Progress in the Treatment of Advanced-Stage Classical Hodgkin Lymphoma in the PET-Adapted Era
Introduction
Classical Hodgkin lymphoma (cHL) is a germinal center B-cell cancer, as demonstrated by somatic hypermutation and clonal immunoglobulin gene alterations.1,2 Clinical indications of a more advanced stage of the disease include enlarged lymph nodes and/or systemic symptoms, including high temperatures, sweats at night, or loss of body weight.3 It is classified into lymphocyte-rich variation, lymphocyte-deficient variant, mixed cellularity, and nodular sclerosis.4 The pathognomonic malignant cells linked to cHL, Reed-Sternberg cells, promote ongoing cell division by expressing the transcription component NF-kB.5,6
Epstein-Barr virus is more frequently linked to cHL, which has a bimodal variation in age with an initial peak around age 20 to 30 years and a subsequent peak around age 50 to 70 years. Patients older than 75 years old may be diagnosed with cHL on occasion.7 Advanced-stage cHL (ASHL) stages III or IV are uniquely problematic due to the aggressiveness of the disease and the intensive treatments that need to be delivered. Prognosis in ASHL has undergone significant improvement in recent decades, with the current 5-year survival rate greater than 80%, which can be attributed almost exclusively to improvement in chemoimmunotherapy and radiotherapy.8
The therapy for HL has changed fundamentally since radiotherapy (RT) became the initial modality in the early 20th century. Although RT alone produced excellent cure rates in early-stage disease, it was less successful in advanced stages and was accompanied by considerable long-term toxicities such as secondary malignancies and cardiovascular difficulties.9 The MOPP treatment, which included mechlorethamine hydrochloride, vincristine sulfate (Oncovin), procarbazine, and prednisone, was developed by researchers from the National Cancer Institute in the US in the 1960s and was thought to be able to cure nearly half of patients who had terminal illness.10 Devita et al reported that in the 1970s, their experience with MOPP was 4 years of freedom from treatment failure (FFTF) of 47%.11 MOPP was complicated by a high incidence of secondary malignancies, and the search for a better option continued.
In 1975, Bonadonna et al compared MOPP with drugs including doxorubicin (Adriamycin), bleomycin, vinblastine, and dacarbazine (ABVD) in 45 patients and found no difference in complete remission induction, with much less toxicity with ABVD.12 ABVD gained wide acceptance and became the standard treatment for ASHL. In the 1980s, a regimen called Stanford V was developed and was later compared with ABVD but failed to show superiority.13,14 In 1990, the German Hodgkin Study Group (GHSG) developed baseline bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine (Oncovin), procarbazine, and prednisolone (BEACOPP) (BB) and escalated BEACOPP (EB).15 GHSG’s first major study in ASHL, HD9, was run between 1993 and 1998 and showed that both BB and EB were more effective than COPP/ABVD, with 10-year FFTF for EB vs COPP/ABVD of 82% vs 64% respectively, with an overall survival (OS) difference of 11% in favor of EB.16
The introduction of functional imaging, specifically fluorodeoxyglucose-PET (FDG-PET), has transformed the treatment of HL. Interim PET, done after 2 cycles of chemotherapy (PET2), has become an effective predictor of early response to treatment. PET2 negativity, as a Deauville score of 1 to 3, is predictive of much-improved outcomes in comparison with PET2 positivity (Deauville 4-5).17 For example, in the RATHL trial, patients with negative PET2 had a 3-year progression-free survival (PFS) rate of 85.7% compared with 67.5% for those with positive PET2.18
The prognostic value of PET2 has been validated in a number of randomized trials and is now a foundation stone in current treatment schemes for ASHL. By identifying those patients who will respond to optimal therapy and those requiring escalated treatment, PET2 allows the treatment approach to become more individualized. PET-adapted approaches are founded on the risk stratification principle. Patients who are low-risk who have a negative PET2 can be de-escalated, thereby minimizing long-term toxicities. Patients who are PET2 positive are high-risk and can be considered for intensification of treatment to enhance outcomes. This strategy not only maximizes therapeutic benefit but also avoids exposing patients to toxic agents unnecessarily, as those patients are likely to go into remission with less intensive therapy.
For example, the HD12 study aimed to reduce EB from 8 cycles (8EB) to 4EB + 4BB and to omit RT because 70% of patients in the HD9 study got RT. HD12 found that a reduction in chemotherapy dose did not reduce toxicity substantially but caused some decrease in efficacy.19 The HD15 study proved 6EB to be more effective and less toxic than 8EB and limited RT to PET-positive residual lesions of more than 2.5 cm.20 Moreover, in the HD18 trial, those with a negative PET2 after 2 escalated BEACOPP cycles were randomly assigned to treatment with 6 or 8 cycles. It was seen that de-escalation to 4 cycles did not undermine results, with 5-year PFS at 92.2% in the de-escalated arm.21 Similarly, in the phase 3 AHL2011 trial (NCT01358747), patients who were PET2 negative were de-escalated to ABVD and attained a 5-year PFS of 85.4% with less toxicity than with standard BEACOPP.22
While GHSG studies reported both PFS and OS benefits of EB compared with ABVD, other studies did not find an OS benefit and even questioned EB’s PFS benefit, and were more concerned about its toxicity.23-25 After Ganesan et al published their phase 2 study results,26 many other randomized studies were published.
PET-adapted randomized studies aimed to find the best first-line regimen and to adjust treatment after 2 cycles according to PET results to maximize efficacy in patients who are less responsive while reducing toxicity in patients with satisfactory responses. Thus, the objective of this review was to assess the role of PET-adapted treatments in improving outcomes in patients with ASHL. By summarizing important randomized trials and discussing their implications, this review seeks to offer an overview of current treatment paradigms and underscore directions for the future management of this disease.
Materials and Methods
Search Strategy
This review aimed to provide an answer to this question: How have PET-adapted treatment strategies advanced the treatment of advanced-stage cHL, and what treatment algorithms have been established based on randomized trials? To answer this question, this review performed an extensive search of existing research studies, clinical trials, and systematic reviews on PET-adapted treatment approaches in ASHL. In this study, electronic databases including PubMed, Web of Science, ScienceDirect, Google Scholar, Scopus, and the Cochrane Library were used to search for relevant data. This research was designed to examine the use, effectiveness, and future perspectives of PET-adapted approaches in ASHL with the goal of maximizing treatment effects while reducing toxicity. The search terms used included the following: “Hodgkin lymphoma,” “advanced-stage Hodgkin lymphoma,” “PET-adapted treatment,” “interim PET,” “PET2,” “ABVD,” “BEACOPP,” “Deauville score,” “treatment escalation,” “treatment de-escalation,” “progression-free survival,” “overall survival,” “randomized controlled trials,” “biomarkers in Hodgkin lymphoma,” “FDG-PET,” and “risk-adapted therapy.” Boolean operators (AND, OR) and proximity operators (NEAR, NEXT, WITHIN) were applied to narrow down the search. The search was restricted to articles published between 2000 and 2024 to guarantee the inclusion of the latest and most applicable studies. Table 1 summarizes the search terms and databases utilized.
TABLE 1. Search Terms and Databases
Search Process
The search methodology used for this review included structured phases to provide a thorough and systematic process. A general search was first done across chosen databases, including PubMed, Web of Science, ScienceDirect, Google Scholar, Scopus, and the Cochrane Library, using defined keywords such as “Hodgkin lymphoma,” “PET-adapted treatment,” “interim PET,” and “BEACOPP.” Titles, abstracts, and index keywords were filtered to assess the relevance of the articles. After the first search, a secondary search that was more specific to identify keywords, Medical Subject Headings terms, and MEDLINE index phrases was conducted.
More studies were drawn from the included articles’ reference lists to guarantee that no important studies were excluded. Articles with titles and abstracts considered to be relevant were obtained for full-text assessment, and those satisfying the eligibility criteria were included in the review. Key data, including study design, patient population, treatment regimens, PET2 results, PFS, OS, and toxicity profiles, were taken from the chosen studies. This review was performed by one author, and although all possible efforts were made to incorporate the most important and pertinent studies, it should be noted that this article is a review and possibly does not include all the complexities of PET-adapted treatment approaches in ASHL.
Eligibility Criteria
The studies were selected using the following inclusion and exclusion criteria:
Inclusion Criteria
- Studies that focused on PET-adapted treatment approaches in ASHL were selected.
- Inclusion criteria included randomized controlled trials, phase 2/3 clinical trials, or systematic reviews.
- Studies that had reported outcomes of PFS, OS, and toxicity profiles were selected.
- Studies that used PET2 as a predictive measure of treatment response were included.
- Studies that reported PET2-guided treatment escalation or de-escalation were included.
- Studies published between 2000 to 2024 and written in English were selected.
Exclusion Criteria
- Studies were excluded if any of the following were true:
- They were published in languages other than English.
- They did not include PET-adapted approaches or advanced-stage Hodgkin lymphoma.
- They did not provide adequate data or evidence to warrant their conclusions.
- They were editorials, opinion pieces, or case reports with no original data.
- They were entirely about early-stage Hodgkin lymphoma or non-Hodgkin lymphoma.
Results
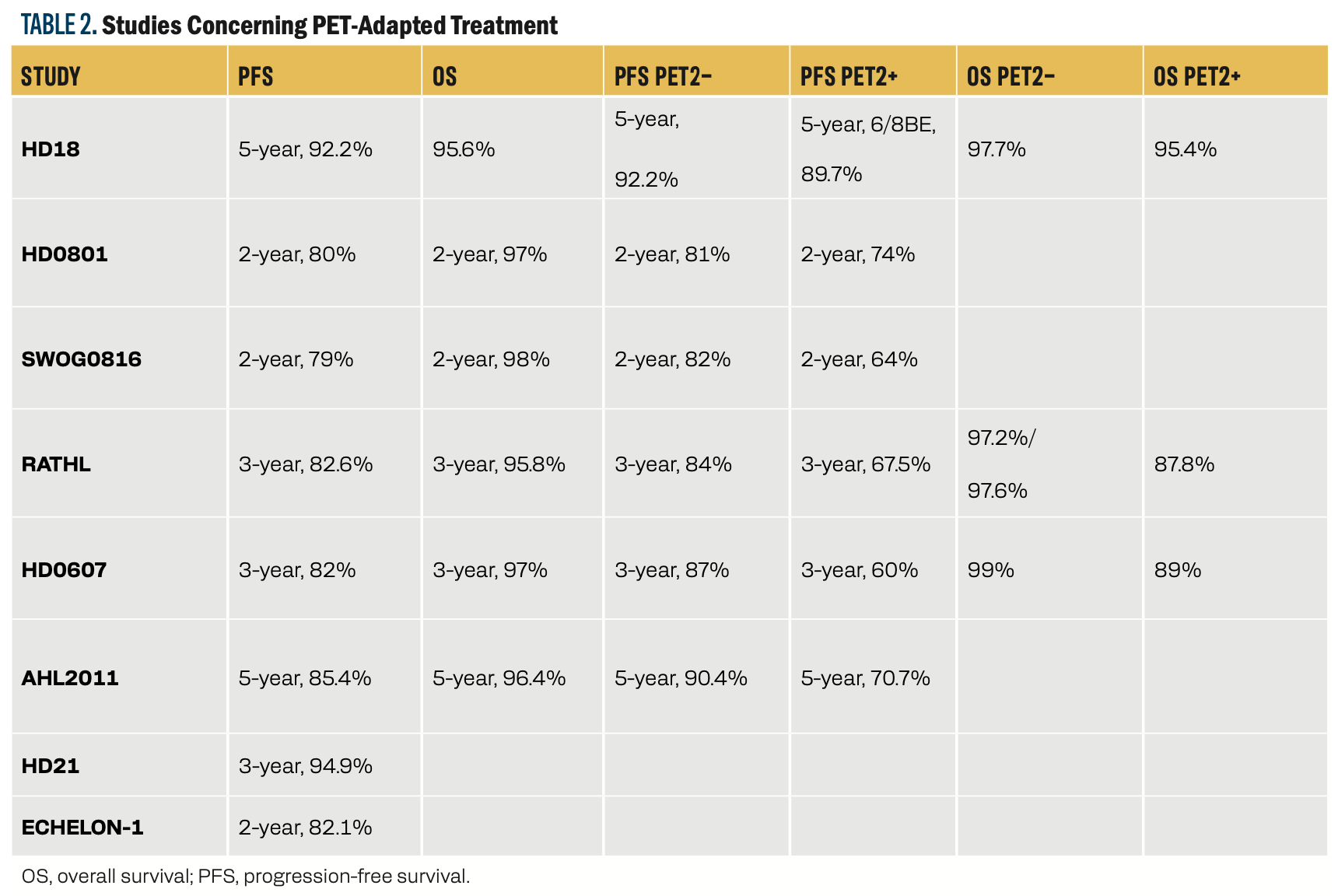
In the literature, some randomized controlled trials have evaluated PET-adapted treatment strategies in advanced-stage cHL, with the resultant treatment algorithms that maximize outcomes (Table 2). The findings from the important trials are detailed below.
TABLE 2. Studies Concerning PET-Adapted Treatment
PET-Guided Treatment De-Escalation
RATHL (2016, Updated 2022)
The phase 3 RATHL trial (NCT00678327) enrolled 1203 patients with stage III/IV cHL between 2008 and 2012. All patients underwent 2 cycles of ABVD and PET2, 83.7% of whom were PET2 negative (Deauville 1-3) and 16.3% of whom were PET2 positive (Deauville 4-5). Patients who were PET2 negative were randomly assigned to receive either 4 cycles of ABVD or AVD (ABVD without bleomycin), and patients who were PET2 positive were randomly assigned to receive 6 cycles of baseline BB or 4 cycles of escalated EB. The 3-year PFS was 85.7% in patients who were PET2 negative receiving ABVD and 84.4% in those receiving AVD, with no difference in efficacy. The PFS at 3 years for patients who were PET2 positive was 67.5%, with no difference between EB and BB. These findings were confirmed by long-term follow-up in 2022, underlining the lessened lung toxicity of AVD.27,28
Key findings: AVD is equivalent to ABVD in patients who are PET2 negative with less lung toxicity. Patients who are PET2 positive are improved by escalation of treatment, although more optimization for this high-risk subgroup remains desirable.
AHL2011 (2018, Updated 2022)
Casasnovas et al enrolled 823 patients with advanced-stage cHL from 2011 through 2014 on either 6 cycles of escalated BEACOPP (EB) or a PET-adapted arm.22,29 In the PET-adapted arm, patients received 2 cycles of EB followed by PET2. Patients who were PET2 negative were de-escalated to 4 cycles of ABVD, and patients who were PET2 positive were given 4 more cycles of EB. A total of 87% of the patients were PET2 negative, and 13% were PET2 positive. The 5-year PFS was comparable in the standard EB arm (86.7%) and PET-adapted arm (85.4%). Follow-up in 2022 reinforced the long-term efficacy and safety of the PET-adapted approach.
Key findings: PET2-directed de-escalation to ABVD in early responders preserves activity with less toxicity. It can be an active alternative to normal EB, especially in PET2-negative patients.
PET-Guided Treatment Escalation
HD0801 (2016)
Zinzani et al conducted a phase 2 trial (NCT00784537) in 512 patients with advanced-stage cHL from 2008 to 2013. All were given 2 cycles of ABVD and PET2. Among these, 80% were PET2 negative (Deauville 1-2) and 20% were PET2 positive (Deauville 3-5). Patients who were PET2 negative were given 4 further cycles of ABVD, while those who were PET2 positive were changed to 4 cycles of ifosfamide, gemcitabine, and vinorelbine (IGEV) with the possibility of autologous stem cell transplant (ASCT).30 The 2-year PFS was 81% for patients who were PET2 negative and 74% for PET2 positive treated with IGEV. The trial proved that treatment intensification early on with ASCT is in favor of patients who were PET2 positive, and it underscores the value of PET2 in determining therapy.
Key findings: Patients who werePET2 positive have improved outcomes with early intensification of treatment, eg, ASCT. PET2-guided therapy enables individualized treatment, maximizing efficacy with minimal unnecessary toxicity in PET2-negative patients.
SWOG 0816 (2016, Updated)
Press et al assessed 336 patients in this phase 2 trial (NCT00822120) with high-stage cHL from 2009 through 2012. Two cycles of ABVD were followed by PET2 in patients.31 Patients who were PET2 negative were continued with an additional 4 cycles of ABVD, and patients who were PET2 positive (Deauville 4-5) were escalated to 6 cycles of escalated BEACOPP (EB). Out of all patients, 82% were PET2 negative and 18% were PET2 positive. The PFS at 2 years was 82% in patients who were PET2 negative and 64% in PET2 positive. A reanalysis reported 5-year PFS of 76% and 66%, respectively.32
Key findings: EB escalation is beneficial for patients who are PET2 positive vs continuation of ABVD. PET2-guided escalation is an important strategy for patients who are high-risk, but improvement is still required for this subgroup.
HD18 (2017, Updated 2021)
Borchmann et al evaluated 1945 patients in a phase 3 trial (NCT00515554) with advanced-stage cHL between 2008 and 2014. Patients were administered 2 cycles of escalated BEACOPP (EB) plus a PET2 scan.21 Patients who were positive on the PET2 scan were assigned to 6 or 8 cycles of EB or 6 cycles of EB with rituximab. Patients who were PET2 negative were assigned randomly to receive either 4 or 6 cycles of EB. Five years later, survival rates in patients who were PET2 positive and PET2 negative who received 4 cycles were 89.7% and 92.2%, respectively. The efficacy and safety of the shorter 4-cycle EB regimen were reaffirmed by a 2021 follow-up.33
Key findings: Four cycles of EB can be safely administered to patients who are PET2 negative instead of 6 cycles without sacrificing efficacy, but lowering toxicity. Rituximab does not enhance outcomes in patients who are PET2 positive.
HD0607 (2018)
In this phase 2 trial (NCT00795613), Gallamini et al studied 782 patients with advanced-stage cHL from 2008 to 2014. Patients were treated with 2 cycles of ABVD and PET2. Patients who were PET2 negative received 4 more cycles of ABVD, and those who were PET2 positive were escalated to 4 cycles of escalated BEACOPP (EB) with or without rituximab. Among the patients, 81% were PET2 negative and 19% were PET2 positive. The 3-year PFS was 87% and 60%, respectively.34
Key findings: Escalation from PET2 to EB is effective and possible for patients who are high-risk. Yet, outcomes in patients who are PET2 positive remain poor, suggesting the urgent need for new treatments.
Novel Therapeutic Approaches
ECHELON-1 Trial (2018, Updated 2022)
Despite the fact that the phase 3 ECHELON-1 (NCT01712490) trial was not a PET-adapted treatment study, it is judicious to mention it here. It was published in 2018.35 The study was run between 2012 and 2016 and included 1334 patients with ASHL, and they were assigned to either ABVD or brentuximab-vedotin, doxorubicin, vinblastine, and dacarbazine (A-AVD). The 3-year PFS was 83.1% with A-AVD and 76.0% with ABVD (P = .005). Researchers did not adjust treatment after PET2, but they reported that more patients treated with A-AVD than those treated with ABVD had negative results at PET2 (89% vs 86%) and that 3-year PFS rates in patients who were PET2 negative and younger than 60 years were 87.2% for A-AVD vs 81.0% for ABVD. A beneficial trend in patients who were PET2positive and younger than 60 years on A-AVD was also observed, with a 3-year PFS rate of 69.2% vs 54.7% with ABVD.36 An updated analysis of ECHELON-1 at a median follow up of 6 years estimated OS to be 93.9% and 89.4% for A-AVD and ABVD, respectively, demonstrating a clear OS benefit with A-AVD vs ABVD, which is why many consider A-AVD as the standard of care for patients with ASHL.37
Key findings: A-AVD is superior in efficacy over ABVD, especially in patients who are PET2 positive, and is an increasingly popular first-line treatment choice.
HD21 Trial (2023, ASCO Update)
The phase 3 HD21 trial (NCT02661503) used brentuximab vedotin (BV) in up-front treatment. It was run between July 2016 and August 2020.38,39 A total of 1482 patients were treated with either EB or brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin, dacarbazine, and dexamethasone (BrECADD)—a modified EB incorporating BV—which was found to be effective and safer compared with EB, with 3-year PFS of 92.3% for EB and 94.9% for BrECADD. The latest data presented at the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting confirmed the superiority of BrECADD over EB with a 4-year PFS of 94.3 vs 90.9 and a 4-year OS of 98.5 vs 98.2, respectively. Most importantly, treatment-associated toxicity was significantly reduced when using BrECADD, evidenced by less gonadal toxicity and fewer infections.40
Key findings: BrECADD is safer and more effective than BEACOPP and can be a potential alternative for advanced-stage cHL.
SWOG1826 Trial (2023, ASCO Update)
Immune checkpoint inhibitors have also been evaluated in first-line treatments: The phase 3 SWOG S1826 study (NCT03907488) assessed 976 patients with ASHL from July 9, 2019, to October 5, 2022, to receive either nivolumab (N)-AVD or BV-AVD. The 1-year PFS was 94% for patients treated with N-AVD and 86% for those who received BV-AVD. With a median follow-up of 2.1 years, the 2-year PFS was 92% with N-AVD, compared with 83% with BV+AVD (HR, 0.45).41,42
Key findings: Nivolumab-based treatment can outperform BV-AVD as a standard of care by providing better PFS for advanced-stage cHL.
Discussion
cHL has previously been managed using standardized chemotherapy protocols, eg, ABVD and BEACOPP, irrespective of response in individual patients.17,43,44 Although these regimens have produced high rates of survival, they are related to considerable toxicity and late complications, such as secondary malignancies, cardiopulmonary disease, and infertility.43
PET-adapted strategies have transformed the treatment of advanced-stage cHL by allowing personalized treatment adjustment according to early response evaluation. PET2 response assessment, undertaken after 2 cycles of chemotherapy, has been confirmed as an excellent prognostic indicator.17,45 Patients with a negative PET2 result (Deauville score 1-3) have outstanding survival rates, permitting treatment reduction with a view to reducing toxicity. On the other hand, patients with positive PET2 scans (Deauville score 4-5) are at increased risk of treatment failure, and, thus, treatment intensification is required to enhance disease control.27
Some randomized controlled trials have addressed the effectiveness of PET-adapted approaches, optimizing treatment pathways to optimize efficacy and toxicity. This review interprets the outcomes from major trials and assesses the effect of de-escalation, escalation, and innovative therapeutic strategies on patient outcomes.
PET-Guided Treatment De-Escalation
One of the most important advantages of PET-adapted therapy is the potential to de-escalate the treatment in patients who are PET2 negative without affecting survival. Various important trials, such as RATHL, AHL2011, and HD18, have proven de-escalation to be a safe strategy for these patients.
The RATHL trial evaluated whether bleomycin could be safely omitted in PET2-negative patients to reduce lung toxicity.27,28 Among 1203 patients, 83.7% were PET2 negative and randomly assigned to ABVD or AVD after 2 initial ABVD cycles. Three-year PFS was equivalent (85.7% vs 84.4%), but AVD resulted in fewer severe lung toxicities.
In the AHL2011 trial, 823 patients received 2 cycles of escalated BEACOPP (EB) followed by PET2.22,30 Patients with negative scans (87%) switched to ABVD, while positive scans (13%) continued EB. Five-year survival was comparable between PET-adapted and standard EB (85.4% vs 86.7%), showing PET2-negative patients can safely transition to ABVD.
The HD18 trial studied 1945 patients, with PET2-negative individuals assigned to 4 or 6 cycles of BEACOPP.21,34 Five-year PFS was 92.2% for 4 cycles and 90.8% for 6, confirming that reducing treatment did not compromise survival but lowered toxicity. These observations make PET-directed de-escalation a normative treatment method, minimizing the toxicity of chemotherapy while preserving cure rates in PET2-negative individuals.
PET-Guided Treatment Escalation
In patients who are PET2 positive, who have a greater chance of relapse, escalation of treatment is necessary to enhance survival. Treatment escalation has been proven to improve the control of disease through several trials, such as SWOG0816, HD0607, and HD18.
The SWOG0816 trial included 336 patients treated with 2 ABVD cycles followed by PET2.32,33 PET2-negative patients (82%) continued ABVD, while PET2-positive patients (18%) escalated to BEACOPP. At 5 years, disease-free survival was 76% vs 66%, showing escalation improves outcomes in high-risk patients.
In addition, the HD0607 trial of 782 patients, PET2 after 2 ABVD cycles guided treatment.35 PET2-negative patients (81%) remained on ABVD, while PET2-positive patients (19%) received BEACOPP ± rituximab. Three-year disease-free survival was 87% for PET2-negative vs 60% for PET2-positive, underscoring the value of early intensification for high-risk patients.
However, a critical subgroup of patients who are PET2 positive, known as slow responders, has been recognized in AHL2011 and HD0801. In AHL2011, 62 patients who were PET2 positive following 2 escalated BEACOPP (EB) cycles became PET2 negative following 2 further escalated BEACOPP (EB) cycles, with no modification of treatment.22 Likewise, 15 patients who were PET2 positive in HD0801 remained on ABVD without regimen change, and 73% attained complete remission.30 This indicates that a proportion of patients who were PET2 positive might later respond without escalation, with issues surrounding overtreatment risk to some patients. With these findings, the issue of how best to modify PET2-based escalation criteria to minimize unnecessary toxicity to patients who will ultimately remain responsive with continued therapy is critical.
Emerging Therapeutic Paradigms: Novel Agents and Immunotherapy
Although PET-adapted chemotherapy continues to be the standard of treatment, new targeted agents and immunotherapy are altering first-line management strategies. The ECHELON-1 trial showed substituting bleomycin with brentuximab vedotin (BV-AVD) improved 3-year PFS (83.1% vs 76.0% for ABVD).36,37 BV-AVD has become an alternate standard of treatment in patients with newly diagnosed cHL.
The HD21 trial brought forth BrECADD, a BEACOPP variant that includes brentuximab vedotin.41 This regimen was more effective, with a 4-year PFS of 94.3% vs 90.9% for BEACOPP, with much less toxicity.
The SWOG1826 trial discovered that N-AVD was superior to BV-AVD, as 92% of patients were still cancer-free at 2 years, vs 83% with BV-AVD.42,43 This indicates that immune checkpoint inhibitors may be an optimal therapy for cHL.
In addition to efficacy, cost and long-term adverse effects are the most important considerations in treatment selection. A comparison of regimens placed AHL2011 at the top in terms of cost-effectiveness, weighing efficacy and safety.46 Because BEACOPP has severe adverse effects, more recent treatments such as brentuximab vedotin and immune checkpoint inhibitors are considered safer alternatives.
The SWOG1826 study proved that the nivolumab-based treatments are capable of outclassing BV-AVD in first-line treatment. Apart from this, Allen et al claimed 100% PFS and OS for first-line sequential pembrolizumab-AVD following a 33.1-month follow-up.47
Combination regimens have also been explored, such as brentuximab vedotin, nivolumab, doxorubicin, and dacarbazine (AN-AD). A phase 2 trial found an 88.3% 2-year PFS in patients who were treatment-naive with cHL, which indicates that BV plus immune checkpoint inhibitors may be an emerging first-line treatment option.48 These data further indicate the expanding use of immunotherapy in optimizing cHL treatment with reduced toxicity.
Strengths and Limitations
PET-adapted therapy approaches have optimized the treatment of stage III/IV cHL by allowing for tailoring therapy. Treatment de-escalation in patients who are PET2 negative can be performed safely, with minimal unnecessary toxicity and still high survival rates, as shown in the RATHL, AHL2011, and HD18 trials.21,22,27 At the same time, treatment intensification favors patients who are PET2 positive, achieving better disease control and survival benefits, as documented in SWOG0816 and HD0607.31,34 PET-adapted treatment has been consistently proven in a series of randomized controlled trials, and it has become a robust and evidence-supported standard of treatment for cHL.
Though it has many benefits, PET-adapted therapy also has some disadvantages. First, the PET scan interpretation inter-reader variability could result in variable treatment plans.44 Second, 10% to 20% of patients who are PET2 negative eventually relapse, thus necessitating further biomarkers such as TARC and gene expression profiling for better risk stratification.48,49 Lastly, patients who are positive on PET2 scans are still at risk, even for more intensive therapy like BEACOPP. This points to the necessity of novel treatment modalities like brentuximab vedotin and immune checkpoint inhibitors.41 There is a potential for future research to address more effective methods for evaluating risk and utilizing biomarkers to make informed treatment choices so that Hodgkin lymphoma management can be more specific and effective.
Future Directions
Future developments in PET-adapted therapy need to be directed toward more accurately pinpointing high-risk patients through combining PET2 imaging with other biomarkers such as TARC, CD68, CD163, and IL-10.48,49 Because PET2 is not always a useful marker, some of these additional markers can make treatment decisions more precise. High levels of TARC (> 800 pg/mL) were associated with worse outcomes independently of PET2 status in the HD0607 trial.49 Other candidate markers, such as patterns of gene expression, soluble CD68, CD163, and IL-10, are being explored in an attempt to enhance the earlier detection of patients at risk.50,51 Future trials must incorporate biomarker-directed treatment algorithms with PET to maximize patient selection for de-escalation or escalation.
New treatments like nivolumab, brentuximab vedotin, and combination immunotherapy must be tested to optimize outcomes in PET2-positive patients. Lastly, long-term follow-up is crucial to evaluate late toxicities of PET-adapted de-escalation so that modification of treatment will not compromise long-term survival or enhance risks of secondary malignancies.
Conclusion
PET-adapted therapy has improved the management of advanced-stage Hodgkin lymphoma through individualized treatment. Individuals with an excellent initial response (PET2 negative) can be treated with reduced intensity to avoid adverse effects, while those with a poor initial response (PET2 positive) are treated with intensified therapy for better outcomes. Research such as RATHL, AHL2011, and HD18 corroborates this, but there are issues, including relapse and accuracy of the scan. New blood tests and new drugs could further tailor treatment options. Long-term monitoring is essential to prevent a reduction in treatment intensity from leading to increased risk factors such as later health issues.
Author Information
Wael Abdulla Moh Khair, MD
Phone: +966 12 731 0800
Email: alhajyousif@gmail.com
Statements and Declarations
Funding
The author did not receive any funding.
Conflict of interest
There are no conflicts of interest to report.
Acknowledgments
None
References
- Küppers R, Rajewsky K, Zhao M, et al. Hodgkin disease: Hodgkin and Reed-Sternberg cells picked from histological sections show clonal immunoglobulin gene rearrangements and appear to be derived from B cells at various stages of development. Proc Natl Acad Sci U S A. Nov 8 1994;91(23):10962-6. doi:10.1073/pnas.91.23.10962
- Lim SH, Johnson PWM. Optimizing therapy in advanced-stage Hodgkin lymphoma. Blood. 2018;131(15):1679-1688. doi:10.1182/blood-2017-09-772640
- Gini G, Cimminiello M, Galieni P, et al. Advanced Stage Hodgkin Lymphoma: Patient Management. Acta Biomed. May 25 2020;91(S-5):5-12. doi:10.23750/abm.v91iS-5.9913
- Wang HW, Balakrishna JP, Pittaluga S, Jaffe ES. Diagnosis of Hodgkin lymphoma in the modern era. Br J Haematol. Jan 2019;184(1):45-59. doi:10.1111/bjh.15614
- Greaves P, Clear A, Coutinho R, et al. Expression of FOXP3, CD68, and CD20 at diagnosis in the microenvironment of classical Hodgkin lymphoma is predictive of outcome. J Clin Oncol. Jan 10 2013;31(2):256-62. doi:10.1200/jco.2011.39.9881
- Liu Y, Abdul Razak FR, Terpstra M, et al. The mutational landscape of Hodgkin lymphoma cell lines determined by whole-exome sequencing. Leukemia. Nov 2014;28(11):2248-51. doi:10.1038/leu.2014.201
- Brice P, de Kerviler E, Friedberg JW. Classical Hodgkin lymphoma. Lancet. Oct 23 2021;398(10310):1518-1527. doi:10.1016/s0140-6736(20)32207-8
- Eichenauer DA, Aleman BMP, André M, et al. Hodgkin lymphoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. Oct 1 2018;29(Suppl 4):iv19-iv29. doi:10.1093/annonc/mdy080
- Patel CG, Michaelson E, Chen Y-H, et al. Reduced Mortality Risk in the Recent Era in Early-Stage Hodgkin Lymphoma Patients Treated With Radiation Therapy With or Without Chemotherapy. International Journal of Radiation Oncology, Biology, Physics. 2018;100(2):498-506. doi:10.1016/j.ijrobp.2017.09.048
- Dann EJ, Casasnovas R-O. Treatment Strategies in Advanced-Stage Hodgkin Lymphoma. Cancers. 2024;16(11):2059.
- Devita VT, Jr., Serpick AA, Carbone PP. Combination chemotherapy in the treatment of advanced Hodgkin's disease. Ann Intern Med. Dec 1970;73(6):881-95. doi:10.7326/0003-4819-73-6-881
- Bonadonna G, Zucali R, Monfardini S, De Lena M, Uslenghi C. Combination chemotherapy of Hodgkin's disease with adriamycin, bleomycin, vinblastine, and imidazole carboxamide versus MOPP. Cancer. Jul 1975;36(1):252-9. doi:10.1002/1097-0142(197507)36:1<252::aid-cncr2820360128>3.0.co;2-7
- Gordon LI, Hong F, Fisher RI, et al. Randomized phase III trial of ABVD versus Stanford V with or without radiation therapy in locally extensive and advanced-stage Hodgkin lymphoma: an intergroup study coordinated by the Eastern Cooperative Oncology Group (E2496). J Clin Oncol. Feb 20 2013;31(6):684-91. doi:10.1200/jco.2012.43.4803
- Supit T, Amin Z. A Case Report: ABVD versus Stanford V Regimen in Unfavorable Classical Hodgkin's Lymphoma. Indonesian Journal of Cancer. 10/01 2013;7:159-166.
- Diehl V, Behringer K. Could BEACOPP be the new standard for the treatment of advanced Hodgkin's lymphoma? Cancer Invest. Jun-Jul 2006;24(4):461-5. doi:10.1080/07357900600705789
- Engert A, Diehl V, Franklin J, et al. Escalated-dose BEACOPP in the treatment of patients with advanced-stage Hodgkin's lymphoma: 10 years of follow-up of the GHSG HD9 study. J Clin Oncol. Sep 20 2009;27(27):4548-54. doi:10.1200/jco.2008.19.8820
- Gallamini A, Hutchings M, Rigacci L, et al. Early interim 2-[18F]fluoro-2-deoxy-D-glucose positron emission tomography is prognostically superior to international prognostic score in advanced-stage Hodgkin's lymphoma: a report from a joint Italian-Danish study. J Clin Oncol. Aug 20 2007;25(24):3746-52. doi:10.1200/jco.2007.11.6525
- Johnson P, Federico M, Kirkwood A, et al. Adapted Treatment Guided by Interim PET-CT Scan in Advanced Hodgkin’s Lymphoma. New England Journal of Medicine. 2016;374(25):2419-2429. doi:doi:10.1056/NEJMoa1510093
- Borchmann P, Haverkamp H, Diehl V, et al. Eight cycles of escalated-dose BEACOPP compared with four cycles of escalated-dose BEACOPP followed by four cycles of baseline-dose BEACOPP with or without radiotherapy in patients with advanced-stage hodgkin's lymphoma: final analysis of the HD12 trial of the German Hodgkin Study Group. J Clin Oncol. Nov 10 2011;29(32):4234-42. doi:10.1200/jco.2010.33.9549
- Engert A, Haverkamp H, Kobe C, et al. Reduced-intensity chemotherapy and PET-guided radiotherapy in patients with advanced stage Hodgkin's lymphoma (HD15 trial): a randomised, open-label, phase 3 non-inferiority trial. Lancet. May 12 2012;379(9828):1791-9. doi:10.1016/s0140-6736(11)61940-5
- Borchmann P, Goergen H, Kobe C, et al. PET-guided treatment in patients with advanced-stage Hodgkin's lymphoma (HD18): final results of an open-label, international, randomised phase 3 trial by the German Hodgkin Study Group. Lancet. Dec 23 2017;390(10114):2790-2802. doi:10.1016/s0140-6736(17)32134-7
- Casasnovas RO, Bouabdallah R, Brice P, et al. PET-adapted treatment for newly diagnosed advanced Hodgkin lymphoma (AHL2011): a randomised, multicentre, non-inferiority, phase 3 study. Lancet Oncol. Feb 2019;20(2):202-215. doi:10.1016/s1470-2045(18)30784-8
- Carde P, Karrasch M, Fortpied C, et al. Eight Cycles of ABVD Versus Four Cycles of BEACOPPescalated Plus Four Cycles of BEACOPPbaseline in Stage III to IV, International Prognostic Score ≥ 3, High-Risk Hodgkin Lymphoma: First Results of the Phase III EORTC 20012 Intergroup Trial. J Clin Oncol. Jun 10 2016;34(17):2028-36. doi:10.1200/jco.2015.64.5648
- Merli F, Luminari S, Gobbi PG, et al. Long-Term Results of the HD2000 Trial Comparing ABVD Versus BEACOPP Versus COPP-EBV-CAD in Untreated Patients With Advanced Hodgkin Lymphoma: A Study by Fondazione Italiana Linfomi. J Clin Oncol. Apr 10 2016;34(11):1175-81. doi:10.1200/jco.2015.62.4817
- Viviani S, Zinzani PL, Rambaldi A, et al. ABVD versus BEACOPP for Hodgkin's lymphoma when high-dose salvage is planned. N Engl J Med. Jul 21 2011;365(3):203-12. doi:10.1056/NEJMoa1100340
- Ganesan P, Rajendranath R, Kannan K, et al. Phase II study of interim PET-CT-guided response-adapted therapy in advanced Hodgkin's lymphoma. Ann Oncol. Jun 2015;26(6):1170-1174. doi:10.1093/annonc/mdv077
- Johnson P, Federico M, Kirkwood A, et al. Adapted Treatment Guided by Interim PET-CT Scan in Advanced Hodgkin's Lymphoma. N Engl J Med. Jun 23 2016;374(25):2419-29. doi:10.1056/NEJMoa1510093
- Luminari S, Fosså A, Trotman J, et al. Long Follow-up of the Response-Adjusted Therapy for Advanced Hodgkin Lymphoma (RATHL) Trial (CRUK/07/033) Confirms the Safety of Both De-Escalation and Intensification of Chemotherapy. Blood. 11/15 2022;140:766-767. doi:10.1182/blood-2022-162780
- Casasnovas R-O, Bouabdallah R, Brice P, et al. Positron Emission Tomography–Driven Strategy in Advanced Hodgkin Lymphoma: Prolonged Follow-Up of the AHL2011 Phase III Lymphoma Study Association Study. Journal of Clinical Oncology. 01/06 2022;40doi:10.1200/JCO.21.01777
- Zinzani PL, Broccoli A, Gioia DM, et al. Interim Positron Emission Tomography Response-Adapted Therapy in Advanced-Stage Hodgkin Lymphoma: Final Results of the Phase II Part of the HD0801 Study. J Clin Oncol. Apr 20 2016;34(12):1376-85. doi:10.1200/jco.2015.63.0699
- Press OW, Li H, Schöder H, et al. US Intergroup Trial of Response-Adapted Therapy for Stage III to IV Hodgkin Lymphoma Using Early Interim Fluorodeoxyglucose-Positron Emission Tomography Imaging: Southwest Oncology Group S0816. J Clin Oncol. Jun 10 2016;34(17):2020-7. doi:10.1200/jco.2015.63.1119
- Stephens DM, Li H, Schöder H, et al. Five-year follow-up of SWOG S0816: limitations and values of a PET-adapted approach with stage III/IV Hodgkin lymphoma. Blood. Oct 10 2019;134(15):1238-1246. doi:10.1182/blood.2019000719
- Kreissl S, Goergen H, Buehnen I, et al. PET-guided eBEACOPP treatment of advanced-stage Hodgkin lymphoma (HD18): follow-up analysis of an international, open-label, randomised, phase 3 trial. Lancet Haematol. Jun 2021;8(6):e398-e409. doi:10.1016/s2352-3026(21)00101-0
- Gallamini A, Tarella C, Viviani S, et al. Early Chemotherapy Intensification With Escalated BEACOPP in Patients With Advanced-Stage Hodgkin Lymphoma With a Positive Interim Positron Emission Tomography/Computed Tomography Scan After Two ABVD Cycles: Long-Term Results of the GITIL/FIL HD 0607 Trial. J Clin Oncol. Feb 10 2018;36(5):454-462. doi:10.1200/jco.2017.75.2543
- Connors JM, Jurczak W, Straus DJ, et al. Brentuximab Vedotin with Chemotherapy for Stage III or IV Hodgkin's Lymphoma. N Engl J Med. Jan 25 2018;378(4):331-344. doi:10.1056/NEJMoa1708984
- Straus DJ, Długosz-Danecka M, Alekseev S, et al. Brentuximab vedotin with chemotherapy for stage III/IV classical Hodgkin lymphoma: 3-year update of the ECHELON-1 study. Blood. Mar 5 2020;135(10):735-742. doi:10.1182/blood.2019003127
- Graham P. C, Stephen M. A, David J. S, et al. Improved overall survival in stage III/IV classical Hodgkin lymphoma: An updated analysis of ECHELON-1. 2023:
- Eichenauer DA, Plütschow A, Kreissl S, et al. Incorporation of brentuximab vedotin into first-line treatment of advanced classical Hodgkin's lymphoma: final analysis of a phase 2 randomised trial by the German Hodgkin Study Group. The Lancet Oncology. 2017;18(12):1680-1687. doi:10.1016/S1470-2045(17)30696-4
- Borchmann P, Moccia A, Greil R, et al. Treatment Related Morbidity in Patients with Classical Hodgkin Lymphoma: Results of the Ongoing, Randomized Phase III HD21 Trial By the German Hodgkin Study Group. Blood. 2022;140(Supplement 1):771-773. doi:10.1182/blood-2022-165917
- Borchmann P, Moccia AA, Greil R, et al. BRECADD IS NON-INFERIOR TO EBEACOPP IN PATIENTS WITH ADVANCED STAGE CLASSICAL HODGKIN LYMPHOMA: EFFICACY RESULTS OF THE GHSG PHASE III HD21 TRIAL. Hematological Oncology. 2023;41(S2):881-882. doi: 10.1002/hon.3196_LBA5
- Herrera AF, LeBlanc M, Castellino SM, et al. NIVOLUMAB(N)-AVD IMPROVES PROGRESSION-FREE SURVIVAL COMPARED TO BRENTUXIMAB VEDOTIN(BV)-AVD IN ADVANCED STAGE (AS) CLASSIC HODGKIN LYMPHOMA (HL): RESULTS OF SWOG S1826. Hematological Oncology. 2023;41(S2):33-35. doi: 10.1002/hon.3163_5
- Herrera AF, LeBlanc M, Castellino SM, et al. Nivolumab+AVD in Advanced-Stage Classic Hodgkin’s Lymphoma. New England Journal of Medicine. 2024;391(15):1379-1389. doi:doi:10.1056/NEJMoa2405888
- Oki Y, Chuang H, Chasen B, et al. The prognostic value of interim positron emission tomography scan in patients with classical Hodgkin lymphoma. Br J Haematol. Apr 2014;165(1):112-6. doi:10.1111/bjh.12715
- Hutchings M, Loft A, Hansen M, et al. FDG-PET after two cycles of chemotherapy predicts treatment failure and progression-free survival in Hodgkin lymphoma. Blood. Jan 1 2006;107(1):52-9. doi:10.1182/blood-2005-06-2252
- Moghbel MC, Mittra E, Gallamini A, et al. Response Assessment Criteria and Their Applications in Lymphoma: Part 2. J Nucl Med. Jan 2017;58(1):13-22. doi:10.2967/jnumed.116.184242
- Vijenthira A, Chan K, Cheung MC, Prica A. Cost-effectiveness of first-line treatment options for patients with advanced-stage Hodgkin lymphoma: a modelling study. Lancet Haematol. Feb 2020;7(2):e146-e156. doi:10.1016/s2352-3026(19)30218-2
- Allen PB, Lu X, Chen Q, et al. Sequential pembrolizumab and AVD are highly effective at any PD-L1 expression level in untreated Hodgkin lymphoma. Blood Adv. Jun 27 2023;7(12):2670-2676. doi:10.1182/bloodadvances.2022008116
- Viviani S, Mazzocchi A, Pavoni C, et al. Early serum TARC reduction predicts prognosis in advanced-stage Hodgkin lymphoma patients treated with a PET-adapted strategy. Hematol Oncol. Oct 2020;38(4):501-508. doi:10.1002/hon.2775
- Khair W. Is thymus and activation-regulated chemokine a forgotten diagnostic and minimal residual disease marker in classical Hodgkin lymphoma? memo - Magazine of European Medical Oncology. 2021/12/01 2021;14(4):406-411. doi:10.1007/s12254-021-00753-x
- Hsi ED, Li H, Nixon AB, et al. Serum levels of TARC, MDC, IL-10, and soluble CD163 in Hodgkin lymphoma: a SWOG S0816 correlative study. Blood. Apr 18 2019;133(16):1762-1765. doi:10.1182/blood-2018-08-870915
- Steidl C, Diepstra A, Lee T, et al. Gene expression profiling of microdissected Hodgkin Reed-Sternberg cells correlates with treatment outcome in classical Hodgkin lymphoma. Blood. Oct 25 2012;120(17):3530-40. doi:10.1182/blood-2012-06-439570
Highlighting Insights From the Marginal Zone Lymphoma Workshop
Clinicians outline the significance of the MZL Workshop, where a gathering of international experts in the field discussed updates in the disease state.