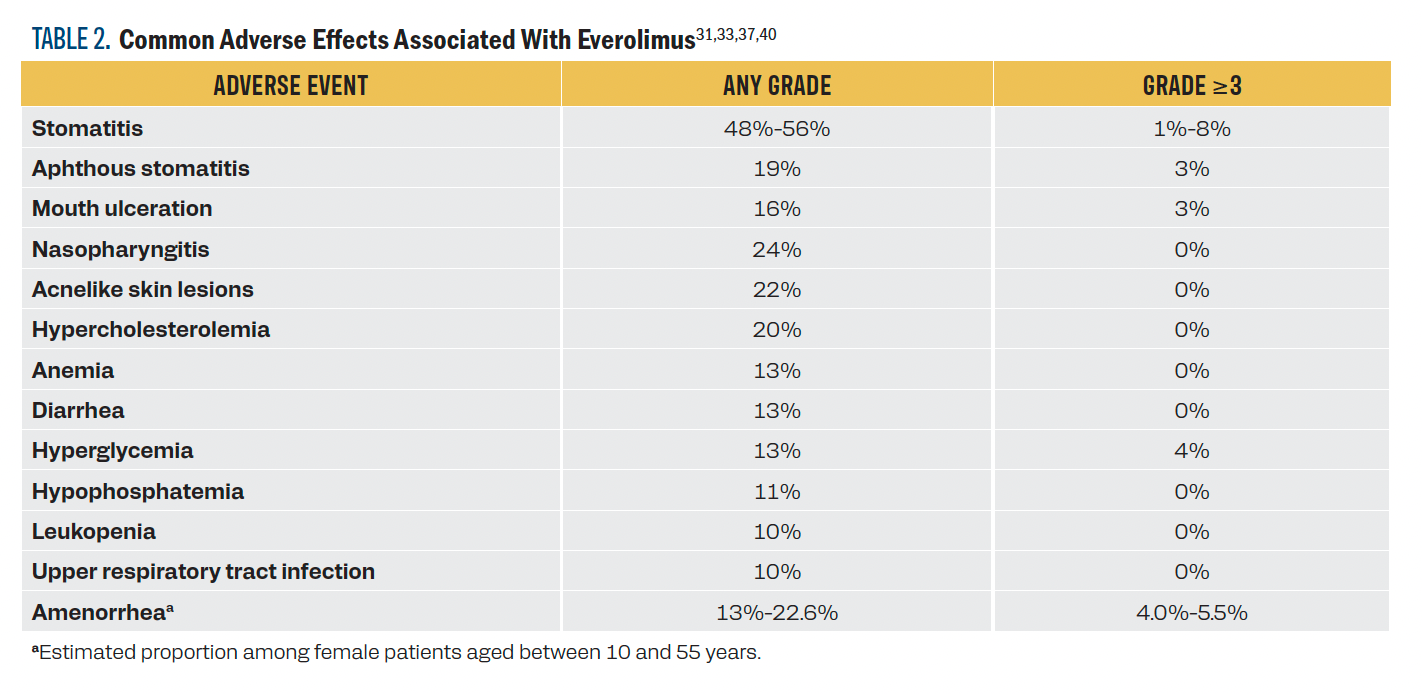
The adverse effects (AEs) of mTOR inhibitors can limit their use. The most frequent AE is stomatitis, a class effect characterized by aphthous-like oral lesions. Stomatitis occurs in about 50% of cases at any grade and in 1% to 8% of cases at grade 3 or higher (Table 2). The best evidence on the prevention of oral mucositis comes from the single-arm phase 2 SWISH trial (NCT02069093). This study evaluated the efficacy of 0.5 mg/5 mL dexamethasone mouthwash 4 times a day in women who are postmenopausal and started everolimus plus exemestane for hormone receptor–positive/HER2-negative metastatic breast cancer. It showed that dexamethasone mouthwashes reduced the frequency of grade 2 or higher stomatitis at 8 weeks (2% in SWISH vs 33% in historical reports from the phase 3 BOLERO-2 study [NCT00863655]).
TABLE 2. Common Adverse Effects Associated With Everolimus31,33,37,40
Fatigue, pneumonitis, rash, hyperglycemia, and hypertriglyceridemia are other important AEs of mTOR inhibitors.
In summary, mTOR inhibitors act as disease modifiers in individuals with TSC, halting the progression of associated tumors (SEGAs, AMLs, LAM, skin lesions), delaying respiratory function deterioration, and improving epilepsy control, all of which converge to enhance quality of life. This case illustrates how personalized management can significantly improve clinical outcomes in rare diseases.38
Outcome
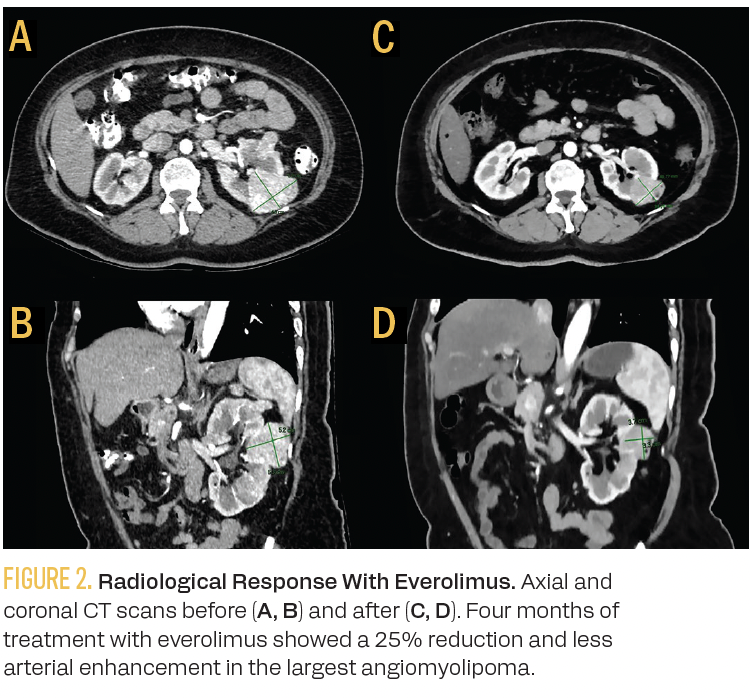
Multigene panel testing showed a heterozygous germline pathogenic variant in TSC2 [c.2172_2176del p.Thr725Profs*35]. After 16 weeks on everolimus 10 mg/day and primary prophylaxis with dexamethasone mouthwash, the patient did not experience any episodes of stomatitis or biochemical abnormalities. She achieved a 25% reduction in the size of the renal AML, with the largest tumor decreasing from 6 cm to 4.5 cm (Figure 2).
Figure 2.
CORRESPONDING AUTHOR
María Teresa Bourlon, MD, MSc
Department of Hematology and Oncology
Instituto Nacional de Ciencias Médicas y Nutrición
Salvador Zubirán
15 Vasco de Quiroga Street
Section XVI, Tlalpan
Ciudad de Mexico 14080, Mexico
Email: maitebourlon@gmail.com
CONFLICT OF INTEREST
José Luis Rodríguez-Olivares, MD: None
Héctor Raúl González-Sánchez, MD: None
Evelyn Lilian Beas-Lozano, MD: None
Elaine T. Lam, MD: None
María Teresa Bourlon, MD, MSc: Speaker for Merck Sharp & Dohme, Merck, Ipsen, Bristol Myers Squibb, Pfizer and Eisai
References
- Santos M, Lanillos J, Roldan-Romero JM, et al. Prevalence of pathogenic germline variants in patients with metastatic renal cell carcinoma. Genet Med. 2021;23(4):698-704. doi:10.1038/s41436-020-01062-0
- NCCN Clinical Practice Guidelines in Oncology. Kidney cancer, version 2.2025. Accessed March 11, 2024. https://shorturl.at/ysIHM
- Ladd R, Davis M, Dyer JA. Genodermatoses with malignant potential. Clin Dermatol. 2020;38(4):432-454. doi:10.1016/j.clindermatol.2020.03.007
- Carley H, Kulkarni A. Genodermatoses – opportunities for early detection and cancer prevention. Curr Genet Med Rep. 2022;10(1):1-13. doi:10.1007/s40142-022-00203-y
- Nickerson ML, Warren MB, Toro JR, et al. Mutations in a novel gene lead to kidney tumors, lung wall defects, and benign tumors of the hair follicle in patients with the Birt-Hogg-Dubé syndrome. Cancer Cell. 2002;2(2):157-164. doi:10.1016/s1535-6108(02)00104-6
- Menko FH, van Steensel MA, Giraud S, et al; European BHD Consortium. Birt-Hogg-Dubé syndrome: diagnosis and management. Lancet Oncol. 2009;10(12):1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Benusiglio PR, Giraud S, Deveaux S, et al; French National Cancer Institute Inherited Predisposition to Kidney Cancer Network. Renal cell tumour characteristics in patients with the Birt-Hogg-Dubé cancer susceptibility syndrome: a retrospective, multicentre study. Orphanet J Rare Dis. 2014;9:163. doi:10.1186/s13023-014-0163-z
- Toro JR, Wei MH, Glenn GM, et al. BHD mutations, clinical and molecular genetic investigations of Birt-Hogg-Dubé syndrome: a new series of 50 families and a review of published reports. J Med Genet. 2008;45(6):321-331. doi:10.1136/jmg.2007.054304
- Lee YR, Chen M, Pandolfi PP. The functions and regulation of the PTEN tumour suppressor: new modes and prospects. Nat Rev Mol Cell Biol. 2018;19(9):547-562. doi:10.1038/s41580-018-0015-0
- Pilarski R, Burt R, Kohlman W, Pho L, Shannon KM, Swisher E. Cowden syndrome and the PTEN hamartoma tumor syndrome: systematic review and revised diagnostic criteria. J Natl Cancer Inst. 2013;105(21):1607-1616. doi:10.1093/jnci/djt277
- Cummings S, Alfonso A, Hughes E, et al. Cancer risk associated with PTEN pathogenic variants identified using multigene hereditary cancer panel testing. JCO Precis Oncol. 2023;7:e2200415. doi:10.1200/PO.22.00415
- Kozman D, Kao CS, Nguyen JK, et al. Renal neoplasia occurring in patients with PTEN hamartoma tumor syndrome : clinicopathologic study of 12 renal cell carcinomas from 9 patients and association with intrarenal “lipomas.” Am J Surg Pathol. 2023;47(9):1001-1010. doi:10.1097/PAS.0000000000002079
- Alanazi AI, Alanezi T, Aljofan ZF, Alarabi A, Elwatidy S. Lhermitte-Duclos disease: a systematic review. Surg Neurol Int. 2023;14:351. doi:10.25259/SNI_555_2023
- Valcarcel-Jimenez L, Frezza C. Fumarate hydratase (FH) and cancer: a paradigm of oncometabolism. Br J Cancer. 2023;129(10):1546-1557. doi:10.1038/s41416-023-02412-w
- Menko FH, Maher ER, Schmidt LS, et al. Hereditary leiomyomatosis and renal cell cancer (HLRCC): renal cancer risk, surveillance and treatment. Fam Cancer. 2014;13(4):637-644. doi:10.1007/s10689-014-9735-2
- Shuch B, Li S, Risch H, Bindra RS, McGillivray PD, Gerstein M. Estimation of the carrier frequency of fumarate hydratase alterations and implications for kidney cancer risk in hereditary leiomyomatosis and renal cancer. Cancer. 2020;126(16):3657-3666. doi:10.1002/cncr.32914
- Tovar EA, Graveel CR. MET in human cancer: germline and somatic mutations. Ann Transl Med. 2017;5(10):205. doi:10.21037/atm.2017.03.64
- Sebai M, Tulasne D, Caputo SM, et al. Novel germline MET pathogenic variants in French patients with papillary renal cell carcinomas type I. Hum Mutat. 2022;43(3):316-327. doi:10.1002/humu.24313
- Jacoba IM, Lu Z. Hereditary papillary renal cell carcinoma. Semin Diagn Pathol. 2024;41(1):28-31. doi:10.1053/j.semdp.2023.12.002
- Crino PB, Nathanson KL, Henske EP. The tuberous sclerosis complex. N Engl J Med. 2006;355(13):1345-1356. doi:10.1056/NEJMra055323
- El-Hashemite N, Zhang H, Henske EP, Kwiatkowski DJ. Mutation in TSC2 and activation of mammalian target of rapamycin signalling pathway in renal angiomyolipoma. Lancet. 2003;361(9366):1348-1349. doi:10.1016/S0140-6736(03)13044-9
- Northrup H, Aronow ME, Bebin EM, et al; International Tuberous Sclerosis Complex Consensus Group. Updated International Tuberous Sclerosis Complex Diagnostic Criteria and Surveillance And Management Recommendations. Pediatr Neurol. 2021;123:50-66. doi:10.1016/j.pediatrneurol.2021.07.011
- Kingswood JC, Belousova E, Benedik MP, et al. Renal manifestations of tuberous sclerosis complex: key findings from the final analysis of the TOSCA study focussing mainly on renal angiomyolipomas. Front Neurol. 2020;11:972. doi:10.3389/fneur.2020.00972
- Zeid M, Sayedin H, Nabi N, et al. Active surveillance for renal angiomyolipoma less than 4 centimeters: a systematic review of cohort studies. Cureus. 2022;14(2):e22678. doi:10.7759/cureus.22678
- Ouzaid I, Autorino R, Fatica R, et al. Active surveillance for renal angiomyolipoma: outcomes and factors predictive of delayed intervention. BJU Int. 2014;114(3):412-417. doi:10.1111/bju.12604
- Swärd J, Henrikson O, Lyrdal D, Peeker R, Lundstam S. Renal angiomyolipoma-patient characteristics and treatment with focus on active surveillance. Scand J Urol. 2020;54(2):141-146. doi:10.1080/21681805.2020.1716066
- Williams JM, Racadio JM, Johnson ND, Donnelly LF, Bissler JJ. Embolization of renal angiomyolipomata in patients with tuberous sclerosis complex. Am J Kidney Dis. 2006;47(1):95-102. doi:10.1053/j.ajkd.2005.09.028
- Kothary N, Soulen MC, Clark TW, et al. Renal angiomyolipoma: long-term results after arterial embolization. J Vasc Interv Radiol. 2005;16(1):45-50. doi:10.1097/01.RVI.0000143769.79774.70
- Metwalli AR, Linehan WM. Nephron-sparing surgery for multifocal and hereditary renal tumors. Curr Opin Urol. 2014;24(5):466-473. doi:10.1097/MOU.0000000000000094
- Bissler JJ, Kingswood JC, Radzikowska E, et al. Everolimus for angiomyolipoma associated with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis (EXIST-2): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet. 2013;381(9869):817-824. doi:10.1016/S0140-6736(12)61767-X
- Bissler JJ, Kingswood JC, Radzikowska E, et al. Everolimus long-term use in patients with tuberous sclerosis complex: four-year update of the EXIST-2 study. PLoS One. 2017;12(8):e0180939. doi:10.1371/journal.pone.0180939
- Bissler JJ, McCormack FX, Young LR, et al. Sirolimus for angiomyolipoma in tuberous sclerosis complex or lymphangioleiomyomatosis. N Engl J Med. 2008;358(2):140-151. doi:10.1056/NEJMoa063564
- Franz DN, Belousova E, Sparagana S, et al. Efficacy and safety of everolimus for subependymal giant cell astrocytomas associated with tuberous sclerosis complex (EXIST-1): a multicentre, randomised, placebo-controlled phase 3 trial. Lancet. 2013;381(9861):125-132. doi:10.1016/S0140-6736(12)61134-9
- Franz DN, Belousova E, Sparagana S, et al. Long-term use of everolimus in patients with tuberous sclerosis complex: final results from the EXIST-1 study. PLoS One. 2016;11(6):e0158476. doi:10.1371/journal.pone.0158476
- McCormack FX, Inoue Y, Moss J, et al; MILES Trial Group. Efficacy and safety of sirolimus in lymphangioleiomyomatosis. N Engl J Med. 2011;364(17):1595-1606. doi:10.1056/NEJMoa1100391
- French JA, Lawson JA, Yapici Z, et al. Adjunctive everolimus therapy for treatment-resistant focal-onset seizures associated with tuberous sclerosis (EXIST-3): a phase 3, randomised, double-blind, placebo-controlled study. Lancet. 2016;388(10056):2153-2163. doi:10.1016/S0140-6736(16)31419-2
- Rugo HS, Seneviratne L, Beck JT, et al. Prevention of everolimus-related stomatitis in women with hormone receptor-positive, HER2-negative metastatic breast cancer using dexamethasone mouthwash (SWISH): a single-arm, phase 2 trial. Lancet Oncol. 2017;18(5):654-662. doi:10.1016/S1470-2045(17)30109-2
- Auvin S, Bissler JJ, Cottin V, et al. A step-wise approach for establishing a multidisciplinary team for the management of tuberous sclerosis complex: a Delphi consensus report. Orphanet J Rare Dis. 2019;14(1):91. doi:10.1186/s13023-019-1072-y
- Krueger DA, Care MM, et al. Everolimus long-term safety and efficacy in subependymal giant cell astrocytoma. Neurology. 2013;80(6):574-80. doi: 10.1212/WNL.0b013e3182815428
- Bobeff K, Krajewska K, et al. Maintenance Therapy With Everolimus for Subependymal Giant Cell Astrocytoma in Patients With Tuberous Sclerosis - Final Results From the EMINENTS Study. Front Neurol. 2021;12:581102. doi: 10.3389/fneur.2021.581102