Managing Acute Myeloid Leukemia in the Elderly
Acute myeloid leukemia (AML) is a disease of the elderly, with the majority of patients diagnosed in their 6th and 7th decade of life. Older patients with AML are less likely to achieve complete remission after induction chemotherapy, and they suffer from higher rates of leukemia relapse compared to younger cohorts. Suboptimal outcomes are the result of adverse biologic characteristics of leukemia in the elderly, as well as the presence of medical comorbidities and patient or physician preferences as to initiating treatment. In addition, there is a distinct lack of randomized, prospective data to guide management decisions for the treatment of AML in the elderly. Patients who are over age 75, with poor performance status, multiple comorbidities, or poor prognostic features, should be considered for a clinical trial or palliative therapy. Elderly patients who are candidates for standard induction chemotherapy and achieve complete remission are unlikely to benefit from intensive postremission therapy and should be referred to a clinical trial when possible. Further prospective trials are needed to identify a tolerable, effective treatment regimen for older patients with AML.
Acute myeloid leukemia (AML) is a disease of the elderly, with the majority of patients diagnosed in their 6th and 7th decade of life. Older patients with AML are less likely to achieve complete remission after induction chemotherapy, and they suffer from higher rates of leukemia relapse compared to younger cohorts. Suboptimal outcomes are the result of adverse biologic characteristics of leukemia in the elderly, as well as the presence of medical comorbidities and patient or physician preferences as to initiating treatment. In addition, there is a distinct lack of randomized, prospective data to guide management decisions for the treatment of AML in the elderly. Patients who are over age 75, with poor performance status, multiple comorbidities, or poor prognostic features, should be considered for a clinical trial or palliative therapy. Elderly patients who are candidates for standard induction chemotherapy and achieve complete remission are unlikely to benefit from intensive postremission therapy and should be referred to a clinical trial when possible. Further prospective trials are needed to identify a tolerable, effective treatment regimen for older patients with AML.
Older adults represent the fastest-growing segment of the population in the United States. Within the next 25 years, 20% of the US population is expected to be older than age 60.[1] Acute myeloid leukemia (AML) is a hematologic malignancy more commonly diagnosed in the older adult, with the median age of diagnosis being 67 years. For older individuals with acute leukemia, the mortality rate closely parallels incidence, thus highlighting the lethal nature of this disease. Several unique challenges in treating leukemia in the elderly ultimately contribute to the significantly inferior clinical outcomes seen in this group compared to younger cohorts. This article will discuss obstacles to treating elderly patients with acute leukemia, the biologic characteristics of AML in the elderly that lead to adverse outcomes, and treatment strategies aimed at improving overall survival.
Clinical Trials
The optimal chemotherapeutic regimen for patients younger than age 60 has been studied extensively in clinical trials of AML. Despite the fact that the majority of patients with acute leukemia are elderly, relatively few clinical trials have investigated treatment strategies in these patients for either curative or palliative intent.
The Southwest Oncology Group (SWOG) reported that the number of patients enrolled on clinical trials over age 65 between 1993 and 1996 were significantly underrepresented.[2] While only 25% of patients aged 65 years or older were enrolled in SWOG trials in the mid-1990s, this age cohort represented 63% of the cancer population in the United States during the same period. One explanation is that several trials simply excluded elderly patients from enrolling. However, even trials that did not exclude elderly patients had lower enrollment rates, which could be attributed to either patient or physician perception of decreased tolerance of cytotoxic chemotherapy or investigational agents. In fact, up to half of oncologists questioned have reported not enrolling patients on clinical trials based on age alone.[3]
Furthermore, older patients who are enrolled in clinical trials may not be representative of the elderly population at large.[4] Restrictive exclusion criteria of many clinical trials select for patients with fewer comorbidities, less advanced age, and a higher baseline performance status. Subsequently, our knowledge of the optimum treatment strategy in the older adult is limited by a distinct lack of reliable clinical trial data.
Biologic Characteristics of AML in the Elderly
TABLE 1

Comparison of FAB and WHO Classification of AML
AML is a clonal disorder that is characterized by an unregulated proliferation of undifferentiated myeloid progenitor cells in the bone marrow and peripheral blood. The classification of AML is based on both morphologic and cytogenetic criteria. A comparison of the French-American-British (FAB) classification system and the more recent World Health Organization (WHO) system is shown in Table 1.[5]
Unfavorable Karyotypes
Molecular and cytogenetic abnormalities are common in leukemia, and are of particular importance in elderly patients with AML (Table 2).[6] The presence of unfavorable karyotypes such as monosomy 5, monosomy 7, chromosome 11q23 abnormalities, and complex karyotype are much more commonly detected in elderly patients. Furthermore, cytogenetic abnormalities associated with favorable outcomes, such as inversion of chromosome 16, a translocation of chromosomes 15 and 17, or a translocation of chromosomes 8 and 21, are seen with considerably less frequency in older adults.
TABLE 2

Prognostic Classification Based on Karyotype Analysis
In a review of the Medical Research Council trials AML 10 (for patients younger than age 55) and AML 11 (for patients older than age 55), a distinct discrepancy was seen in karyotype distribution between the two age groups. Favorable cytogenetic profiles were detected in 24% vs 7% and unfavorable cytogenetic profiles in 6% vs 13% in younger vs older cohorts, respectively.[7] Furthermore, the presence of poor-risk karyotype profiles appears to continually increase with age. A recent report of five SWOG trials conducted between 1998 and 2006 indicated that the percentage of patients with favorable cytogenetics declined and the percentage with unfavorable cytogenetics rose precipitously for age groups less than 55, between 55 and 65, between 65 and 75, and greater than 75.[8] Thus, the presence of poor-risk cytogenetics increases with age and is associated with adverse outcomes in elderly patients with AML.
Cancer History
Antecedent hematologic malignancies are also associated with poor outcomes in patients with AML. Elderly patients are more likely to have a diagnosis of myelodysplastic syndrome (MDS) or a myeloproliferative disorder preceding the development of AML, and these patients consistently respond poorly to chemotherapy. Systemic exposure to chemotherapeutics that are known to induce a secondary AML, such as alkylating agents and topoisomerase II inhibitors are also more commonly experienced in elderly patients. Furthermore, adverse cytogenetic profiles are present in higher frequency in patients with preceding MDS and those with exposure to alkylating agents (chromosome 5 or 7 abnormalities) and topoisomerase II inhibitors (chromosome 11q23 abnormalities).[9] Together, these two factors contribute to inferior responses to standard treatment protocols.
MDR-1
In more recent years, the presence of a multidrug resistance phenotype has been identified as a negative prognostic finding in AML. The gene responsible for this phenotype (MDR-1) is located on the long arm of chromosome 7, and encodes a 170-kD glycoprotein or P-glycoprotein (PgP), which functions as an ATP-dependent efflux pump. In vitro models have shown that PgP overexpression has led to increased cellular export of antineoplastic drugs, including vinca alkaloids, anthracyclines, epipodophyllotoxins, and paclitaxel.[10]
MDR-1 is more commonly expressed in elderly patients and is associated with an increased resistance to anthracycline chemotherapy, with an associated decreased rate of complete remission and an inferior disease-free survival and overall survival.[11] In a comparison of patients younger than age 55 enrolled on the SWOG 8600 trial to patients older than age 55 enrolled on the SWOG 9031 trial, MDR-1 overexpression was detected in 30% vs 71% of younger and older patients, respectively. Thus, MDR-1 overexpression is a significant contributor to the likelihood of an adverse clinical outcome for elderly patients with AML.
Other Biomarkers
Numerous additional molecular markers have been identified in AML that affect response to chemotherapy and overall clinical outcomes. One example is the presence of fms-like tyrosine kinase (FLT-3) internal tandem duplications or point mutations, which have been shown in multiple studies to result in adverse outcomes.[12] While these mutations are not unique to elderly patients with AML, they appear to confer an inferior response to chemotherapy in both younger and older patients.
In contrast, the presence of nucleophosmin gene mutations (NPM1) has recently been linked to chemo-responsiveness in patients with AML.[13,14] The NPM1 gene encodes nucleophosmin, a nucleocytoplasmic shuttling protein that is normally present in the nucleolus and is responsible for maintaining genomic integrity. Mutations in exon 12 of the NPM1 gene result in cytoplasmic translocation of the protein and subsequent inhibition of the ARF/p53 tumor-suppressor pathway. The presence of cytoplasmic nucleophosmin in leukemic blasts of patients with AML has been linked to a normal karyotype, increased frequency of FLT-3 mutations, and a favorable response to induction chemotherapy.
While these molecular characteristics are not unique to elderly patients with AML, the presence of these, along with karyotype analysis and MDR-1 expression, can help determine a gene-expression profile that may guide therapeutic decisions.
Special Treatment Considerations in the Elderly
FIGURE 1
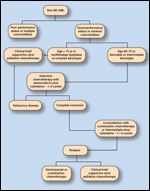
Treatment Algorithm for Elderly Patients With Newly Diagnosed AML
One substantial obstacle in treating older leukemia patients with induction chemotherapy is the presence of medical comorbidities, which can limit their ability to tolerate traditional chemotherapy regimens used to treat AML (Figure 1). Over 25% of cancer patients over age 75 have six or more comorbidities.[15] The presence and severity of comorbidities has an independent adverse effect on the survival of patients with all types of cancer.[16] This may be due, in part, to the physical burden of the medical condition, the increased risk of toxicity from intensive chemotherapy, or drug interactions between chemotherapeutic agents and drugs used to treat various medical conditions. For example, cardiotoxicity secondary to anthracyclines, which are commonly used to treat acute leukemia, occurs more frequently in elderly patients.
Furthermore, elderly patients are less likely to be offered intensive induction, owing largely to physician or patient concerns of excessive toxicity with standard chemotherapy regimens. In a review of Medicare claims between 1991 and 1996 of all patients older than age 60 diagnosed with AML, only 30% of patients received intensive chemotherapy.[17] For those patients who received induction chemotherapy, median survival was significantly prolonged compared to the entire leukemia group (8 vs 2 months).
However, it is not clear that intensive vs nonintensive options are offered to patients based on functional status or comorbidities alone. In a prospective analysis of elderly patients with AML and advanced MDS, 51% of patients opted for a less aggressive treatment algorithm.[18] Patients receiving intensive chemotherapy tended to be younger than those choosing best supportive care or nonintensive chemotherapy; however, baseline performance status or quality-of-life scores were not significantly different between the two groups. Thus, inferior outcomes may be, in part, attributable to elderly patients receiving low-intensity treatment regimens based on either physician or patient perception of their ability to tolerate cytotoxic agents.
Induction Chemotherapy
TABLE 3

Outcomes After Induction Therapy for AML in Younger (< 55–60 yr) and Older (> 55–60 yr) Patients
Standard induction chemotherapy for AML in adults has traditionally consisted of a combination of cytarabine and an anthracycline, with or without additional cytotoxic agents. Regardless of the induction regimen utilized, elderly patients are less likely than their younger counterparts to achieve complete remission from their leukemia (30%-50% vs 60%-80%, see Table 3).[7,11,19-28] Furthermore, long-term outcomes in this setting remain dismal for elderly patients, who have a median survival of only 6 to 12 months. Alternative strategies have been investigated to improve remission rates, prolong long-term survival, and minimize treatement-related toxicities that lead to increased morbidity and mortality. These strategies include the use of less-intensive chemotherapy regimens, altered dosing or choice of anthracyclines, addition of alternative chemotherapies, maintenance chemotherapy, and the use of growth factors in priming or neutrophil recovery.
Given the suboptimal outcomes using induction chemotherapy in elderly AML patients, several groups have investigated whether standard induction is superior to supportive care or low-dose chemotherapy for patients over age 50 to 65.[29-31] However, only one trial-by the European Organization for Research and Treatment of Cancer (EORTC)-provided prospective, randomized data to suggest that outcomes from induction therapy were superior to palliative care alone, showing median overall survivals of 22 vs 11 weeks, respectively (P = .015).[30] That said, the majority of patients enrolled in this trial were between the ages of 66 and 75, and thus, it remains unclear whether standard induction chemotherapy continues to be beneficial for patients older than age 75.
A retrospective study examined this question by comparing outcomes for patients over age 75 to those between 65 and 74 who received either induction chemotherapy, low-dose palliative chemotherapy, or supportive care alone.[32] For the cohort of patients older than 75 who received a standard anthracycline-based regimen, complete remission and overall survival rates were not significantly different from those achieved by the younger group and were also comparable to historical studies of elderly patients.
Contrary to this finding, a retrospective review of patients older than age 75 treated in Italy between 1987 and 1996 found no benefit in terms of overall survival in patients treated with induction therapy vs low-dose chemotherapy or supportive care.[33] However, for the small number of patients who were able to attain a complete remission, survival was improved. A similar finding was reported by M. D. Anderson, suggesting that patients older than age 80 should not receive standard induction therapy. This is based on a single-institution experience with 30 patients over the age of 80 treated with either an anthracycline alone, an anthracycline plus cytarabine, or fludarabine with cytarabine. The median survival for these patients was a mere 3 to 4 weeks, which compared to a median overall survival of 10 weeks for age-matched controls of a historical cohort who did not receive induction chemotherapy.[34]
Other investigators have suggested that the decision to offer induction chemotherapy should not be based on age alone, but rather on the presence of adverse features predictive of an inferior response to chemotherapy. In a review of over 150 patients with AML over the age of 60 treated with intensive induction chemotherapy, the presence of a complex karyotype was highly predictive of outcome.[35] A median survival of 15 months was attained for patients with a normal karyotype, compared with 4 months for those with a complex karyotype. With a lack of prospective data to guide management of patients over age 75, each patient must be considered individually, with the decision regarding induction chemotherapy based on comorbidities, cytogenetics, and patient preference.
Elderly patients who are considered candidates for standard induction therapy will receive an anthracycline-based regimen, such as daunorubicin at 45 to 60 mg/m2 for 3 days combined with cytarabine at 100 mg/m2 by continuous infusion over 7 days (ie, "7 + 3"). Multiple trials have assessed the utility of combining additional agents, such as thioguanine (Tabloid) or etoposide to the standard anthracycline-based protocol.[19,36] There have been no clear improvements in overall survival when these agents are substituted or combined with cytarabine and an anthracycline.
The optimum choice of anthracycline has also been investigated, and neither mitoxantrone (Novantrone) nor idarubicin has yielded improvements in overall survival for elderly patients when compared to standard doses of daunorubicin.[20] Because of the increased risk of cardiotoxicity with anthracyclines in the elderly, alternative doses of daunorubicin have been explored. A large meta-analysis of over 2,000 patients demonstrated a benefit in terms of complete remission rates and disease-free survival for patients who received a total of at least 90 mg/m2 of daunorubicin.[37] The incidence of early death was identical between the two groups, and thus, there is no definitive benefit associated with reducing the dose of daunorubicin in elderly patients.
Growth Factors
Leukemic blasts in elderly patients with AML are more commonly resistant to standard chemotherapy, as evidenced by inferior remission rates compared to younger cohorts. Growth factor priming in elderly patients with leukemia has been of interest to several groups.[20,38] Both granulocyte colony-stimulating factor (G-CSF [Neupogen]) and granulocyte-macrophage colony-stimulating factor (GM-CSF [Leukine]) have been shown to alter the cell-cycle kinetics of the leukemic blasts, rendering them more susceptible to agents such as cytarabine, which is a cell-cycle dependent cytotoxic drug.[39] In younger patients, the addition of G-CSF to induction chemotherapy with idarubicin and cytarabine followed by amsacrine and cytarabine resulted in significant improvements in disease-free and overall survival.[38] However, a large Eastern Cooperative Oncology Group (ECOG) study revealed no improvements in complete remission or overall survival from GM-CSF priming in elderly AML patients given just prior to induction chemotherapy.[20]
A more common use of growth factors, both in clinical trials and in standard practice, has been in the postchemotherapy stage of treatment. There is generally a 2- to 3-week delay in neutrophil reconstitution following the completion of chemotherapy, and elderly patients in particular are at high risk for severe infection and early death. One complicating factor has been the theoretical concern that growth factor use would stimulate residual leukemia cells to proliferate, thus limiting the effectiveness of induction therapy. Preclinical studies have supported this theory with the finding that G-CSF and GM-CSF can upregulate procaspase protein levels in leukemia cell lines, further promoting cell survival and proliferation.[39] However, several groups have investigated the use of growth factors on various days following the completion of chemotherapy, and there has been no evidence of inferior clinical outcome.[21,36,40-42] With the use of G-CSF or GM-CSF following induction, a 2- to 6-day improvement in neutrophil recovery time can be seen, and in some cases with reduced numbers of severe infections. Only one study to date has reported an improved overall survival with the use of GM-CSF.[41]
Given the current evidence, the use of growth factor support post-induction chemotherapy is unlikely to be harmful and may reduce the number of days the patient remains neutropenic and thus susceptible to severe infections.
Consolidation Chemotherapy
The majority of older adult patients with AML who achieve remission with induction chemotherapy will experience relapse of their disease, with only 10% to 20% of elderly patients achieving long-term survival. Given this dismal prognosis, attempts to reduce the risk of relapse with consolidation chemotherapy, maintenance therapy, and transplant have been explored.
Younger patients with good or intermediate prognosis have been shown to benefit from one to four cycles of high-dose cytarabine, although the optimal number of cycles has not yet been defined.[22] Unfortunately, a standard consolidation regimen in elderly patients has not been identified, owing in part to the resistant nature of their disease and also to their relative intolerance of high-dose chemotherapy.
The Cancer and Leukemia Group B (CALGB) investigated the optimal dose of consolidation cytarabine for adults who achieved completed remission after standard induction therapy.[22] While patients younger than age 60 experienced improved disease-free survival when treated with high-dose cytarabine (3 g/m2), the outcomes for older patients were not significantly improved. This finding was related, in part, to the excessive toxicity of high-dose cytarabine in the elderly cohort. Less than one-third of elderly patients were able to complete all four cycles of consolidation, and reports of significant neurotoxicity were common. As a result, high-dose cytarabine cannot be routinely recommended as consolidation therapy for patients older than age 60.
Strategies utilizing an intermediate dose of cytarabine (1-1.5 g/m2) have shown acceptable tolerability, but with disappointingly low disease-free survival rates.[20,43] Studies using one to three courses of a regimen similar to that used for induction have produced long-term survival rates similar to those achieved with intermediate doses of cytarabine.[19,36,42] Thus, a standard consolidation regimen for elderly patients with AML has not been established, although one to two cycles of intermediate-dose cytarabine or repeated cycles of combination chemotherapy represent reasonable options.
Maintenance Therapy
As an alternative strategy to improve long-term outcomes in elderly patients with AML, various maintenance therapies have been investigated as postremission therapy. Interferon, interleukin (IL)-2 (Proleukin), low-dose cytarabine, and combination chemotherapy are some of the treatments that have been administered for various lengths of time postconsolidation.[20,22,23,33,36,38] Unfortunately, in studies that have compared maintenance therapy to placebo in the elderly, these strategies have been unsuccessful in reducing the risk of relapse or improving overall survival.[22,23,36]
Transplant
Older adults are underrepresented in transplantation trials in AML, making it difficult to predict survival and treatment-related mortality rates. Historically, patients older than 55 to 60 have not been considered for transplant secondary to concerns of early death or debilitating morbidity. Over the past several years, however, there have been substantial changes in transplantation protocols, with the use of more tolerable conditioning regimens, substitution of bone marrow stem cells for peripheral blood stem cells in transplant, and improved supportive care measures that have allowed for a renewed interest in transplantation procedures for the elderly.
Autologous transplantation has been investigated in the elderly, and while tolerable regimens have been reported, high relapse rates and concerns for excessive toxicity remain.[44-46] Thus, this modality is not routinely recommended outside of a clinical trial setting. Outcomes have also been generally poor for ablative allogeneic transplants in elderly patients with AML due to excessive treatment-related mortality.[47]
Somewhat more promising outcomes have been seen with non-myeloblative and reduced-intensity allogeneic regimens.[48-50] Reduced-intensity strategies have been employed to take advantage of a T-cell-mediated graft-vs-leukemia (GVL) effect without the added toxicity of high-dose chemotherapy. In a small study of 19 elderly patients with active leukemia, a reduced-intensity conditioning regimen followed by allogeneic stem cell transplant resulted in an overall 1-year survival of 68% and a 1-year nonrelapse mortality of 22%.[49]
Other nonmyeloablative regimens have produced similarly promising outcomes with relatively low transplant-related mortality and favorable 1-year survival rates.[48,51] However, these allogeneic strategies are associated with significant relapse rates, treatment-related morbidity secondary to graft-vs-host disease, and persistent infections. Thus, allogeneic transplant with nonmyeloablative or reduced-intensity conditioning regimens may be considered on an individual patient basis, preferably in the setting of a clinical trial.
Salvage Chemotherapy
Despite the high frequency of leukemia relapse following induction chemotherapy, elderly patients are rarely candidates for salvage therapy in either the relapsed or refractory setting. For those who are able to receive further treatment, several single and combination chemotherapy regimens have been utilized with no clearly superior strategy.[52-55] Gemtuzumab ozogamicin (Mylotarg) is a humanized monoclonal antibody directed against CD33 that has been approved for the treatment of older adults with AML in first relapse. Although a complete remission rate of 26% has been reported, the agent is associated with significant toxicity, prolonged myelosuppression, and hepatic veno-occlusive disease.[52]
Novel Therapies
Traditional cytotoxic chemotherapy is either unsuccessful at eradicating the malignant leukemic clone or the cause of excessive toxicity in the majority of elderly patients with AML. For this reason, alternative therapies with improved efficacy and favorable toxicity profiles are being actively investigated. Several agents have shown modest activity as single agents in the treatment of AML, including farnesyl transferase inhibitors, nucleoside analogs, FLT-3 inhibitors, histone deacetylase inhibitors, and DNA methyltransferase inhibitors.
Tipifarnib (Zarnestra), an orally bioavailable inhibitor of farnesyl transferase, is an example of one of these drugs that is currently under active investigation. Significant antileukemic activity was confirmed in a multicenter, phase II trial involving elderly patients with untreated poor-risk AML or high-risk MDS who were not otherwise candidates for standard induction therapy.[56] Phase I and II trials with FLT-3 inhibitors, such as CEP-70, PKC, and sunitinib malate (SU11248, Sutent) have also shown activity as single agents, and trials that combine FLT-3 inhibitors with cytotoxic chemotherapy are ongoing. Hypomethylating agents have acquired new interest in recent years, although initial studies with these drugs in leukemia began 3 decades ago.[57]
REFERENCE GUIDE
Therapeutic Agents
Mentioned in This Article
Amsacrine
Azacitidine (Vidaza)
Cytarabine
Daunorubicin
Decitabine (Dacogen)
Etoposide
Fludarabine
Gemtuzumab ozogamicin
(Mylotarg)
Granulocyte colony-stimulating
factor (G-CSF [Neupogen])
Granulocyte-monocyte
colony-stimulating factor
(GM-CSF [Leukine])
Idarubicin
Interferon
Interleukin (IL)-2 (Proleukin)
Mitoxantrone (Novantrone)
Paclitaxel
Sunitinib malate (Sutent)
Thioguanine (Tabloid)
Tipifarnib (Zarnestra)
Brand names are listed in parentheses only if a drug is not available generically and is marketed as no more than two trademarked or registered products. More familiar alternative generic designations may also be included parenthetically.
Azacitidine (Vidaza) is a DNA methyltransferase inhibitor that can cause direct tumor cell cytotoxicty through inhibition of thymidylate synthase and, at lower doses, induces hypomethylation of CpG islands at the promoter regions of tumor-suppressor genes.[58] Small, phase I/II studies in AML or MDS/AML have yielded modest complete remission rates and overall response rates of approximately 50%.[59,60] In the same family of agents is 5-aza-2´-deoxycytidine, or decitabine (Dacogen), which forms an irreversible bond with DNA methyltransferase leading to gene hypomethylation and subsequent reactivation of silenced genes.[58] Decitabine has been investigated in several trials including patients with AML or MDS/AML and has produced overall response rates ranging from 17% to 50%.[61,62] Larger studies are needed to confirm these results, but hypomethylating agents show considerable promise in the treatment of poor-risk AML or AML/MDS who are not candidates for intensive therapy.
Conclusions
AML in the elderly freqently presents with high-risk features that predict resistance to standard cytotoxic chemotherapy. As a result, elderly patients are less likely to attain complete remission following induction chemotherapy and suffer from high rates of relapse compared to younger cohorts. In addition, medical comorbidities in the elderly often preclude the administration of standard doses and regimens of chemotherapy. However, elderly patients who are candidates for induction therapy may benefit from improved survival as compared to those who receive palliative care alone. The decision to offer standard therapies should be based on patient preference, the presence of comorbidities, and prognostic characteristics. Strong consideration should be given to enrollment in clinical trials in order to investigate less toxic and more efficacious regimens for this unique population of patients.
Financial Disclosure:The author has no significant financial interest or other relationship with the manufacturers of any products or providers of any service mentioned in this article.
References:
1. Cohen HJ, Muss HB: The Cancer and Leukemia Group B Cancer in the Elderly Committee: Addressing a major cancer need. Clin Cancer Res 12(11 suppl):3606s-3611s, 2006.
2. Hutchins L, Unger J, Crowley J, et al: Underrepresentation of patients 65 years of age or older in cancer-treatment trials. N Engl J Med 341:2061-2067, 1999.
3. Benson A III, Pregler J, Bean J, et al: Oncologists' reluctance to accrue patients onto clinical trials: An Illinois Cancer Center study. J Clin Oncol 9:2067-2075, 1991.
4. Mengis C, Aebi S, Tobler A, et al: Assessment of differences in patient populations selected for or excluded from participation in clinical phase III acute myelogenous leukemia trials. J Clin Oncol 21:3933-3939, 2003.
5. Vardiman J, Harris N, Brunning R: The World Health Organization (WHO) classification of the myeloid neoplasms. Blood 100:2292-2302, 2002.
6. Slovak M, Kipecky K, Cassileth P, et al: Karyotypic analysis predicts outcome of preremission and postremission therapy in adult acute myeloid leukemia: A Southwest Oncology Group/Eastern Cooperative Oncology Group study. Blood 96:4075-4083, 2000.
7. Grimwade D, Walker H, Harrison G, et al: The predictive value of hierarchal cytogenetic classification in older adults with acute myeloid leukemia (AML): Analysis of 1065 patients entered into the United Kingdom Medical Research Council AML 11 trial. Blood 98:1312-1320, 2001.
8. Applebaum F, Gundacker H, Head D, et al: Age and acute myeloid leukemia. Blood 107:3481-3485, 2006.
9. Pedersen-Bjergaard J, Philip P, Larsen S, et al: Chromosome aberrations and prognostic factors in therapy-related myelodysplasia and acute nonlymphocytic leukemia. Blood 76:1083-1091, 1990.
10. Chin K, Pastav I, Gottesman M: Function and regulation of the human multidrug resistance gene. Adv Cancer Res 60:157-180, 1993.
11. Leith C, Kopecky K, Godwin J, et al: Acute myeloid leukemia in the elderly: Assessment of multidrug resistance (MDR1) and cytogenetics distinguishes biologic subgroups with remarkably distinct responses to standard chemotherapy. A Southwest Oncology Group study. Blood 89:3323-3329, 1997.
12. Thiede C, Steudel C, Mohr B, et al: Analysis of FLT3-activating mutations in 979 patients with acute myelogenous leukemia: Association with FAB subtypes and identification of subgroups with poor prognosis. Blood 99:4326-4335, 2002.
13. Falini B, Mecucci C, Tiacci E, et al: Cytoplasmic nucleophosmin in acute myelogenous leukemia with a normal karyotype. N Engl J Med 352:254-266, 2005.
14. Wilson C, Davidson G, Martin S, et al: Gene expression profiling of adult acute myeloid leukemia identifies novel biologic clusters for risk classification and outcome prediction. Blood 108:685-696, 2006.
15. Yancik R: Cancer burden in the aged: An epidemiologic and demographic overview. Cancer 80:1273-1283, 1997.
16. Piccirillo J, Tierney R, Costas I, et al: Prognostic importance of comrobidity in a hospital-based cancer registry. JAMA 291:2441-2447, 2004.
17. Menzin J, Lang K, Earle C, et al: The outcomes and costs of acute myeloid leukemia among the elderly. Arch Intern Med 162:1597-1603, 2002.
18. Sekeres M, Stone R, Zahrieh D, et al: Decision-making and quality of life in older adults with acute myeloid leukemia or advanced myelodysplastic syndrome. Leukemia 18:809-816, 2004.
19. Anderson J, Kipecky K, Willman C, et al: Outcome after induction chemotherapy for older patients with acute myeloid leukemia is not improved with mitoxantrone and etoposide compared to cytarabine and daunorubicin: A Southwest Oncology Group study. Blood 100:3896-3876, 2002.
20. Rowe J, Neuberg D, Friedenberg W, et al: A phase 3 study of three induction regimens and of priming with GM-CSF in older adults with acute myeloid leukemia: A trial by the Eastern Cooperative Oncology Group. Blood 103:479-485, 2004.
21. Heil G, Hoelzer D, Sanz M, et al: A randomized, double-blind, placebo-controlled, phase III study of filgrastim in remission induction and consolidation therapy for adults with de novo acute myeloid leukemia. Blood 90:4710-4718, 1997.
22. Mayer R, Davis R, Schiffer C, et al: Intensive postremission chemotherapy in adults with acute myeloid leukemia. N Engl J Med 331:896-903, 1994.
23. Preisler H, Davis R, Kirshner J, et al: Comparison of three remission induction regimens and two postinduction strategies for the treatment of acute nonlymphocytic leukemia: A Cancer and Leukemia Group B study. Blood 69:1441-1449, 1987.
24. Buchner T, Urbanitz D, Hiddemann W, et al: Intensified induction and consolidation with or without maintenance chemotherapy for acute myeloid leukemia (AML): Two multicenter studies of the German AML Cooperative Group. J Clin Oncol 3:1583-1589, 1985.
25. Dillmann R, Davis R, Green M, et al: A comparative study of two different doses of cytarabine for acute myeloid leukemia: A phase III trial of the the Cancer and Leukemia Group B. Blood 78:2520-2526, 1991.
26. Vogler W, Velez-Garcia E, Weiner R, et al: A phase III trial comparing idarubicin and daunorubicin combination with cytarabine in acute myelogenous leukemia: A Southeastern Cancer Study Group study. J Clin Oncol 10:1103-1111, 1992.
27. Rees J, Gray R, Wheatley K: Dose intensification in acute myeloid leukaemia: greater effectiveness at lower cost. Principal report of the Medical Research Council's AML9 study. MRC Leukaemia in Adults Working Party. Br J Haematol 94:89-98, 1996.
28. Cassileth P, Harrington D, Appelbaum F, et al: Chemotherapy compared with autologous or allogeneic bone marrow transplantation in the management of acute myeloid leukemia in first remission. N Engl J Med 339:1649-1656, 1998.
29. Sebban C, Archimbaud E, Coiffier B, et al: Treatment of acute myeloid leukemia in elderly patients. A retrospective study. Cancer 61:227-231, 1988.
30. Keating M, McCredie K, Benjamin R, et al: Treatment of patients over 50 years of age with acute myelogenous leukemia with a combination of rubidazone and cytosine arabinoside, vincristine, and prednisone (ROAP). Blood 58:584-591, 1981.
31. Lowenberg B, Zittoun R, Kerkhofs H, et al: On the value of intensive remission-induction chemotherapy in elderly patients of 65+ years with acute myeloid leukemia: A randomized phase III study of the European Organization for Research and Treatment of Cancer Leukemia Group. J Clin Oncol 7:1268-1274, 1989.
32. Vey N, Coso D, Bardou V, et al: The benefit of induction chemotherapy in patients age > or = 75 years. Cancer 101:325-331, 2004.
33. Ferrara F, Annuziata M, Copia C, et al: Therapeutic options and treatment results for patients over 75 years of age with acute myeloid leukemia. Haematologica 83:126-131, 1998.
34. DeLima M, Ghaddar H, Pierce S, et al: Treatment of newly-diagnosed acute myelogenous leukaemia in patients aged 80 years and above. Br J Haematol 93:89-95, 1996.
35. Knipp S, Hildebrandt B, Giagounidis A, et al: Intensive chemotherapy is not recommended for patients with AML or high-risk MDS aged over 60 years with complex karyotype anomalies (abstract 72). Blood 104(11):24a, 2004.
36. Goldstone A, Burnett A, Wheatley K, et al: Attempts to improve treatment outcomes in acute myeloid leukemia (AML) in older patients: The results of the United Kingdom Medical Research Council AML11 trial. Blood 98:1302-1311, 2001.
37. Hiddemann W, Kern W, Schoch C, et al: Management of acute myeloid leukemia in elderly patients. J Clin Oncol 17:3569-3576, 1999.
38. Lowenberg B, Putten W, Theobald M, et al: Effect of priming with granulocyte colony-stimulating factor on the outcome of chemotherapy for acute myeloid leukemia. N Engl J Med 349:743-752, 2003.
39. Faderl S, Harris D, Van Q, et al: Granulocyte-macrophage colony-stimulating factor (GM-CSF) induces antiapoptotic and proapoptotic signals in acute myeloid leukemia. Blood 102:630-637, 2003.
40. Dombret H, Chastang C, Fenaux P, et al: A controlled study of recombinant human granulocyte colony-stimulating factor in elderly patients after treatment for acute myelogenous leukemia. N Engl J Med 332:1678-1683, 1995.
41. Rowe J, Anderson J, Mazza J, et al: A randomized placebo-controlled phase III study of granulocyte-macrophage colony-stimulating factor in adult patients (>55 to 70 years of age) with acute myelogenous leukemia: A study of the Eastern Cooperative Oncology Group (E1490). Blood 86:457-462, 1995.
42. Stone R, Berg D, George S, et al: Granulocyte-macrophage colony-stimulating factor after initial chemotherapy for elderly patients with primary acute myelogenous leukemia. N Engl J Med 332:1671-1677, 1995.
43. Sperr W, Piribauer M, Wimazal F, et al: A novel effective and safe consolidation for patients over 60 years with acute myeloid leukemia: Intermediate dose cytarabine (2 x 1 g/m2 on days 1,3, and 5). Clin Cancer Res 10:3965-3971, 2004.
44. Archimbaud E, Jehn U, Thomas X, et al: Multicenter randomized phase II trial of idarubicin vs mitoxantrone, combined with VP-16 and cytarabine for induction/consolidation therapy, followed by a feasibility study of autologous peripheral blood stem cell transplantation in elderly patients with acute myeloid leukemia. Leukemia 13:843-849, 1999.
45. Gorin N, Labopin M, Pichard P, et al: Feasibility and recent improvement of autologous stem cell transplantation for acute myelocytic leukaemia in patients over 60 years of age: Importance of the source of stem cells. Br J Haematol 110:887-893, 2000.
46. Cahn J, Labopin M, Mandelli F, et al: Autologous bone marrow transplantation for first remission acute myeloblastic leukemia in patients older than 50 years: A retrospective analysis of the European Bone Marrow Transplant Group. Blood 85:575-579, 1995.
47. Wallen H, Gooley T, Deeg H, et al: Ablative allogeneic hematopoietic cell transplantation in adults 60 years of age and older. J Clin Oncol 23:3439-3446, 2005.
48. McSweeney P, Niederwieser D, Shizuru J, et al: Hematopoietic cell transplantation in older patients with hematologic malignancies: Replacing high-dose cytotoxic therapy with graft-vs-tumor effects. Blood 97:3390-3400, 2001.
49. Bertz H, Potthoff K, Finke J: Allogeneic stem-cell transplantation from related and unrelated donors in older patients with myeloid leukemia. J Clin Oncol 21:1480-1484, 2003.
50. Claxton D, Ehmann C, Rybka W: Control of advanced and refractory acute myelogenous leukaemia with sirolimus-based non-myeloablative allogeneic stem cell transplantation. Br J Haematol 130:256-264, 2005.
51. Mohty M, de Lavallade H, Ladaique P, et al: The role of reduced intensity conditioning allogeneic stem cell transplantation in patients with acute myeloid leukemia: A donor vs no donor comparison. Leukemia 19:891-893, 2005.
52. Sievers E, Larson R, Stadtmauer E, et al: Efficacy and safety of gemtuzumab ozogamicin in patients with CD33-positive acute myeloid leukemia in first relapse. J Clin Oncol 19:3244-3254, 2001.
53. Ossenkoppele G, Graveland W, Sonneveld P, et al: The value of fludarabine in addition to ARA-C and G-CSF in the treatment of patients with high-risk myelodysplastic syndromes and AML in elderly patients. Blood 103:2908-2913, 2004.
54. Estey E, Plunkett W, Handhi V, et al: Fludarabine and arabinoxylcytosine therapy of refractory and relapsed acute myelogenous leukemia. Leuk Lymphoma 9:343-350, 1993.
55. Welborn J, Kopecky K, Meyers F, et al: Carboplatin infusion in relapsed and refractory acute myeloid leukemia-a Southwest Onocology Group trial. Leukemia 9:1126-1129, 1995.
56. Lancet J, Gotlib J, Gojo I, et al: Tipifarnib (ZarnestraTM) in previously untreated poor-risk AML of the elderly: Updated results of a mutlicenter phase 2 trial (abstract 874). Blood 104(11):249a, 2004.
57. Vogler W, Miller D, Keller J: 5-Azacytidine (NSC 102816): A new drug for the treatment of myeloblastic leukemia. Blood 48:331-337, 1976.
58. Christman J: 5-Azacytidine and 5-aza-2´-deoxycytine as inhibitors of DNA methylation: Mechanistic studies and their implications for cancer therapy. Oncogene 21:5438-5495, 2002.
59. Gore S, Baylin S, Dauses T, et al: Changes in promoter methylation and gene expression in patients with MDS and MDS-AML treated with 5-azacitidine and sodium phenylbutyrate (abstract 469). Blood 104(11):137a, 2004.
60. Shadduck R, Rossetti J, Faroun Y, et al: AML induction therapy with outpatient azacitidine (abstract 1800). Blood 104(11):499a, 2004.
61. Issa J, Garcia-Manero G, Giles F, et al: Phase I study of low-dose prolonged exposure schedules of the hypomethylating agents 5-aza-2´-deoxycytidine (decitabine) in hematopoietic malignancies. Blood 103:1635-1640, 2004.
62. Kantargian H, Issa JP, Rosenfeld CS, et al: Decitabine improves patient outcomes in myelodysplastic syndromes: results of a phase III randomized study. Cancer 106:1794-1803, 2006.